- +86-0533-2185556
- WhatsApp: +86 15965530500
- admin@hangyubiotech.com
Your Location:Home >Products >Pharmaceutical intermediates >76549-02-5
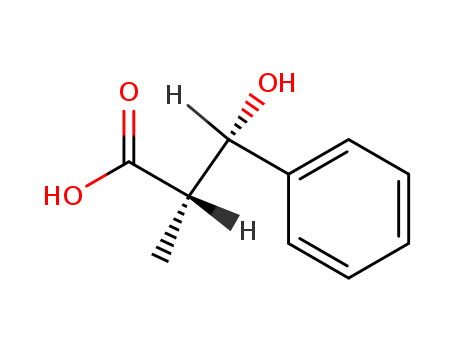

Purity:99%
The synthesis of cis- and trans-2-oxo-2-...
A new non-cross-linked polystyrene suppo...
The catalytic asymmetric aldol reaction ...
Sorting tags are oligomeric structures t...
A systematic examination of enolboration...
The first catalytic asymmetric hetero-Di...
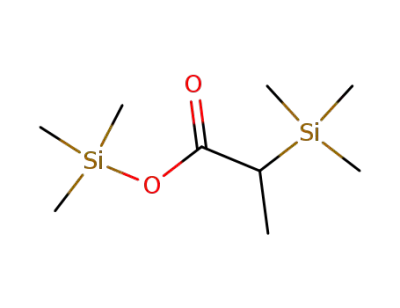
2-(Trimethylsilyl)propansaeure-trimethylsilylester

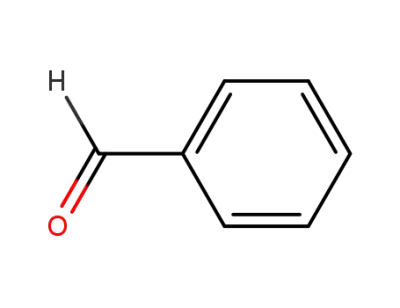
benzaldehyde

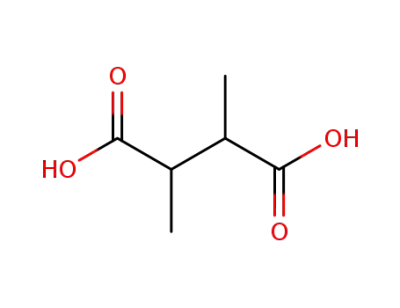
2,3-dimethylsuccinic acid

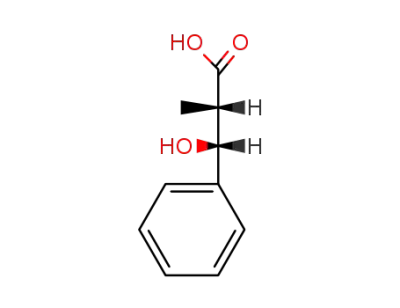
(2RS, 3SR)-3-hydroxy-2-methyl-3-phenylpropanoic acid

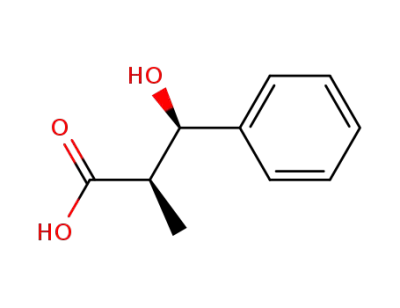
syn-3-hydroxy-2-methyl-3-phenylpropanoic acid
| Conditions | Yield |
|---|---|
|
Product distribution;
other catalysts discussed;
|
![(S)-3-((2S,3S)-3-hydroxy-2-methyl-3-phenylpropionyl)-4-[4-(2-{2-[2-(2-methoxyethoxy)ethoxy]ethoxy}ethoxy)benzyl]oxazolidin-2-one](/upload/2026/5/0b89f4d3-6872-4aae-a1f9-18672dd61d24.png)
(S)-3-((2S,3S)-3-hydroxy-2-methyl-3-phenylpropionyl)-4-[4-(2-{2-[2-(2-methoxyethoxy)ethoxy]ethoxy}ethoxy)benzyl]oxazolidin-2-one

