- +86-0533-2185556
- WhatsApp: +86 15965530500
- admin@hangyubiotech.com
Your Location:Home >Products >API >16834-13-2
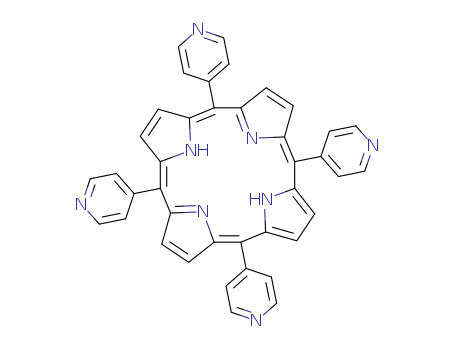

Purity:99%
|
Purification Methods |
Purify it by chromatography on alumina (neutral, Grade I), with CHCl3/MeOH (80:20) followed by recrystallisation from CH2Cl2/MeOH [Yamashita et al. J Phys Chem 91 3055 1987]. [Kalyanasundaram Inorg Chem 23 2453 1984, Okuno et al. Synthesis 537 1980.] |
|
General Description |
5,10,15,20-tetra-4-pyridyl-21H,23H-porphine (H2TPyP)?can react with [Re6(μ3-Se)8(Pet3)5(NCMe]2+ and 2,4,6-tri-4-pyridyl-1,3,5-triazine to form tri- and tetra complexes. 1 Trace amounts of nitroaromatic explosives can be detected by porphyrin functionalized graphene. It behaves as a sensitive amperometric sensor. 2 |
InChI:InChI=1/C40H26N8/c1-2-30-38(26-11-19-42-20-12-26)32-5-6-34(47-32)40(28-15-23-44-24-16-28)36-8-7-35(48-36)39(27-13-21-43-22-14-27)33-4-3-31(46-33)37(29(1)45-30)25-9-17-41-18-10-25/h1-24,45,48H/b37-29-,37-31-,38-30-,38-32-,39-33-,39-35-,40-34-,40-36-
Quaternized tetra(4-pyridyl)porphine der...
A series of chiral synthetic compounds i...
Porphyrins are well-known anticancer age...
The preparation and properties of lumine...
The interaction modes and nuclease activ...
A novel tetracationic porphyrin-platinum...
For constructing a bifunctional antioxid...
To achieve the synthesis of (Fe3O4@SnO2/...
This article describes a new synthetic m...
Aiming at preparation of new biologicall...
Photodynamic inactivation (PDI) is an ef...
Tetracationic (TMPyP) and tricationic po...
A meso-pyridyl porphyrin, 5-(4-(2-(4-(S-...
Studies on the synthesis, structural elu...
An entirely earth-abundant chromophore-r...
The combined use of porphyrins and fluor...
The present work sheds light on the role...
The synthesis and characterization of ca...
The self-assembly of the oppositely char...
A supramolecular nanocapsule was constru...
The synthesis of one-step meso-porphyrin...
This work presents the synthesis and cha...
Fe(iii)-meso-tetra(pyridyl)porphyrins ar...
The selective oxidation of sulfides with...
A series of magnetic, ionic-substituted ...
-
The water-soluble cationic nickel(II) co...
The extracoordination reactions of nitro...
A series of water-soluble fluorinated ca...
The push-pull electronic and steric effe...
The development of new photodynamic ther...
Despite advances achieved over the last ...
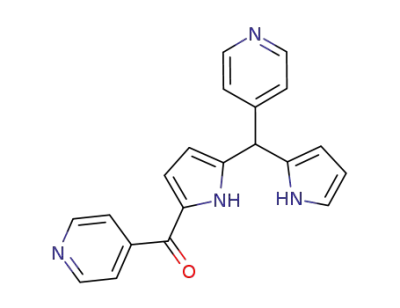
1-isonicotinoyl-5-(4-pyridyl)dipyrromethane

