- +86-0533-2185556
- WhatsApp: +86 15965530500
- admin@hangyubiotech.com
Your Location:Home >Products >Customization >83321-23-7

Purity:99%
5-Oxo-6,8,11,14-eicosatetraenoic acid (5...
The present invention provides a novel p...
5-Oxo-6,8,11,14-eicosatetraenoic acid (5...
The present disclosure provides processe...
![[1-(hydroxymethyl)cyclopropyl]methanol](/upload/2026/5/f6350ce0-61bd-46ae-879d-fafcb204bcdb.png)
[1-(hydroxymethyl)cyclopropyl]methanol

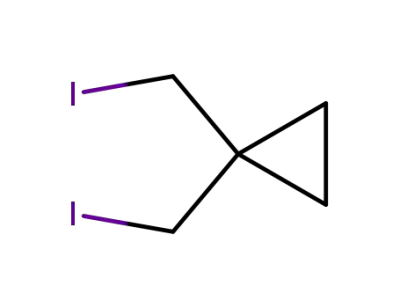
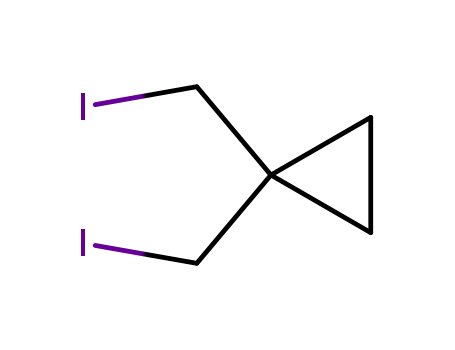
1,1-bis(iodomethyl)cyclopropane
| Conditions | Yield |
|---|---|
|
With
1H-imidazole; iodine; triphenylphosphine;
In
dichloromethane;
at 0 - 20 ℃;
for 2.5h;
|
96% |
|
With
chloro-trimethyl-silane; sodium iodide;
In
acetonitrile;
for 16h;
Ambient temperature;
|
80% |
|
With
1H-imidazole; iodine; triphenylphosphine;
In
dichloromethane;
at 0 - 10 ℃;
for 2.5h;
|
70% |
|
With
1H-imidazole; iodine; triphenylphosphine;
In
dichloromethane;
at 10 ℃;
for 3h;
|
70% |
|
With
1H-imidazole; iodine; triphenylphosphine;
In
dichloromethane;
at 0 - 15 ℃;
for 3.5h;
|
46% |
|
Multi-step reaction with 2 steps
2: acetone; sodium iodide
With
acetone; sodium iodide;
|
|
|
Multi-step reaction with 2 steps
1: triethylamine / acetone / 3 h / 0 - 20 °C
2: sodium iodide / acetone / 35 °C
With
triethylamine; sodium iodide;
In
acetone;
|
|
|
With
1H-imidazole; iodine; triphenylphosphine;
In
chlorosulfonic acid;
at -10 - 20 ℃;
for 3h;
Inert atmosphere;
|
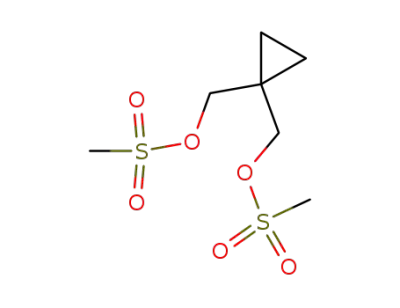
cyclopropane-1,1-diylbis(methylene) dimethanesulfonate


