- +86-0533-2185556
- WhatsApp: +86 15965530500
- admin@hangyubiotech.com
Your Location:Home >Products >Inhibitor >1224713-90-9
Purity:99%
Transthyretin (TTR) is a homotetrameric ...
A series of novel resveratrol derivative...
The present invention relates to Stilben...
The invention relates to the use of hydr...
![5-[(1E)-2-(4-bromophenyl)ethenyl]-1,3-dimethoxybenzene](/upload/2026/5/cdaf5730-9c72-493c-969e-882d5281a96a.png)
5-[(1E)-2-(4-bromophenyl)ethenyl]-1,3-dimethoxybenzene

![5-[(1E)-2-(4-bromophenyl)ethenyl]-1,3-benzenediol](/upload/2026/5/9ae1cd12-8e0e-4ea3-9c2a-5036cbd4dd43.png)
5-[(1E)-2-(4-bromophenyl)ethenyl]-1,3-benzenediol
| Conditions | Yield |
|---|---|
|
With
boron tribromide;
In
dichloromethane;
at -20 - 20 ℃;
Inert atmosphere;
|
73% |
|
5-[(1E)-2-(4-bromophenyl)ethenyl]-1,3-dimethoxybenzene;
With
boron tribromide;
In
dichloromethane;
at 0 - 20 ℃;
With
water;
In
dichloromethane;
Cooling with ice;
|
70% |
|
With
boron tribromide;
In
dichloromethane;
at 0 - 22 ℃;
for 4h;
Inert atmosphere;
|
70% |
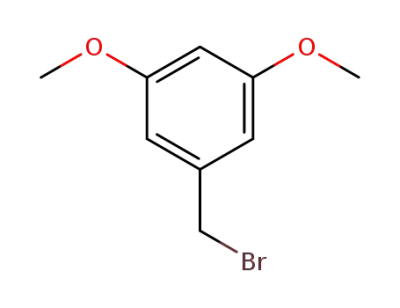
3,5-dimethoxybenzyl bromide

![5-[(1E)-2-(4-bromophenyl)ethenyl]-1,3-benzenediol](/upload/2026/5/9ae1cd12-8e0e-4ea3-9c2a-5036cbd4dd43.png)
5-[(1E)-2-(4-bromophenyl)ethenyl]-1,3-benzenediol
| Conditions | Yield |
|---|---|
|
Multi-step reaction with 3 steps
1.1: 10 h / 120 °C
2.1: sodium methylate / N,N-dimethyl-formamide / 0.5 h / 0 °C / Inert atmosphere
2.2: 20 °C / Inert atmosphere
3.1: boron tribromide / dichloromethane / -20 - 20 °C / Inert atmosphere
With
sodium methylate; boron tribromide;
In
dichloromethane; N,N-dimethyl-formamide;
|
|
|
Multi-step reaction with 2 steps
1.1: triethyl phosphite / neat (no solvent) / 4 h / 150 °C / Inert atmosphere
1.2: 0.33 h / 0 °C / Inert atmosphere
1.3: 0 - 22 °C / Inert atmosphere
2.1: boron tribromide / dichloromethane / 4 h / 0 - 22 °C / Inert atmosphere
With
boron tribromide; triethyl phosphite;
In
dichloromethane;
1.1: |Wittig Olefination / 1.2: |Wittig Olefination / 1.3: |Wittig Olefination;
|
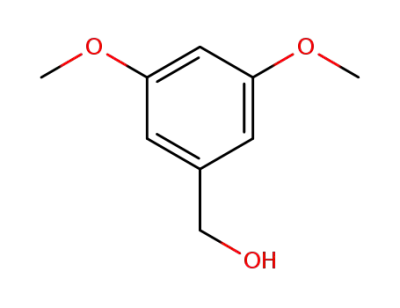
3,5-dimethoxybenzyl alcohol

3,5-dimethoxybenzyl bromide
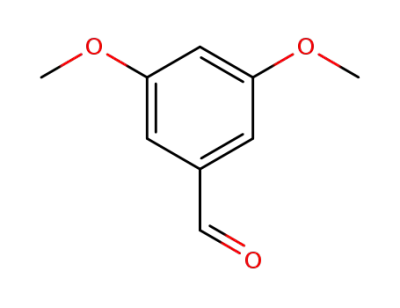
3,5-dimethoxybenzaldehdye
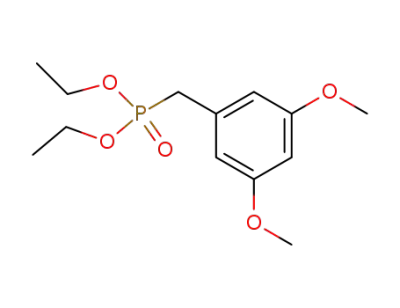
diethyl 3,5-dimethoxybenzylphosphonate
(4-[(E)-2-(3,5-dihydroxyphenyl)vinyl]phenyl)-boronic acid
CAS:56-94-0
CAS:1000018-10-9
CAS:2264-01-9
CAS:1338575-28-2