- +86-0533-2185556
- WhatsApp: +86 15965530500
- admin@hangyubiotech.com
Your Location:Home >Products >API >14527-51-6
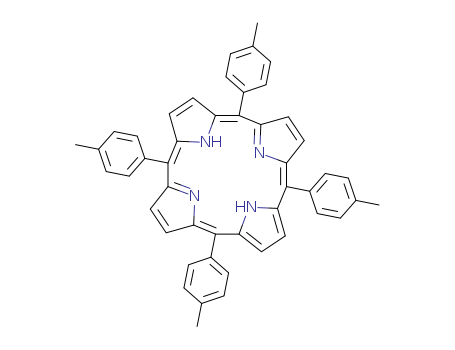
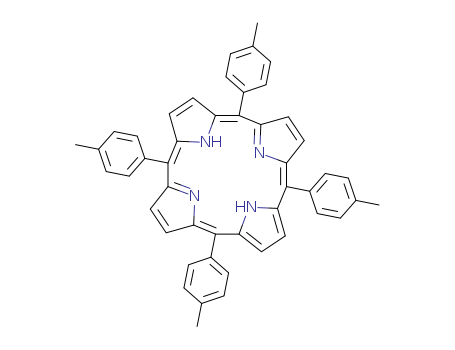
pd_meltingpoint:>300℃
Purity:99%
InChI:InChI=1/C48H38N4/c1-29-5-13-33(14-6-29)45-37-21-23-39(49-37)46(34-15-7-30(2)8-16-34)41-25-27-43(51-41)48(36-19-11-32(4)12-20-36)44-28-26-42(52-44)47(40-24-22-38(45)50-40)35-17-9-31(3)10-18-35/h5-28,49-50H,1-4H3/b45-37-,45-38-,46-39-,46-41-,47-40-,47-42-,48-43-,48-44-
The synthesis of meso-tetraarylporphyrin...
A simple method has been employed to syn...
The first direct synthesis of A3-type un...
The Cu(II) and Ag(II) complexes of tetra...
Photodynamic therapy (PDT) is a noninvas...
Thiaporphyrins with one, two and four un...
Several different synthetic approaches e...
Hybrid materials that link light capture...
A new porous silicate, FSM-16 with a por...
Structurally controlled aggregation cour...
A series of meso-furyl porphyrins with f...
NOBF4 reacts with para-substituted meso-...
[MediaObject not available: see fulltext...
-
Addition of one of a variety of salts to...
meso-Tetraphenylporphyrin syntheses from...
This paper is aimed to investigate the c...
The syntheses of diboryl porphyrin compl...
Synthesis and crystal structure analysis...
In place of widely used dichloromethane,...
We have synthesized a porphyrin-anthraqu...
We investigated the preparation of tetra...
The synthesis of new directly azobenzene...
Two types of cobalt porphyrins, viz., me...
The direct sources of aliphatic acids in...
The synthesis of dipyrromethanes from py...
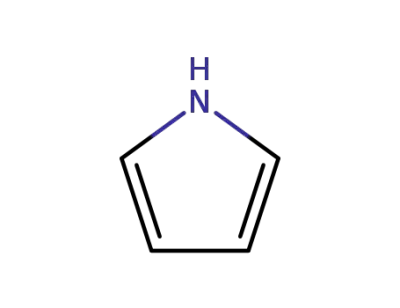
pyrrole

