- +86-0533-2185556
- WhatsApp: +86 15965530500
- admin@hangyubiotech.com
Your Location:Home >Products >API >57450-61-0
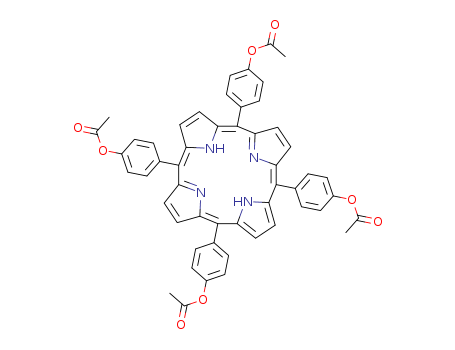

Purity:99%
Towards mimicking natural photosystems, ...
A supramolecular photosensitizer was con...
Biochemical activities at a membrane int...
Sixteen porphyrins, including neutral, a...
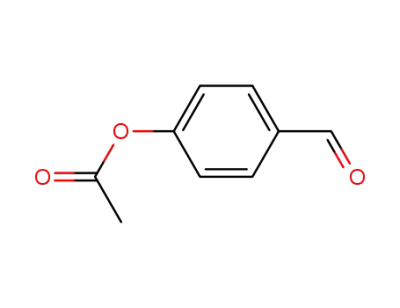
4-formylphenyl acetate

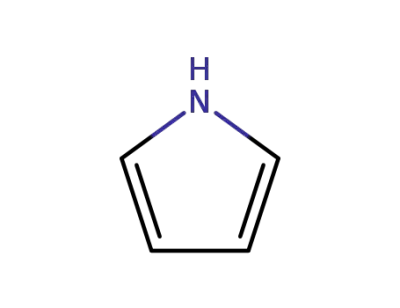
pyrrole

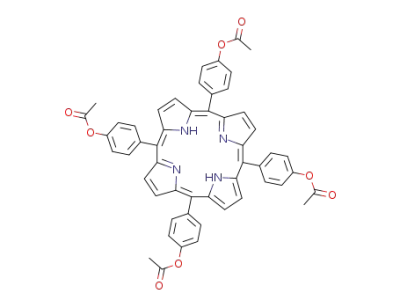
5,10,15,20-tetrakis(p-acetoxyphenyl)porphyrin
| Conditions | Yield |
|---|---|
|
4-formylphenyl acetate; pyrrole;
With
boron trifluoride diethyl etherate;
In
dichloromethane;
at 20 ℃;
for 0.5h;
With
selenium(IV) oxide;
In
dichloromethane;
at 20 ℃;
for 1h;
|
28% |
|
4-formylphenyl acetate; pyrrole;
With
boron trifluoride diethyl etherate;
In
dichloromethane;
at 20 ℃;
for 0.5h;
With
selenium(IV) oxide;
In
dichloromethane;
for 1h;
|
26% |
|
4-formylphenyl acetate;
With
benzoic acid;
at 200 ℃;
for 0.0833333h;
under 2736.18 Torr;
pyrrole;
at 200 ℃;
for 0.333333h;
|
19% |
|
With
propionic acid;
for 0.5h;
Reflux;
|
19% |
|
With
propionic acid;
for 0.5h;
Reflux;
|
19% |
|
With
propionic acid;
at 135 ℃;
for 4h;
Inert atmosphere;
|
18.1% |
|
With
propionic acid;
|
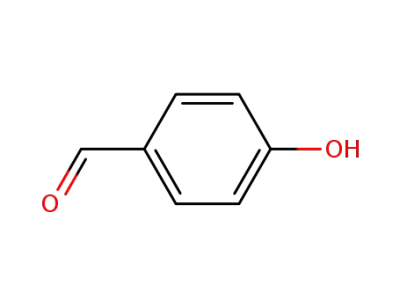
4-hydroxy-benzaldehyde


5,10,15,20-tetrakis(p-acetoxyphenyl)porphyrin
| Conditions | Yield |
|---|---|
|
Multi-step reaction with 2 steps
1: triethylamine; dmap / dichloromethane
2: propionic acid
With
dmap; propionic acid; triethylamine;
In
dichloromethane;
|

4-formylphenyl acetate

pyrrole

4-hydroxy-benzaldehyde
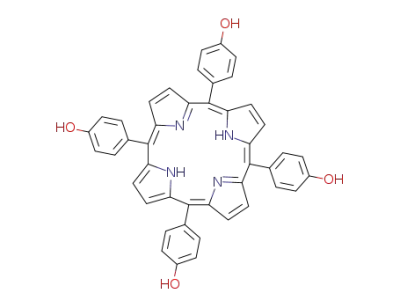
5,10,15,20-tetrakis(4'-hydroxyphenyl)porphyrin
CAS:196929-78-9
CAS:2210228-45-6
CAS:8047-67-4
CAS:1308833-36-4