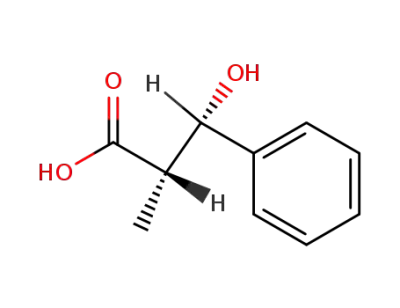
(-)-(2S,3S)-3-hydroxy-2-methyl-3-phenylpropanoic acid
| Conditions | Yield |
|---|---|
|
With
dihydrogen peroxide; lithium hydroxide;
In
tetrahydrofuran; water;
at 0 - 20 ℃;
for 2h;
|
91% |
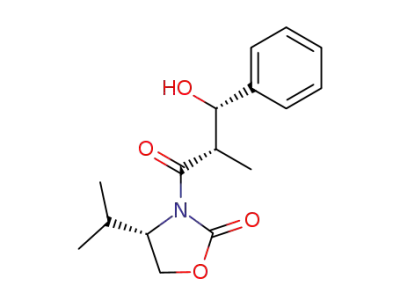
(4S)-3-((2S,3S)-3-hydroxy-2-methyl-1-oxo-3-phenylpropyl)-4-(1-methylethyl)-2-oxazolidinone
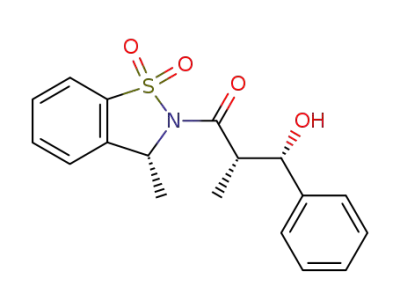
(2S,3S)-3-Hydroxy-2-methyl-1-((R)-3-methyl-1,1-dioxo-1,3-dihydro-1λ6-benzo[d]isothiazol-2-yl)-3-phenyl-propan-1-one
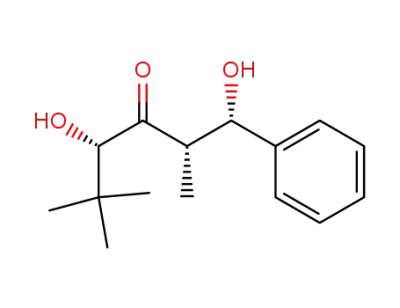
(1S,2S,4S)-1,4-dihydroxy-2,5,5-trimethyl-1-phenyl-3-hexanone
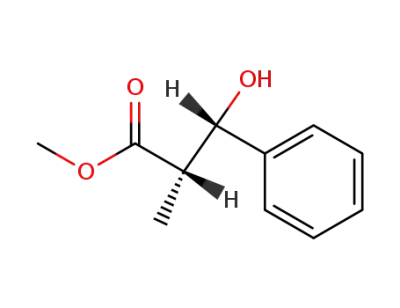
methyl (2S,3S)-3-hydroxy-2-methyl-3-phenylpropanoate

methyl (2S,3S)-3-hydroxy-2-methyl-3-phenylpropanoate
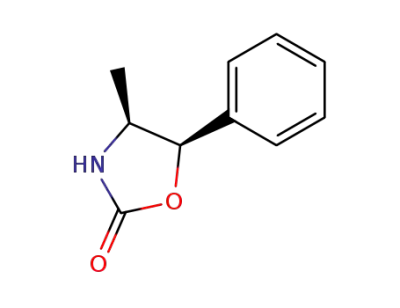
(4S,5R)-4-methyl-5-phenyl-oxazolidin-2-one
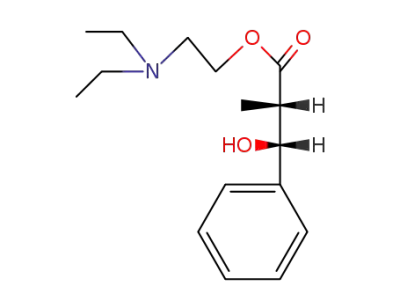
(2RS,3SR)-3-hydroxy-2-methyl-3-phenyl-propionic acid-(2-diethylamino-ethyl ester)
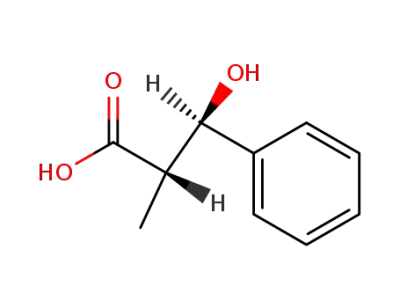
(+)-(2S,3R)-3-hydroxy-2-methyl-3-phenylpropanoic acid
CAS:110802-84-1
CAS:78303-71-6
CAS:907204-31-3
CAS:103694-84-4