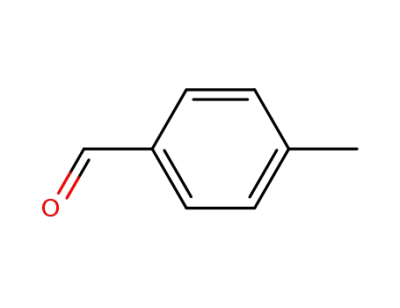
4-methyl-benzaldehyde


5,10,15,20-tetra(p-tolyl)porphyrin
| Conditions | Yield |
|---|---|
|
With
propionic acid;
In
diethyl ether;
at 60 ℃;
for 0.5h;
|
70% |
|
pyrrole; 4-methyl-benzaldehyde;
With
Tulsion-63 resin; Triethyl orthoacetate;
In
dichloromethane;
at 20 ℃;
for 16h;
With
chloranil;
In
dichloromethane;
for 2h;
Heating;
|
68% |
|
In
propionic acid;
for 5h;
Reflux;
|
51% |
|
With
vanadium(V) oxychloride;
In
dichloromethane;
for 1h;
Ambient temperature;
other reagents: TiCl4, VO(OEt)Cl2, VO(OiPr)Cl2, Mn(OAc)3;
|
50% |
|
In
propionic acid; nitrobenzene;
for 0.75h;
Reflux;
|
45% |
|
pyrrole; 4-methyl-benzaldehyde;
In
ethylbenzene; propionic acid;
at 159.6 ℃;
for 0.166667h;
under 1596.11 Torr;
Large scale;
Green chemistry;
With
oxygen;
In
ethylbenzene; propionic acid;
for 0.166667h;
Large scale;
Green chemistry;
|
42.2% |
|
pyrrole; 4-methyl-benzaldehyde;
With
propionic acid;
In
ethylbenzene;
at 159.6 ℃;
for 0.166667h;
under 1596.11 Torr;
With
oxygen;
In
ethylbenzene;
for 0.166667h;
Reflux;
|
41.3% |
|
With
air; chloroacetic acid;
In
xylene;
for 1.5h;
Heating;
|
39% |
|
With
propionic acid; propionic acid anhydride;
|
35% |
|
With
propionic acid;
for 2h;
Reflux;
|
33.4% |
|
4-methyl-benzaldehyde;
With
benzoic acid;
at 200 ℃;
for 0.0833333h;
under 2736.18 Torr;
pyrrole;
at 200 ℃;
for 0.333333h;
|
30% |
|
With
boron trifluoride diethyl etherate;
In
dichloromethane;
for 1h;
Heating;
|
27% |
|
pyrrole; 4-methyl-benzaldehyde;
With
boron trifluoride diethyl etherate;
In
dichloromethane;
at 20 ℃;
for 0.5h;
With
selenium(IV) oxide;
In
dichloromethane;
for 1h;
|
23% |
|
With
propionic acid;
at 120 ℃;
for 1h;
Reagent/catalyst;
|
21% |
|
at 200 ℃;
for 0.25h;
|
20% |
|
In
propionic acid;
for 0.5h;
Heating;
|
19.1% |
|
pyrrole; 4-methyl-benzaldehyde;
With
trifluoroacetic acid;
In
dichloromethane;
for 72h;
Reflux;
With
triethylamine; 2,3-dicyano-5,6-dichloro-p-benzoquinone;
In
dichloromethane;
at 20 ℃;
for 1h;
|
8% |
|
With
propionic acid;
In
nitrobenzene;
at 120 ℃;
for 1h;
|
|
|
pyrrole; 4-methyl-benzaldehyde;
With
trifluoroacetic acid;
In
dichloromethane;
at 20 ℃;
for 1h;
With
2,3-dicyano-5,6-dichloro-p-benzoquinone;
In
dichloromethane;
|
39 % Spectr. |
|
In
propionic acid; nitrobenzene;
at 120 ℃;
for 1h;
|
|
|
With
propionic acid;
for 30h;
Heating;
|
|
|
With
chloranil; trifluoroacetic acid;
Yield given. Multistep reaction;
1) CH2Cl2, 2 h, r.t., 2) 1 h, reflux;
|
|
|
With
chloranil;
aluminum oxide; FSM-16(2.8 nm);
Yield given. Multistep reaction;
1) CH2Cl2, r.t., 1 h, 2) CH2Cl2, 45 deg C, 1 h;
|
|
|
|
|
|
With
trifluoroacetic acid;
In
xylene;
for 1.5h;
Heating;
|
|
|
|
|
|
With
trifluoroacetic acid;
In
dichloromethane; water;
for 2h;
|
|
|
In
propionic acid;
Heating;
|
|
|
With
propionic acid;
Heating;
|
|
|
With
propionic acid;
for 2h;
Reflux;
|
|
|
In
propionic acid;
Reflux;
|
|
|
With
propionic acid;
for 12h;
Reflux;
|
|
|
In
propionic acid;
for 0.5h;
Reflux;
|
|
|
With
propionic acid;
for 0.5h;
Reflux;
|
|
|
With
propionic acid;
for 0.5h;
Reflux;
|
|
|
With
hydrogenchloride;
In
methanol; water;
for 1h;
Reflux;
Inert atmosphere;
|
|
|
With
propionic acid;
for 1h;
Reflux;
|
|
|
With
propionic acid;
Reflux;
|
|
|
In
propionic acid;
at 145 ℃;
for 2h;
Inert atmosphere;
Darkness;
|
|
|
With
propionic acid;
at 145 ℃;
for 2h;
Inert atmosphere;
Darkness;
|
|
|
With
propionic acid;
at 141 ℃;
for 0.5h;
|
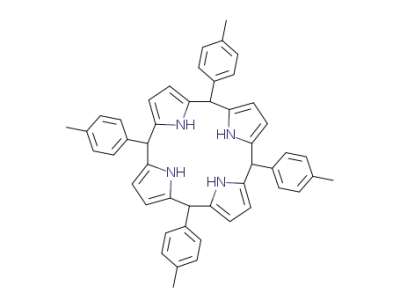
5,10,15,20-Tetra-p-tolyl-porphyrinogene


