- +86-0533-2185556
- WhatsApp: +86 15965530500
- admin@hangyubiotech.com
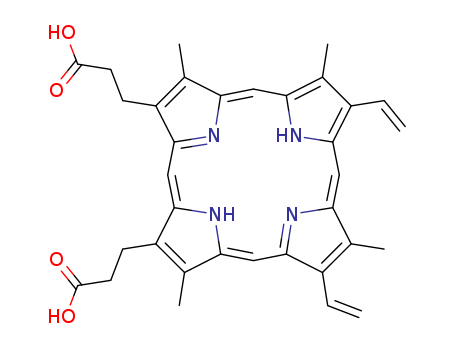

pd_meltingpoint:>300 °C(Solv: acetone (67-64-1))
Purity:99%
|
Biochem/physiol Actions |
Protoporphyrin IX?levels are elevated in?tumor?cells due to?metabolism anomalies?compared to normal?cells. |
|
Enzyme inhibitor |
This iiron-free, immediate precusor of heme (FWfree-acid = 562.67 g/mol; CAS 553-12-8; brownish-yellow solid; soluble in a number of organic solvents; disodium and dipotassium salts solublized in the presence of Tween 80) has lmax values in 25% HCl are 602.4, 582.2, and 557.2 nm. Protoporphryrin IX is also an activator of guanylate cyclase. See also Iron Protoporphyrin IX; Heme; Hemin Target(s): aminolevulinate aminotransferase; 5-aminolevulinate synthase; glutamate: glyoxylate aminotransferase; glutathione S-transferase; glyoxalase I, or lactoylglutathione lyase; guanylate cyclase; heme oxygenase; hydroxymethylbilane synthase, or porphobilinogen deaminase; nitric-oxide synthase; porphobilinogen synthase, or 5-aminolevulinate dehydratase; succinyl-CoA synthetase; tryptophan pyrrolase, or tryptophan 2,3-dioxygenase; and uroporphyrinogen decarboxylase. |
|
Purification Methods |
Protoporphyrin IX (3,18-divinyl-2,7,13,17-tetramethylporphin-8,12-dipropionic acid, ooporphyrin) [553-12-8] M 562.7, pKEst ~ 4.8. Protoporphyrin IX is purified by dissolving (4g) in 98-100% HCOOH (85mL), diluting with dry Et2O (700mL) and keeping at 0o overnight. The precipitate is collected and washed with Et2O, then H2O, and dried in a vacuum at 50o over P2O5. It crystallises from aqueous pyridine and from Et2O in monoclinic, brownish-yellow prisms. The UV max values in 25% HCl are 557.2, 582.2 and 602.4nm. It is freely soluble in ethanolic HCl, AcOH, CHCl3, and Et2O containing AcOH. It forms sparingly soluble diNa and diK salts. [Ramsey Biochemical Preparations 3 39 1953, UV: Holden Aust J. Exptl Biol and Med Sci 15 412 1937, Garnick J Biol Chem 175 333 1948, IR: Falk & Willis Aust J Sci Res [A] 4 579 1951, Beilstein 26 IV 3042.] |
|
Toxicity evaluation |
protoporphyrin IX, is highly toxic in the presence of light and molecular oxygen, killing photosynthetic plants very quickly through the generation of singlet oxygen. |
|
Definition |
ChEBI: A cyclic tetrapyrrole that consists of porphyrin bearing four methyl substituents at positions 3, 8, 13 and 17, two vinyl substituents at positions 7 and 12 and two 2-carboxyethyl substituents at positions 2 and 18. The parent of the class of protoporphyri s. |
|
General Description |
Protoporphyrin IX belongs to the porphyrin family, which are a class of tetrapyrroles. It is the iron-free form of hemin and amphiphilic in nature. It is the precursor of heme in its biosynthetic pathway. |
InChI:InChI=1/C34H34N4O4/c1-7-21-17(3)25-13-26-19(5)23(9-11-33(39)40)31(37-26)16-32-24(10-12-34(41)42)20(6)28(38-32)15-30-22(8-2)18(4)27(36-30)14-29(21)35-25/h7-8,13-16,35-36H,1-2,9-12H2,3-6H3,(H,39,40)(H,41,42)/b25-13u,26-13-,27-14-,28-15u,29-14u,30-15-,31-16u,32-16-
In an effort to asses the effect of Val3...
The photooxidation of protoporphyrin IX ...
-
Iron accumulation in substantia nigra pa...
The components of haematoporphyrin deriv...
-
Defects in the human protoporphyrinogen ...
Components of 'haematoporphyrin derivati...
One unsaturated polymer support was prep...
Heme is an essential cofactor and signal...
HemN is a radical S-adenosyl-l-methionin...
The present application relates generall...
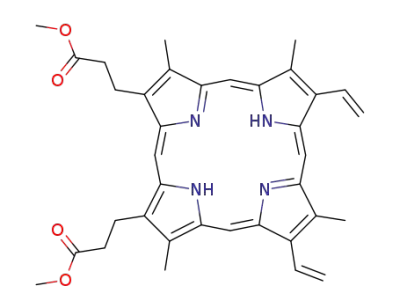
protoporphyrin IX dimethyl ester

