- +86-0533-2185556
- WhatsApp: +86 15965530500
- admin@hangyubiotech.com
Your Location:Home >Products >API >136247-07-9
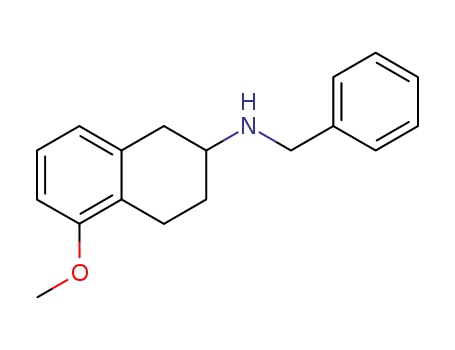

Purity:99%
|
General Description |
Rotigotine is a chemical compound that falls under the category of dopamine agonists. It is used as a medication for the treatment of Parkinson's disease and restless legs syndrome. The chemical structure of rotigotine consists of a 5-methoxy-1,2,3,4-tetrahydro-N-(phenylmethyl)- 2-Naphthalenamine. 5-methoxy-1,2,3,4-tetrahydro-N-(phenylmethyl)- 2-Naphthalenamine (Rotigotine) acts as a dopamine receptor agonist, specifically targeting the D2, D3, and D4 dopamine receptors in the brain. By stimulating these receptors, rotigotine helps to alleviate the motor symptoms of Parkinson's disease, such as tremors, stiffness, and slow movements, as well as reduce the symptoms of restless legs syndrome. It is typically administered as a transdermal patch for continuous delivery of the medication over a 24-hour period. |
InChI:InChI=1/C18H21NO/c1-20-18-9-5-8-15-12-16(10-11-17(15)18)19-13-14-6-3-2-4-7-14/h2-9,16,19H,10-13H2,1H3
The present invention relates to heteroc...
The present invention relates to heteroc...
The present invention refers to a combin...
Novel 2-aminotetralin derivatives were s...
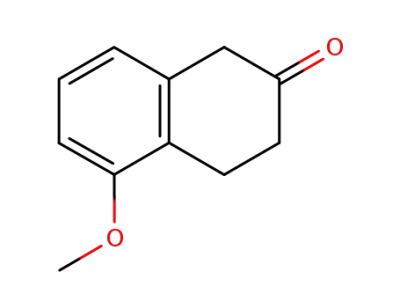
5-methoxy-2-tetralone

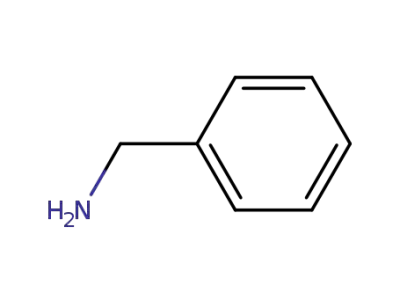
benzylamine

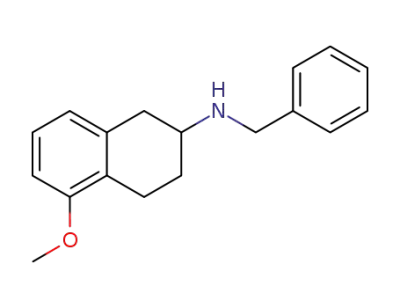
2-(N-benzylamino)-5-methoxytetralin
| Conditions | Yield |
|---|---|
|
5-methoxy-2-tetralone; benzylamine;
With
acetic acid;
In
dichloromethane;
at 20 ℃;
for 4h;
With
sodium tris(acetoxy)borohydride;
In
dichloromethane;
at 0 - 20 ℃;
for 15.5h;
With
water; sodium hydrogencarbonate;
In
dichloromethane;
at 0 ℃;
for 0.25h;
pH=8.0;
|
74% |
|
5-methoxy-2-tetralone; benzylamine;
With
acetic acid;
In
dichloromethane;
at 20 ℃;
for 4h;
With
sodium tris(acetoxy)borohydride;
In
dichloromethane;
at 0 - 20 ℃;
for 16.83h;
With
sodium hydrogencarbonate;
In
dichloromethane; water;
at 0 ℃;
for 0.25h;
pH=8.0;
|
74% |
|
5-methoxy-2-tetralone; benzylamine;
With
acetic acid;
In
dichloromethane;
at 20 ℃;
for 4h;
With
sodium tris(acetoxy)borohydride;
In
dichloromethane;
at 0 - 20 ℃;
for 16.8333h;
With
sodium hydrogencarbonate;
In
dichloromethane; water;
at 0 ℃;
for 0.25h;
pH=8.0;
|
74% |
|
With
hydrogen; toluene-4-sulfonic acid;
platinum(IV) oxide;
Yield given. Multistep reaction;
1.) toluene, reflux, 2 h, 2.) EtOH, 32 psi, 1 h;
|
|
|
With
sodium tris(acetoxy)borohydride;
In
1,2-dichloro-ethane;
at 20 ℃;
|
![Benzyl-[5-methoxy-3,4-dihydro-1H-naphthalen-(2E)-ylidene]-amine](/upload/2026/5/ef14013a-7183-4490-abe2-9cacf77d82f3.png)
Benzyl-[5-methoxy-3,4-dihydro-1H-naphthalen-(2E)-ylidene]-amine


