- +86-0533-2185556
- WhatsApp: +86 15965530500
- admin@hangyubiotech.com
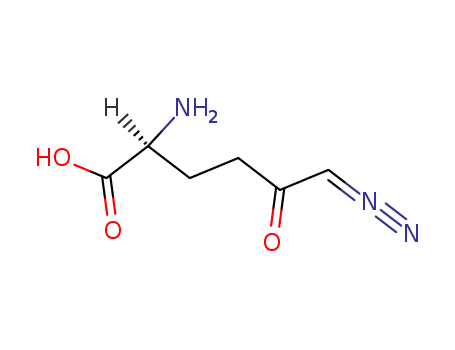

pd_meltingpoint:-145oC (dec.)
Purity:99%
|
Biochem/physiol Actions |
DON is used to study mechanisms of glutamine utilizing enzymes such as carbamoyl phosphate synthase and cytidine triphosphate synthase. |
|
Purification Methods |
Crystallise it from EtOH, H2O/EtOH, MeOH, 95% aqueous MeOH or H2O/Me2CO. [DeWald & Moor J Am Chem Soc 80 3944 1958, Dion et al. J Am Chem Soc 78 3075 1956, Beilstein 4 IV 3278.] |
|
General Description |
Chemical structure: amino acid derivatives |
InChI:InChI=1/C6H9N3O3/c7-5(6(11)12)2-1-4(10)3-9-8/h3,5H,1-2,7H2,(H-,10,11,12)/b4-3+/t5-/m0/s1
A series of electrophilic glutamine anal...
The transfer of the metallocarbene deriv...
The reaction of carbamate derivatives of...
-
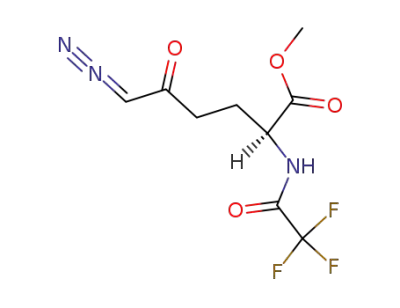
6-Diazo-5-oxo-N-Tfa-L-norleucine-OMe

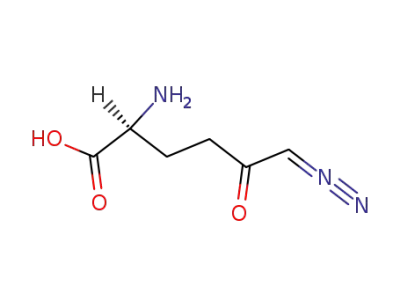
6-diazo-5-oxo-L-norleucine
| Conditions | Yield |
|---|---|
|
With
sodium hydroxide;
In
methanol;
for 0.5h;
Ambient temperature;
|
88% |
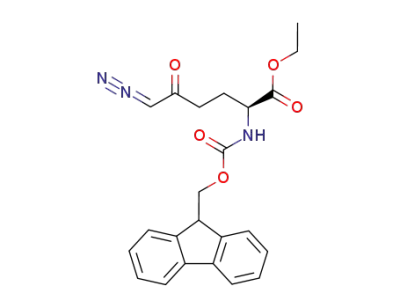
ethyl 2-((((9H-fluoren-9-yl)methoxy)carbonyl)amino)-6-diazo-5-oxohexanoate


6-diazo-5-oxo-L-norleucine
| Conditions | Yield |
|---|---|
|
With
piperidine;
for 0.0333333h;
|
81% |
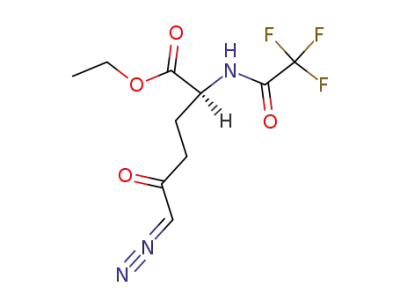
(S)-6-diazo-5-oxo-2-(2,2,2-trifluoro-acetylamino)-hexanoic acid ethyl ester
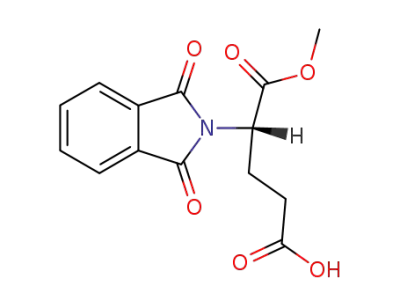
(2S)-2-(1,3-dioxo-1,3-dihydroisoindol-2-yl)pentanedioic acid α-methyl ester

6-Diazo-5-oxo-N-Tfa-L-norleucine-OMe

ethyl 2-((((9H-fluoren-9-yl)methoxy)carbonyl)amino)-6-diazo-5-oxohexanoate
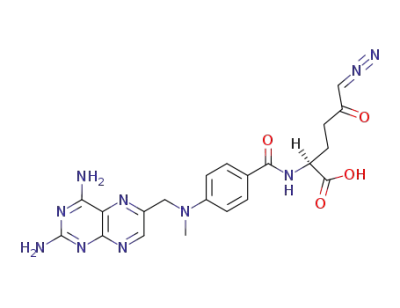
4-amino-4-deoxy-N10-methylpteroyl-(6-diazo-5-oxo)-L-norleucine
CAS:112163-33-4
CAS:112-84-5
CAS:444930-42-1
CAS:943764-99-6