- +86-0533-2185556
- WhatsApp: +86 15965530500
- admin@hangyubiotech.com
Your Location:Home >Products >API >956104-40-8
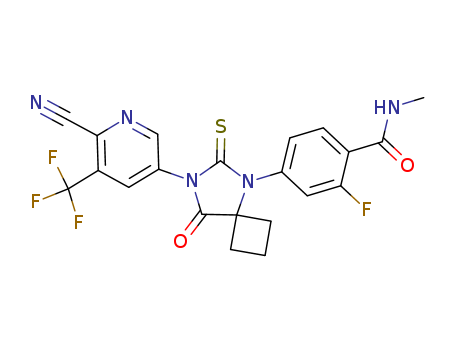

Purity:99%
The present application relates a proces...
Aspect of the present application relate...
The invention relates to an intermediate...
The invention relates to a method for pr...
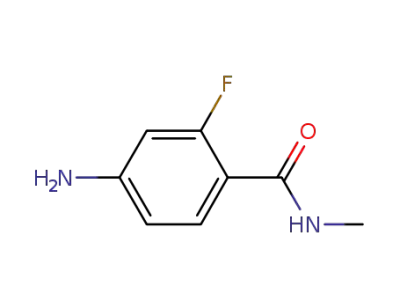
2-fluoro-N-methyl-4-amino-benzamide

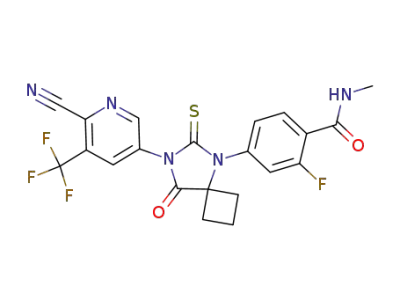
ARN-509
| Conditions | Yield |
|---|---|
|
Multi-step reaction with 2 steps
1.1: acetic acid / 16 h / 80 °C
2.1: N,N-dimethyl acetamide / 16 h / 60 °C
2.2: 1 h / Reflux
With
acetic acid;
In
N,N-dimethyl acetamide;
|
|
|
Multi-step reaction with 2 steps
1.1: acetic acid / 24 h / 80 °C
2.1: N,N-dimethyl-formamide / 20 h / 80 °C / Microwave irradiation
2.2: 2 h
With
acetic acid;
In
N,N-dimethyl-formamide;
|
|
|
Multi-step reaction with 2 steps
1.1: acetic acid / 16 h / 80 °C / Sealed tube
2.1: N,N-dimethyl acetamide / 16 h / 60 °C / Inert atmosphere
2.2: 2 h / Reflux; Inert atmosphere
With
acetic acid;
In
N,N-dimethyl acetamide;
|
|
|
Multi-step reaction with 2 steps
1.1: triethylamine / dichloromethane / 20 h / 20 °C
1.2: 4 h / Reflux
1.3: 16 h / 20 °C
2.1: N,N-dimethyl-formamide / 16 h / 80 °C / Sealed tube
With
triethylamine;
In
dichloromethane; N,N-dimethyl-formamide;
|
|
|
Multi-step reaction with 3 steps
1.1: triethylamine / dichloromethane / 20 h / 20 °C
1.2: 4 h / Reflux
1.3: 16 h / 20 °C
2.1: thionyl chloride / 16 h / 0 - 40 °C
3.1: pyridine / 4.75 h / 60 °C
With
pyridine; thionyl chloride; triethylamine;
In
dichloromethane;
|
|
|
Multi-step reaction with 3 steps
1.1: triethylamine / dichloromethane / 20 h / 20 °C
1.2: 4 h / Reflux
1.3: 16 h / 20 °C
2.1: dicyclohexyl-carbodiimide / dichloromethane; ethyl acetate / 2 h / -15 - 0 °C
3.1: pyridine / 112 h / 20 - 60 °C / Sealed tube
With
pyridine; triethylamine; dicyclohexyl-carbodiimide;
In
dichloromethane; ethyl acetate;
|
|
|
Multi-step reaction with 2 steps
1.1: acetic acid / tetrahydrofuran / 26.17 h / 25 - 85 °C
2.1: Isopropyl acetate; N,N-dimethyl-formamide / 65 - 70 °C
2.2: 3 h / 25 - 85 °C
With
acetic acid;
In
tetrahydrofuran; Isopropyl acetate; N,N-dimethyl-formamide;
|
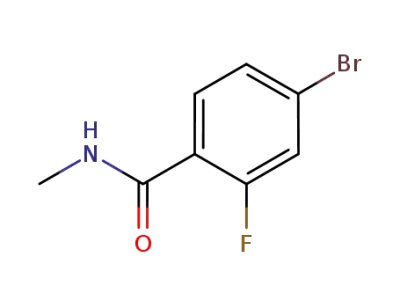
4-bromo-2-fluoro-N-methylbenzanamide


ARN-509
| Conditions | Yield |
|---|---|
|
Multi-step reaction with 3 steps
1.1: potassium carbonate / N,N-dimethyl-formamide; water / 18 h / 100 °C
2.1: potassium carbonate / N,N-dimethyl-formamide / 0.17 h / 50 °C
2.2: 10 h / 0 - 20 °C
3.1: dimethyl sulfoxide / 1 h / 85 °C / Inert atmosphere
With
potassium carbonate;
In
water; dimethyl sulfoxide; N,N-dimethyl-formamide;
|
|
|
Multi-step reaction with 3 steps