2-(N-benzylamino)-5-methoxytetralin
| Conditions | Yield |
|---|---|
|
With
hydrogen;
platinum(IV) oxide;
In
ethanol;
for 2.5h;
under 1520 Torr;
|

5-methoxy-2-tetralone

benzylamine
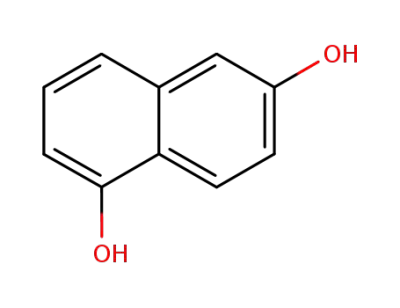
1,6-dihydroxynaphthalene
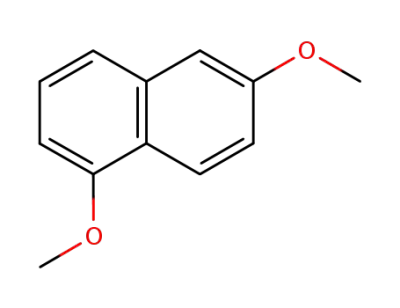
1,6-dimethoxynaphthalene
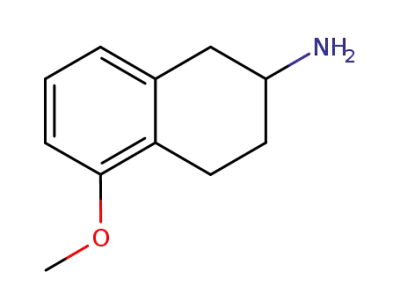
2-Amino-5-methoxy-1,2,3,4-tetrahydronaphthalene
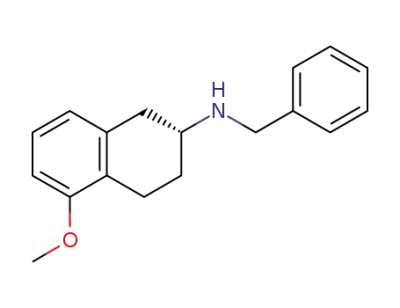
(R)-(+)-2-(N-benzylamino)-5-methoxytetralin
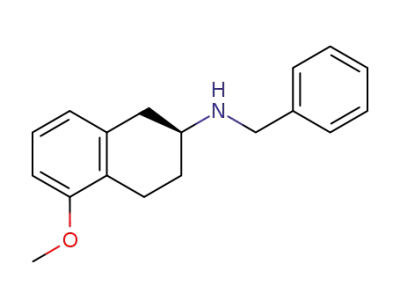
(S)-(-)-2-(N-benzylamino)-5-methoxytetralin
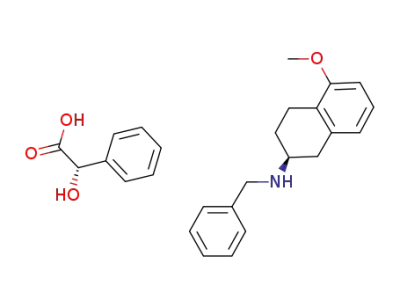
C8H8O3*C18H21NO
CAS:112163-33-4
CAS:112-84-5
CAS:783348-36-7
CAS:5585-96-6