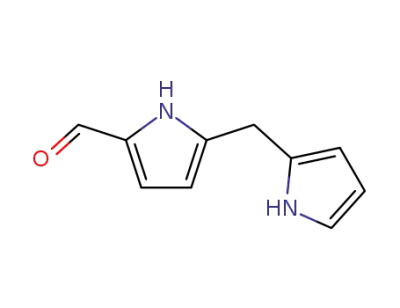
5-formyl-2,2'-dipyrrylmethane

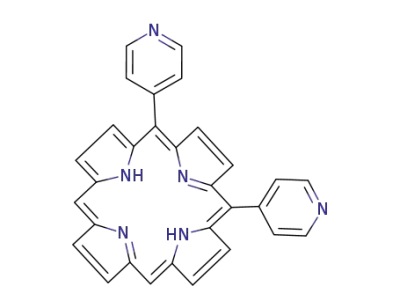
5,10-di(4-pyridyl)porphyrin

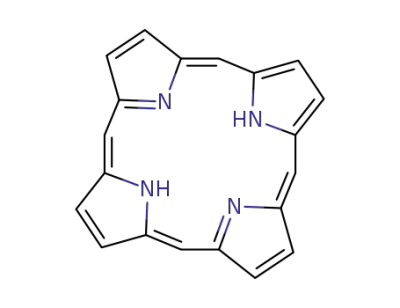
porphyrin

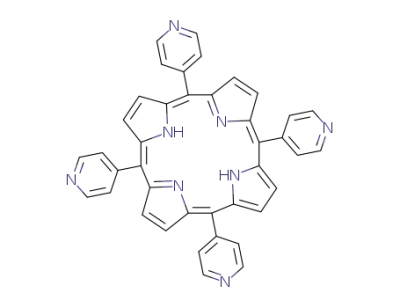
5,10,15,20-tetra(4-pyridyl)-21H,23H-porphine
| Conditions | Yield |
|---|---|
|
With
air; 1,8-diazabicyclo[5.4.0]undec-7-ene; magnesium bromide;
In
toluene;
at 115 ℃;
for 1.5h;
microwave irradiation;
|
27% 16% 14% |
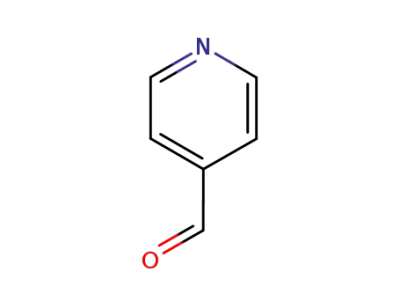
pyridine-4-carbaldehyde