1.1: potassium carbonate / N,N-dimethyl-formamide; water / 18 h / 100 °C
2.1: potassium carbonate / N,N-dimethyl-formamide / 0.17 h / 50 °C
2.2: 5 h / 50 °C
3.1: dimethyl sulfoxide / 85 °C / Inert atmosphere
With
potassium carbonate;
In
water; dimethyl sulfoxide; N,N-dimethyl-formamide;
|
|
|
Multi-step reaction with 3 steps
1.1: potassium carbonate / N,N-dimethyl-formamide; water / 18 h / 100 °C
2.1: dmap / dichloromethane / 0.17 h
2.2: 24 h / 0 - 20 °C
3.1: dimethyl sulfoxide / 8 h / 85 °C / Inert atmosphere
With
dmap; potassium carbonate;
In
dichloromethane; water; dimethyl sulfoxide; N,N-dimethyl-formamide;
|
|
|
Multi-step reaction with 3 steps
1.1: potassium carbonate / N,N-dimethyl-formamide; water / 18 h / 100 °C
2.1: dmap / dichloromethane / 0.17 h
2.2: 24 h / 0 - 20 °C
3.1: dimethyl sulfoxide / 85 °C / Inert atmosphere
With
dmap; potassium carbonate;
In
dichloromethane; water; dimethyl sulfoxide; N,N-dimethyl-formamide;
|
|
|
Multi-step reaction with 3 steps
1: potassium carbonate / N,N-dimethyl-formamide; water / 18 h / 100 °C
2: potassium carbonate / N,N-dimethyl-formamide; water / 1 h / 30 - 40 °C
3: dimethyl sulfoxide / 8 h / 80 °C
With
potassium carbonate;
In
water; dimethyl sulfoxide; N,N-dimethyl-formamide;
|
|
|
Multi-step reaction with 3 steps
1: potassium carbonate / N,N-dimethyl-formamide; water / 18 h / 100 °C
2: potassium carbonate / N,N-dimethyl-formamide; water / 5 h / 30 - 50 °C
3: dimethyl sulfoxide / 5 h / 85 °C
With
potassium carbonate;
In
water; dimethyl sulfoxide; N,N-dimethyl-formamide;
|
|
|
Multi-step reaction with 3 steps
1: 2-acetylcyclohexanone; potassium carbonate; copper(l) chloride / N,N-dimethyl acetamide; water / 8 h / 95 - 105 °C / Inert atmosphere
2: potassium carbonate / N,N-dimethyl acetamide / 1 h / 20 - 45 °C / Inert atmosphere
3: acetonitrile / 40 h / 85 °C
With
2-acetylcyclohexanone; potassium carbonate; copper(l) chloride;
In
N,N-dimethyl acetamide; water; acetonitrile;
|
|
|
Multi-step reaction with 4 steps
1: caesium carbonate; copper(l) iodide / N,N-dimethyl acetamide / 90 - 95 °C / Inert atmosphere
2: thionyl chloride / 40 - 45 °C
3: N-ethyl-N,N-diisopropylamine / methanol / Reflux
4: potassium carbonate; copper(l) iodide / N,N-dimethyl-formamide / 100 - 110 °C / Inert atmosphere
With
copper(l) iodide; thionyl chloride; potassium carbonate; caesium carbonate; N-ethyl-N,N-diisopropylamine;
In
methanol; N,N-dimethyl acetamide; N,N-dimethyl-formamide;
|
|
|
Multi-step reaction with 2 steps
1: copper(l) iodide; potassium carbonate; triethylamine; 2-acetylcyclohexanone / water; N,N-dimethyl-formamide / 60 h / 95 - 100 °C
2: N,N-dimethyl-formamide / 16 h / 80 °C / Sealed tube
With
copper(l) iodide; 2-acetylcyclohexanone; potassium carbonate; triethylamine;
