- +86-0533-2185556
- WhatsApp: +86 15965530500
- admin@hangyubiotech.com
Your Location:Home >Products >Pharmaceutical intermediates >4233-33-4
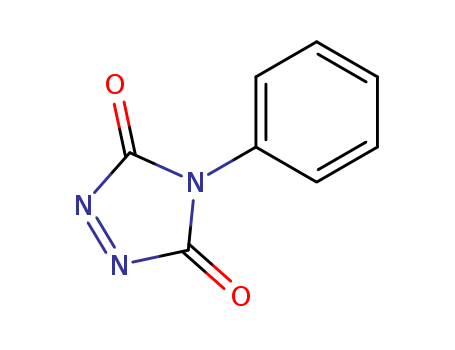

pd_meltingpoint:165-170 °C (dec.)(lit.)
Purity:99%
|
Synthesis Reference(s) |
Synthetic Communications, 17, p. 409, 1987 DOI: 10.1080/00397918708063918 |
|
Purification Methods |
PTAD forms carmine red needles by sublimation (ice cold finger) at 100o/0.1mm, and/or by recrystallisation from EtOH. IR: 1760 and 1780 cm-1 . [Cookson et al. Org Synth 51 121 1971, Moore max et al. J Org Chem 39 3700 1974, Beilstein 26 I 57, 26 III/IV 540.] |
|
General Description |
4-Phenyl-1,2,4-triazoline-3,5-dione, also known as Cookson reagent, is a strong dienophile, which gives a stable Diels-Alder adduct quantitatively within a short time and under mild conditions. It is commonly used as a protecting group of the diene moiety for the synthesis of vitamin D3 (VD3)-related compounds. |
InChI:InChI=1/C8H5N3O2/c12-7-9-10-8(13)11(7)6-4-2-1-3-5-6/h1-5H
4-Arylurazoles are selectively oxidized ...
Diels-Alder cycloaddition to several der...
Provided are a method for separating a D...
Provided is a compound having the formul...
-
Localized singlet diradicals are key int...
(1R,8S)-4,15-dimethyl-11-phenyl-9,11,13-triazatetracyclo[6.5.2.02,7.09,13]pentadeca-2,4,6,14-tetraene-10,12-dione

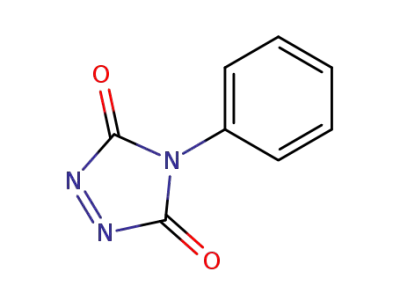
4-Phenyl-1,2,4-triazolidine-3,5-dione

2.6-dimethylnaphthalene
| Conditions | Yield |
|---|---|
|
at 20 ℃;
Equilibrium constant;
Thermodynamic data;
Kinetics;
Activation energy;
|
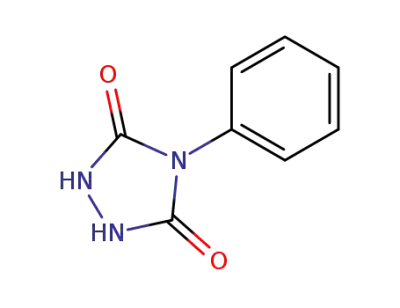
4-Phenylurazole


