- +86-0533-2185556
- WhatsApp: +86 15965530500
- admin@hangyubiotech.com
Your Location:Home >Products >API >1146629-75-5
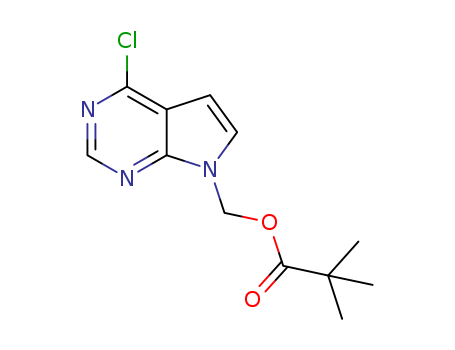

Purity:99%
Nine compounds potentially related to th...
Aiming to develop potent JAK inhibitors,...
The invention relates to a piperidine-co...
Provided in the present invention are a ...
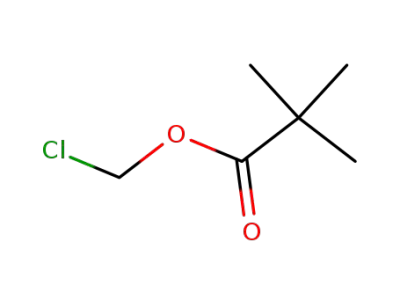
Chloromethyl pivalate

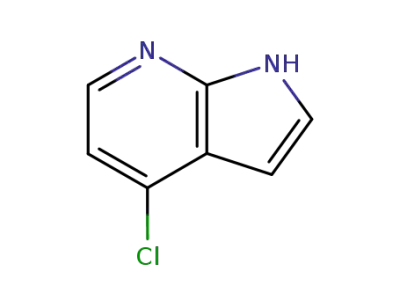
4-chloro-7-azaindole

![(4-chloro-7H-pyrrolo[2,3-d]pyrimidin-7-yl)methyl 2,2-dimethylpropanoate](/upload/2026/5/adfdd678-87b1-46e9-abd5-39f7647c8d49.png)
(4-chloro-7H-pyrrolo[2,3-d]pyrimidin-7-yl)methyl 2,2-dimethylpropanoate
| Conditions | Yield |
|---|---|
|
With
potassium carbonate;
In
N,N-dimethyl-formamide;
at 20 ℃;
for 16h;
|
8.47 g |

Chloromethyl pivalate

![4-chloro-1H-pyrrolo[2,3-d]pyrimidine](/upload/2026/5/bb1833bf-8b6a-43fa-9778-fc0a24e651c3.png)
4-chloro-1H-pyrrolo[2,3-d]pyrimidine

![(4-chloro-7H-pyrrolo[2,3-d]pyrimidin-7-yl)methyl 2,2-dimethylpropanoate](/upload/2026/5/adfdd678-87b1-46e9-abd5-39f7647c8d49.png)
(4-chloro-7H-pyrrolo[2,3-d]pyrimidin-7-yl)methyl 2,2-dimethylpropanoate
| Conditions | Yield |
|---|---|
|
With
potassium carbonate;
In
N,N-dimethyl-formamide;
at 20 ℃;
for 14h;
|
98.85% |
|
4-chloro-1H-pyrrolo[2,3-d]pyrimidine;
With
sodium hydride;
In
tetrahydrofuran;
at 0 - 20 ℃;
Inert atmosphere;
Chloromethyl pivalate;
In
tetrahydrofuran;
at 0 - 20 ℃;
Inert atmosphere;
|
91% |
|
4-chloro-1H-pyrrolo[2,3-d]pyrimidine;
With
sodium hydride;
In
tetrahydrofuran; mineral oil;
at 0 - 20 ℃;
Inert atmosphere;
Chloromethyl pivalate;
In
tetrahydrofuran; mineral oil;
at 0 - 20 ℃;
Product distribution / selectivity;
Inert atmosphere;
|
91% |
|
4-chloro-1H-pyrrolo[2,3-d]pyrimidine;
With
sodium t-butanolate;
In
tetrahydrofuran;
at -10 - 20 ℃;
Inert atmosphere;
Chloromethyl pivalate;
In
tetrahydrofuran;
at 20 ℃;
Reagent/catalyst;
Temperature;
Solvent;
|
91.6% |
|
4-chloro-1H-pyrrolo[2,3-d]pyrimidine;
With
sodium hydride;
In
tetrahydrofuran;
at 0 ℃;
for 0.5h;
Chloromethyl pivalate;
In
tetrahydrofuran;
at 20 ℃;
for 1.5h;
|
85% |
|
4-chloro-1H-pyrrolo[2,3-d]pyrimidine;
With
sodium hydride;
In
tetrahydrofuran;
for 0.5h;
Cooling with ice;
Chloromethyl pivalate;
In
tetrahydrofuran;
at 20 ℃;
for 1.5h;
Inert atmosphere;
|
85% |
|
4-chloro-1H-pyrrolo[2,3-d]pyrimidine;
With
sodium hydride;
In
1,2-dimethoxyethane; mineral oil;
at 0 - 20 ℃;
Chloromethyl pivalate;
In
1,2-dimethoxyethane; mineral oil;
at 0 - 20 ℃;
|
|
|
4-chloro-1H-pyrrolo[2,3-d]pyrimidine;
With
sodium hydride;
In
tetrahydrofuran; mineral oil;
at 0 ℃;
for 0.166667h;
Chloromethyl pivalate;
In
tetrahydrofuran; mineral oil;
at 0 - 20 ℃;
for 1.5h;
|
|
|
4-chloro-1H-pyrrolo[2,3-d]pyrimidine;
With
sodium hydride;
In
tetrahydrofuran; mineral oil;
at 0 ℃;
for 0.166667h;
Chloromethyl pivalate;
In
tetrahydrofuran; mineral oil;
at 0 - 20 ℃;
for 1.5h;
|
|
|
4-chloro-1H-pyrrolo[2,3-d]pyrimidine;
With
sodium hydride;
In
tetrahydrofuran; mineral oil;
at 0 ℃;
for 0.166667h;
Chloromethyl pivalate;
In
tetrahydrofuran; mineral oil;
at 0 - 20 ℃;
for 1.5h;
|
170 mg |
|
With
sodium hydroxide;
In
N,N-dimethyl-formamide;
Inert atmosphere;
|
|
|
4-chloro-1H-pyrrolo[2,3-d]pyrimidine;
With
sodium hydride;
In
N,N-dimethyl acetamide;
at 10 - 15 ℃;
for 1h;
Inert atmosphere;
Chloromethyl pivalate;
In
tetrahydrofuran; N,N-dimethyl acetamide;
at 20 ℃;
for 3h;
Inert atmosphere;
|
530 g |