In
water; N,N-dimethyl-formamide;
|
|
|
Multi-step reaction with 3 steps
1: copper(l) iodide; potassium carbonate; triethylamine; 2-acetylcyclohexanone / water; N,N-dimethyl-formamide / 60 h / 95 - 100 °C
2: thionyl chloride / 16 h / 0 - 40 °C
3: pyridine / 4.75 h / 60 °C
With
pyridine; copper(l) iodide; thionyl chloride; 2-acetylcyclohexanone; potassium carbonate; triethylamine;
In
water; N,N-dimethyl-formamide;
|
|
|
Multi-step reaction with 3 steps
1: copper(l) iodide; potassium carbonate; triethylamine; 2-acetylcyclohexanone / water; N,N-dimethyl-formamide / 60 h / 95 - 100 °C
2: dicyclohexyl-carbodiimide / dichloromethane; ethyl acetate / 2 h / -15 - 0 °C
3: pyridine / 112 h / 20 - 60 °C / Sealed tube
With
pyridine; copper(l) iodide; 2-acetylcyclohexanone; potassium carbonate; triethylamine; dicyclohexyl-carbodiimide;
In
dichloromethane; water; ethyl acetate; N,N-dimethyl-formamide;
|
|
|
Multi-step reaction with 3 steps
1.1: potassium carbonate; copper(l) chloride / N,N-dimethyl-formamide / 10.5 h / 20 - 110 °C / Inert atmosphere
2.1: potassium carbonate / N,N-dimethyl-formamide / 0.5 h / 20 °C
2.2: 2 h / 40 °C
3.1: N,N-dimethyl-formamide / 8 h / 85 °C / Inert atmosphere
With
potassium carbonate; copper(l) chloride;
In
N,N-dimethyl-formamide;
|
|
|
Multi-step reaction with 3 steps
1: potassium carbonate; copper(l) chloride / N,N-dimethyl-formamide / 10.5 h / 20 - 110 °C / Inert atmosphere
2: sulfuric acid / 20 h / Reflux
3: N,N-dimethyl-formamide / 8 h / 85 °C / Inert atmosphere
With
sulfuric acid; potassium carbonate; copper(l) chloride;
In
N,N-dimethyl-formamide;
|
|
|
Multi-step reaction with 3 steps
1: potassium carbonate; copper(l) chloride / N,N-dimethyl-formamide / 10.5 h / 20 - 110 °C / Inert atmosphere
2: acetyl chloride / 10 h / Reflux
3: dimethylsulfoxide-d6 / 12 h / 85 °C / Inert atmosphere
With
potassium carbonate; acetyl chloride; copper(l) chloride;
In
dimethylsulfoxide-d6; N,N-dimethyl-formamide;
|
|
|
Multi-step reaction with 3 steps
1: potassium carbonate; copper(l) chloride / N,N-dimethyl-formamide / 10.5 h / 20 - 110 °C / Inert atmosphere
2: acetyl chloride / 10 h / Reflux
3: dimethyl sulfoxide; Isopropyl acetate / 12 h / 85 °C / Inert atmosphere
With
potassium carbonate; acetyl chloride; copper(l) chloride;
In
Isopropyl acetate; dimethyl sulfoxide; N,N-dimethyl-formamide;
|
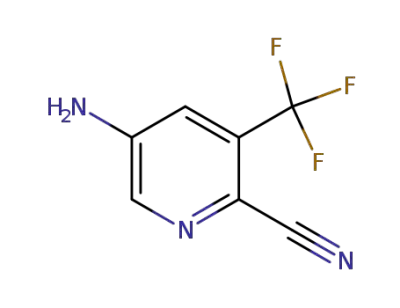
5-amino-3-(trifluoromethyl)pyridine-2-carbonitrile
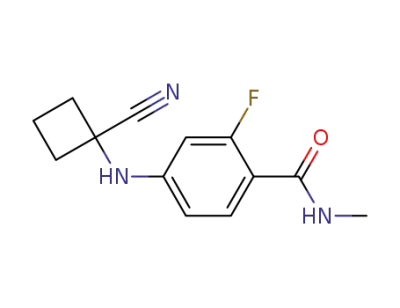
4-(1-cyano-cyclobutanylamino)-2-fluoro-N-methyl-benzamide
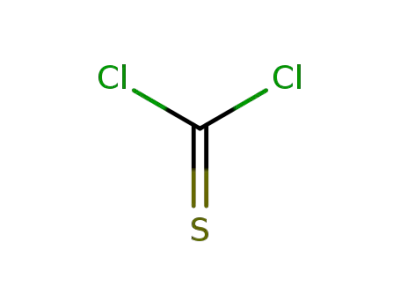
thiophosgene
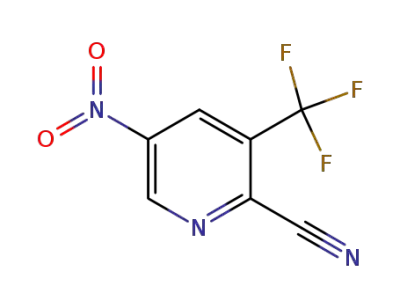
2-cyano-3-(trifluoromethyl)-5-nitropyridine
CAS:117724-63-7
CAS:2169919-21-3
CAS:2936625-34-0
CAS:97229-11-3