4-Phenyl-1,2,4-triazolidine-3,5-dione
| Conditions | Yield |
|---|---|
|
With
tetrabutylammonium periodite;
In
acetone;
at 20 ℃;
for 0.5h;
|
100% |
|
With
SiO2-supported HNO3;
In
dichloromethane;
at 20 ℃;
for 0.583333h;
chemoselective reaction;
|
99% |
|
With
chlorine;
In
ethyl acetate;
for 18h;
|
98% |
|
With
tetrachlorosilane; silica gel; sodium nitrite;
In
dichloromethane;
at 20 ℃;
for 3h;
|
98% |
|
With
ICl supported on silica gel;
In
dichloromethane;
at 20 ℃;
for 0.166667h;
|
98% |
|
With
aluminum(III) nitrate nonahydrate;
In
dichloromethane;
at 20 ℃;
for 2h;
|
98% |
|
With
guanidine nitrate;
In
dichloromethane;
at 20 ℃;
for 0.75h;
|
98% |
|
With
ammonium nitrate; aluminum(III) hydrogen sulfate;
In
dichloromethane;
at 20 ℃;
for 0.666667h;
|
98% |
|
With
aluminium trichloride; benzyltriphenylphosphonium chlorate;
In
dichloromethane;
at 20 ℃;
for 0.25h;
|
97% |
|
With
1,3-dibromo-5,5-dimethylimidazolidine-2,4-dione;
In
dichloromethane;
at 20 ℃;
for 2h;
|
97% |
|
With
sodium hydrogen sulfate; silica gel; sodium nitrite;
In
dichloromethane;
at 20 ℃;
for 1h;
|
96% |
|
With
hydrogensulfate; silica gel; sodium nitrite;
In
dichloromethane;
at 20 ℃;
for 2h;
|
96% |
|
With
1,3,4,6-tetrachloro-3α,6α-diphenyl glycoluril;
In
dichloromethane;
at 20 ℃;
for 1h;
|
96% |
|
With
aluminum (III) chloride; benzyltriphenylphosphonium nitrate;
In
dichloromethane;
for 0.5h;
|
96% |
|
With
[bis(acetoxy)iodo]benzene;
In
dichloromethane;
for 0.25h;
Ambient temperature;
|
95% |
|
With
potassium dichromate; aluminium trichloride;
for 0.05h;
|
95% |
|
With
silica gel; zirconium(IV) chloride; 1,4-diazabicyclo[2,2,2]octane-N,N′-dioxide;
In
dichloromethane;
at 20 ℃;
for 1h;
|
95% |
|
With
1,1'-(ethane-1,2-diyl)dipyridinium bistribromide; water;
In
dichloromethane;
at 20 ℃;
for 0.5h;
|
95% |
|
With
poly(ethylene glycol) impregnated with dinitrogen tetroxide;
In
dichloromethane;
at 20 ℃;
for 0.5h;
|
95% |
|
With
nitrourea;
In
dichloromethane;
at 20 ℃;
for 1h;
|
95% |
|
With
magnesium hydrogen sulfate; silica gel; sodium nitrite;
In
dichloromethane;
at 20 ℃;
for 1h;
|
94% |
|
With
benzyltriphenylphosphonium peroxymonosulfate;
aluminium trichloride;
at 20 ℃;
for 0.166667h;
grinding;
|
94% |
|
With
Oxone; silica gel; sodium nitrite;
In
dichloromethane;
at 20 ℃;
for 1.5h;
|
94% |
|
With
chromium (VI) oxide; 1-n-butyl-3-methylimidazolim bromide;
at 20 ℃;
for 1.5h;
|
94% |
|
With
calcium hypochlorite;
In
dichloromethane;
at 20 ℃;
for 0.25h;
|
92% |
|
With
KMnO4/alumina;
at 20 ℃;
for 0.133333h;
|
90% |
|
With
dinitrogen tetraoxide;
In
dichloromethane;
at 0 ℃;
|
86% |
|
With
silica gel; iodic acid; sodium nitrite;
In
dichloromethane;
at 20 ℃;
for 1.5h;
|
86% |
|
With
dihydrogen peroxide;
at 70 ℃;
for 3h;
|
84% |
|
With
N-Bromosuccinimide;
In
dichloromethane;
at 0 ℃;
for 0.333333h;
|
82% |
|
With
n-propylsilica kryptofix 21-supported dinitrogen tetroxide;
In
dichloromethane;
at 20 ℃;
|
82% |
|
With
melamine-(H2SO4)3; C3H6N6*3HNO3;
In
neat (no solvent);
Green chemistry;
|
81% |
|
With
oxalic acid; silica gel; sodium nitrite;
In
dichloromethane;
at 20 ℃;
for 1h;
|
80% |
|
With
trichloroisocyanuric acid;
In
dichloromethane;
at 20 ℃;
for 0.5h;
|
74% |
|
With
iodosylbenzene;
In
ethyl acetate;
at 25 ℃;
for 4h;
Darkness;
Inert atmosphere;
|
65% |
|
With
tert-butylhypochlorite;
In
ethyl acetate;
at 20 ℃;
for 1.66667h;
Inert atmosphere;
|
38% |
|
With
tert-butylhypochlorite;
|
|
|
With
perchloric acid; sodium perchlorate;
In
acetonitrile;
at 0 ℃;
electrolyse, Pt-electrode;
|
|
|
With
benzeneseleninic anhydride;
In
tetrahydrofuran;
for 0.0666667h;
Yield given;
Ambient temperature;
|
|
|
With
tert-butylhypochlorite;
In
dichloromethane;
at 0 ℃;
|
|
|
With
tert-butylhypochlorite;
In
acetone;
at -60 ℃;
|
|
|
With
dinitrogen tetraoxide; sodium sulfate;
In
dichloromethane;
at 0 ℃;
|
|
|
With
tert-butylhypochlorite;
In
ethyl acetate;
at 15 - 20 ℃;
for 0.666667h;
|
65.6 g |
|
With
tert-butylhypochlorite;
In
acetone;
|
|
|
With
tert-butylhypochlorite;
In
ethyl acetate;
at 0 ℃;
for 0.0833333h;
|
|
|
With
tert-butylhypochlorite;
In
acetone;
|
|
|
In
sulfuric acid; acetonitrile;
Electrolysis;
|
|
|
With
tert-butylhypochlorite;
In
ethyl acetate;
at 0 ℃;
for 0.166667h;
|
|
|
With
[NO(1+)*18-crown-6*H(NO3)2(1-)]; silica gel;
In
dichloromethane;
at 20 ℃;
|
|
|
With
N,N,2,3,4,5,6-heptachloroaniline;
In
dichloromethane;
at 20 ℃;
for 1h;
|
|
|
With
ferric nitrate;
|
|
|
With
tert-butylhypochlorite;
In
dichloromethane;
at 0 - 20 ℃;
for 2h;
|
|
|
With
dinitrogen trioxide; dinitrogen tetraoxide; Nitrogen dioxide;
In
dichloromethane;
at 0 - 5 ℃;
for 0.5h;
|
|
|
With
tert-butylhypochlorite;
|
|
|
pH=2;
pH-value;
Solvent;
Kinetics;
Electrochemical reaction;
aq. phosphate buffer;
|
|
|
With
calcium hypochlorite; sodium sulfate;
In
dichloromethane;
at 20 ℃;
for 2h;
|