1,1-bis(iodomethyl)cyclopropane
| Conditions | Yield |
|---|---|
|
With
sodium iodide;
In
acetone;
at 35 ℃;
|
94% |
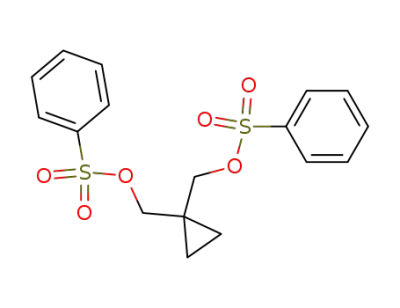
1,1-bis-benzenesulfonyloxymethyl-cyclopropane
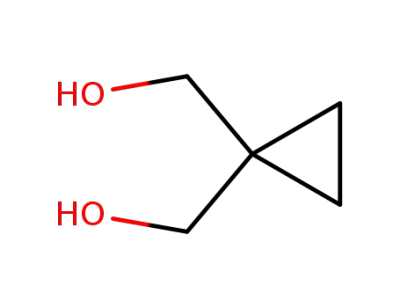
[1-(hydroxymethyl)cyclopropyl]methanol
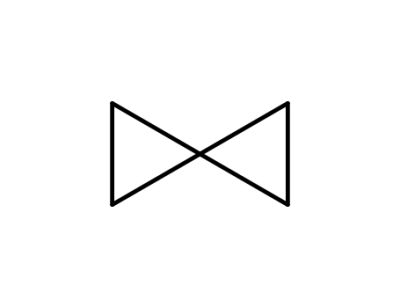
spiropentane
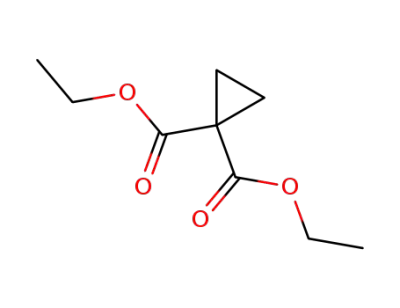
diethyl cyclopropane-1,1-dicarboxylate
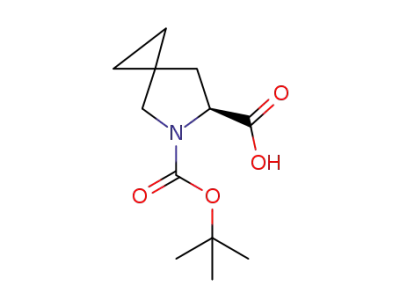
(6S)-5-[(tert-butoxy)carbonyl]-5-azaspiro[2.4]heptane-6-carboxylic acid
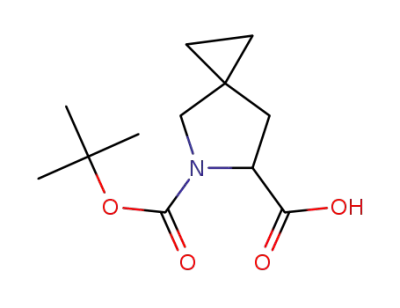
(±)-5-(tert-butoxycarbonyl)-5-azaspiro[2.4]heptane-6-carboxylic acid
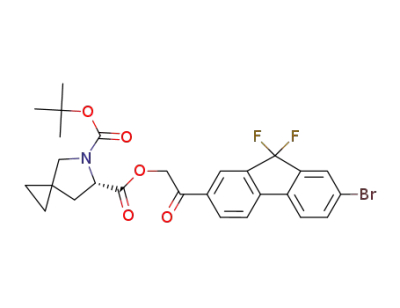
(S)-6-(2-(7-bromo-9,9-difluoro-9H-fluoren-2-yl)-2-oxoethyl) 5-tert-butyl 5-azaspiro[2.4]heptane-5,6-dicarboxylate
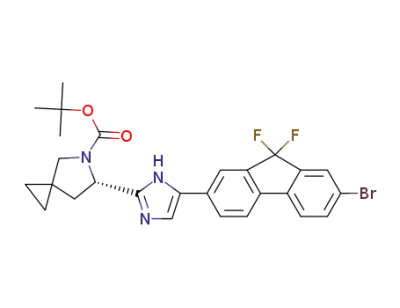
(6S)-6-[5-(7-bromo-9,9-difluoro-9H-fluoren-2-yl)-1H-imidazol-2-yl]-5-azaspiro[2.4]heptane-5-carboxylic acid 1,1-dimethylethyl ester
CAS:1861-48-9
CAS:905579-51-3
CAS:58069-82-2