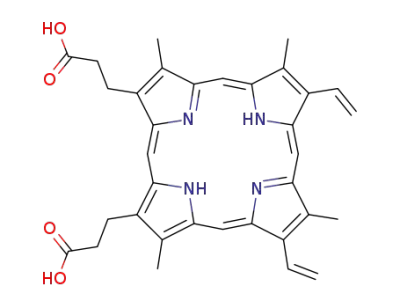
protoporphyrin IX
| Conditions | Yield |
|---|---|
|
With
potassium hydroxide;
In
methanol;
Reflux;
Inert atmosphere;
Darkness;
|
76.6% |
|
With
hydrogenchloride;
In
water;
|
|
|
With
zinc diacetate;
In
methanol; dichloromethane; chloroform; water;
|
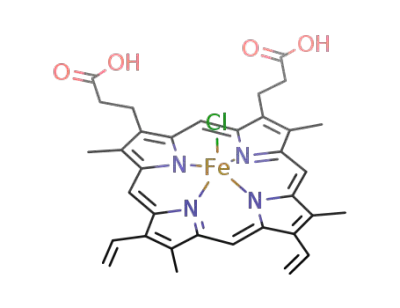
hemin


protoporphyrin IX
| Conditions | Yield |
|---|---|
|
With
ammonium hydroxide; phosphoric acid tributyl ester; sulfuric acid;
In
benzene;
at 40 ℃;
for 1.5h;
|
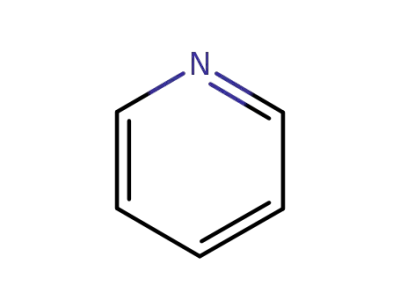
pyridine
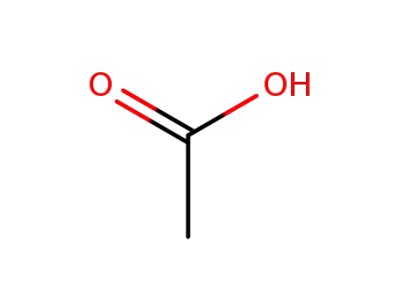
acetic acid
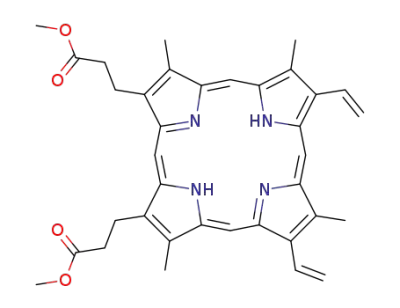
3,3'-(3,7,12,17-tetramethyl-8,13-divinyl-21H,23H-porphine-2,18-diyl)-bis-propionic acid dimethyl ester
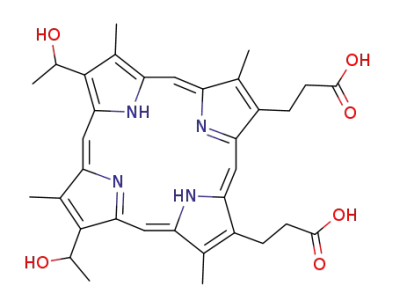
hematoporphyrin

3,3'-(3,7,12,17-tetramethyl-8,13-divinyl-21H,23H-porphine-2,18-diyl)-bis-propionic acid dimethyl ester
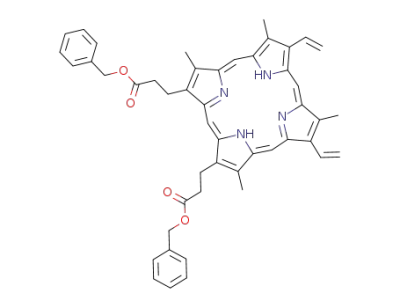
protoporphyrin-dibenzyl ester
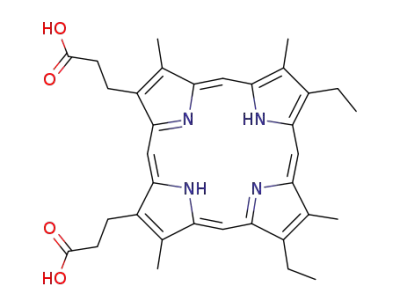
mesoporphyrin IX
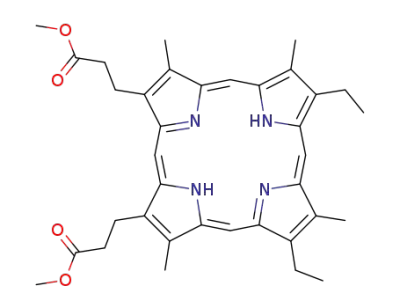
mesoporphyrin IX dimethyl ester
CAS:112163-33-4
CAS:112-84-5
CAS:956025-83-5
CAS:71989-17-8