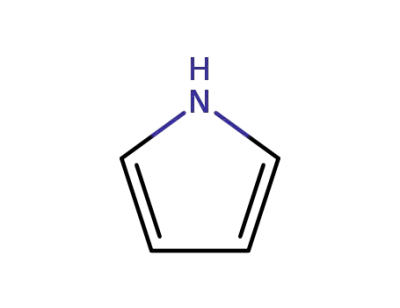
pyrrole


5,10,15,20-tetra(4-pyridyl)-21H,23H-porphine
| Conditions | Yield |
|---|---|
|
With
propionic acid;
for 3h;
Reflux;
Darkness;
|
49% |
|
With
propionic acid;
for 3h;
Reflux;
Darkness;
|
49% |
|
With
salicylic acid;
In
xylene;
for 3.16667h;
Heating;
|
32% |
|
In
propionic acid;
for 1.5h;
Reflux;
|
27% |
|
In
propionic acid;
for 1h;
Reflux;
|
24% |
|
In
propionic acid;
for 0.75h;
Heating;
|
23% |
|
In
propionic acid;
for 3h;
Reflux;
Inert atmosphere;
|
23% |
|
In
propionic acid;
at 50 ℃;
for 1.25h;
Inert atmosphere;
|
21% |
|
With
acetic anhydride; propionic acid;
for 0.5h;
Reflux;
|
21.3% |
|
With
propionic acid;
for 3h;
Reflux;
|
20% |
|
With
propionic acid;
for 2h;
Reflux;
|
20% |
|
With
propionic acid;
for 2h;
Reflux;
|
20% |
|
With
propionic acid;
for 2.5h;
Heating;
|
17% |
|
With
propionic acid;
at 140 ℃;
for 1.5h;
Darkness;
|
15% |
|
In
propionic acid;
at 150 ℃;
for 1h;
Darkness;
|
13% |
|
pyridine-4-carbaldehyde; pyrrole;
With
propionic acid;
for 2h;
Heating / reflux;
With
ammonia; water;
|
12% |
|
With
1-butylimidazolium trifluoroacetate;
at 120 ℃;
for 1h;
Reagent/catalyst;
Green chemistry;
|
11% |
|
In
acetic acid; nitrobenzene;
at 120 ℃;
for 1h;
|
11% |
|
With
zinc diacetate;
at 200 ℃;
for 0.25h;
|
10% |
|
With
propionic acid;
for 45h;
Heating;
|
10.3% |
|
pyridine-4-carbaldehyde; pyrrole;
With
propionic acid;
for 3h;
Reflux;
Inert atmosphere;
In
methanol;
at 80 ℃;
for 1h;
|
10% |
|
With
propionic acid;
for 3h;
Reflux;
|
10% |
|
With
propionic acid;
for 0.75h;
Reflux;
|
7.6% |
|
With
propionic acid;
In
nitrobenzene;
at 20 ℃;
for 2h;
Reflux;
|
2.4% |
|
With
propionic acid;
Heating;
|
|
|
With
propionic acid;
Heating;
|
|
|
With
propionic acid;
for 0.5h;
Heating;
|
|
|
With
acetic anhydride; propionic acid;
at 130 ℃;
|
|
|
With
salicylic acid;
In
5,5-dimethyl-1,3-cyclohexadiene;
for 1.5h;
Reflux;
|
|
|
With
propionic acid;
|
|
|
pyridine-4-carbaldehyde;
With
zinc diacetate;
at 200 ℃;
for 0.0166667h;
under 2736.18 Torr;
pyrrole;
at 200 ℃;
for 0.333333h;
|
|
|
With
propionic acid;
for 1h;
Reflux;
|
|
|
With
acetic acid;
In
nitrobenzene;
at 130 ℃;
for 3h;
|
0.2% |
|
In
propionic acid;
at 90 - 141 ℃;
for 3h;
|
|
|
pyridine-4-carbaldehyde;
With
acetic anhydride; propionic acid;
at 130 ℃;
pyrrole;
for 1.5h;
Reflux;
|
|
|
With
acetic acid; propionic acid;
for 1h;
Inert atmosphere;
Reflux;
|
|
|
With
acetic acid;
In
propionic acid;
for 1h;
Reflux;
|
|
|
With
propionic acid;
for 1.5h;
Reflux;
|
|
|
With
propionic acid;
|
|
|
pyridine-4-carbaldehyde; pyrrole;
With
propionic acid;
for 1.5h;
Reflux;
With
2,3-dicyano-5,6-dichloro-p-benzoquinone;
In
chloroform;
for 0.5h;
Reflux;
|

pyridine-4-carbaldehyde

pyrrole
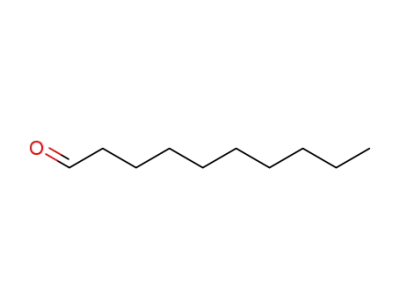
caprinaldehyde
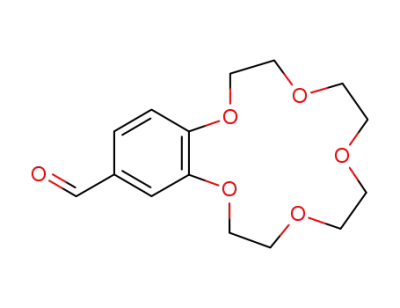
benzo-15-crown-5-ether

5,10,15,20-tetra(N-methylpyridinium-4-yl)porphine
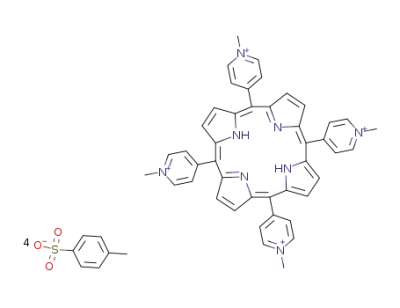
tetra(4-N-methylpyridyl)porphyrin tosylate

5,10,15,20-tetrakis(4-methylpyridinium)porphyrin tetrahydrochloride
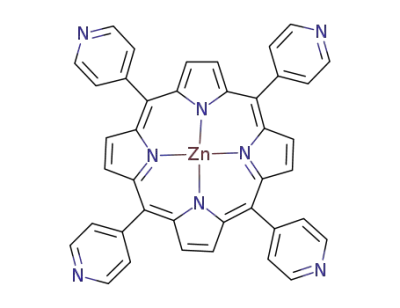
5,10,15,20-tetra(4-pyridyl)porphyrin zinc
CAS:112163-33-4
CAS:112-84-5
CAS:660431-66-3
CAS:120-29-6