Chloromethyl pivalate
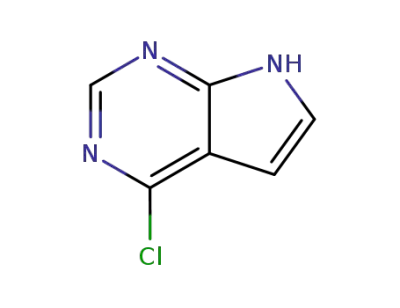
4-chloro-1H-pyrrolo[2,3-d]pyrimidine

4-chloro-7-azaindole
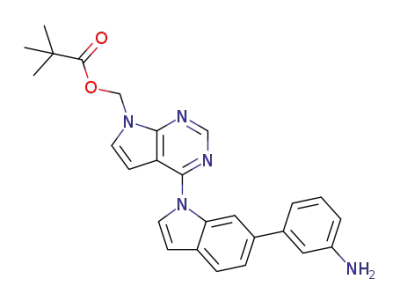
(4-(6-(3-aminophenyl)-1H-indole-1-yl)-7H-pyrrolo[2,3-d]pyrimidine-7-yl)methyl pivalate
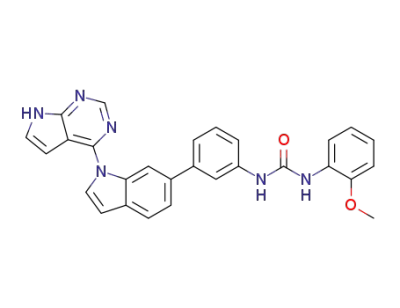
1-(3-(1-(7H-pyrrolo[2,3-d]pyrimidine-4-yl)-1H-indole-6-yl)phenyl)-3-(2-methoxyphenyl)urea
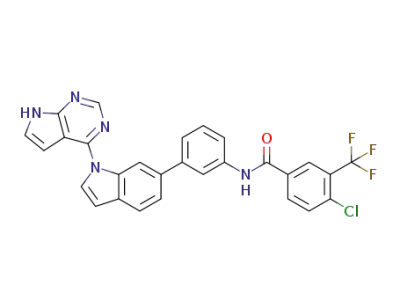
N-(3-(1-(7H-pyrrolo[2,3-d]pyrimidin-4-yl)-1H-indol-6-yl)phenyl)-4-chloro-3-(trifluoromethyl)benzamide
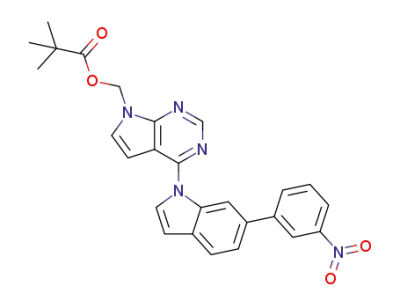
(4-(6-(3-nitrophenyl)-1H-indole-1-yl)-7H-pyrrolo[2,3-d]pyrimidine-7-yl)methyl pivalate
CAS:115473-15-9
CAS:118685-33-9
CAS:841301-32-4