4-Phenylurazole
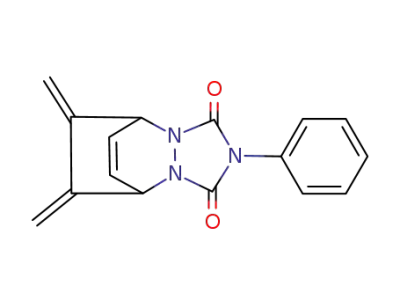
10,11-Dimethylen-4-phenyl-2,4,6-triazatricyclo<5.2.2.02,6>undec-8-en-3,5-dion
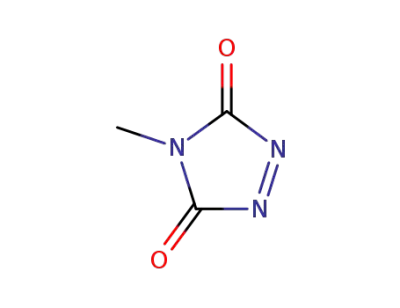
4-methyl-1,2,4-triazoline-3,5-dione
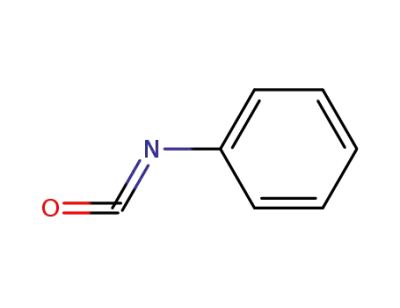
phenyl isocyanate
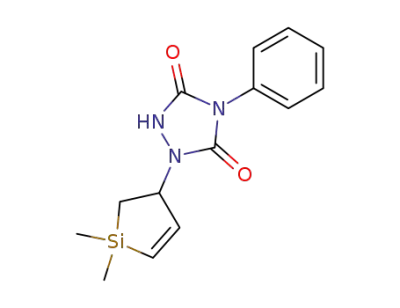
1-(1,1-dimethyl-2,3-dihydro-1H-silol-3-yl)-4-phenyl-[1,2,4]triazolidine-3,5-dione
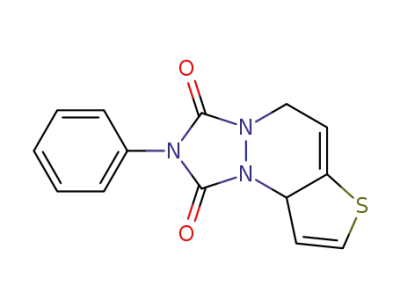
5,9a-dihydro-2-phenylthieno<3,2-c><1,2,4>triazolo<1,2-a>pyridazine-1,3-dione
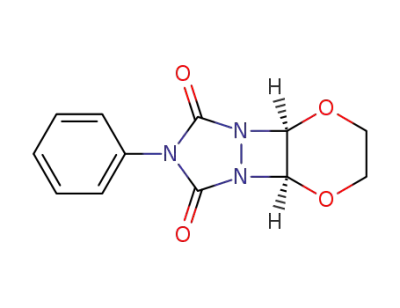
7-phenyl-(4ar,9ac)-tetrahydro-[1,4]dioxino[2',3':3,4][1,2]diazeto[1,2-a][1,2,4]triazole-6,8-dione
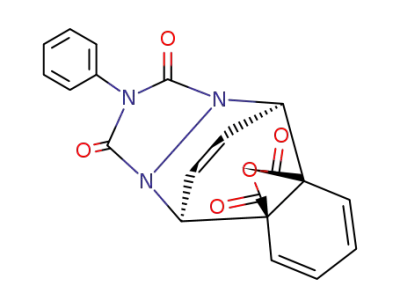
1,3-dioxo-2-phenyl-2,3-dihydro-1H-5r,10c-etheno-[1,2,4]triazolo[1,2-b]phthalazine-5at,9at-dicarboxylic acid anhydride
CAS:112163-33-4
CAS:112-84-5
CAS:16009-13-5
CAS:58069-82-2