5,10,15,20-tetra(p-tolyl)porphyrin
| Conditions | Yield |
|---|---|
|
With
chloranil;
In
tetrahydrofuran; water;
Yield given;
|
|
|
With
oxygen; 2,3-dicyano-5,6-dichloro-p-benzoquinone;
iron(II) phthalocyanine μ-oxo dimer;
In
dichloromethane;
for 2h;
|
660 mg |
|
With
air;
In
dichloromethane;
at 39 ℃;
for 4h;
|
|
|
With
air;
In
dichloromethane;
at 39 ℃;
|
|
|
With
air;
In
dichloromethane;
at 39 ℃;
for 3h;
|
|
|
for 1.5h;
UV-irradiation;
|
|
|
With
2,3-dicyano-5,6-dichloro-p-benzoquinone;
In
dichloromethane;
for 2h;
Inert atmosphere;
Darkness;
|
117 mg |
|
With
1-hydroxy-3H-benz[d][1,2]iodoxole-1,3-dione;
In
dichloromethane;
at 39 ℃;
|
|
|
With
ammonium cerium (IV) nitrate;
In
dichloromethane;
at 20 ℃;
for 0.333333h;
Green chemistry;
|

pyrrole

4-methyl-benzaldehyde
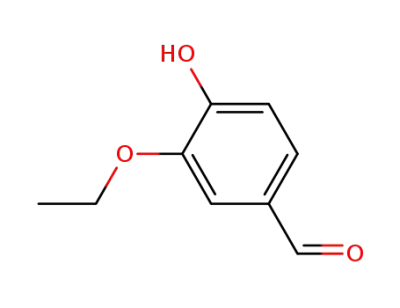
4-hydroxy-3-ethoxybenzaldehyde
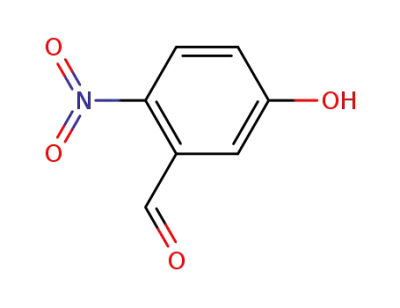
2-nitro-5-hydroxybenzaldehyde
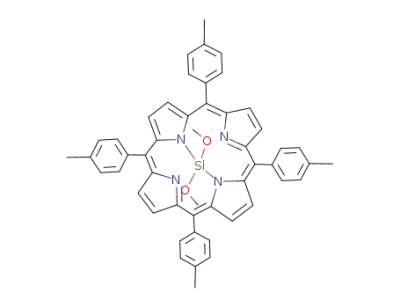
Dimethoxo<5,10,15,20-tetrakis(4-methylphenyl)porphyrinato>silicium(IV)
CAS:112163-33-4
CAS:112-84-5
CAS:1380723-44-3
CAS:1245916-14-6