- +86-0533-2185556
- WhatsApp: +86 15965530500
- admin@hangyubiotech.com
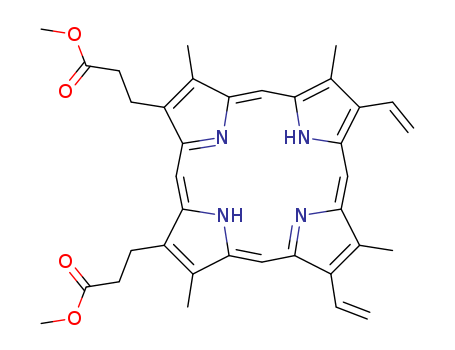

pd_meltingpoint:225-228 °C(lit.)
Purity:99%
|
Purification Methods |
The crude dimethyl ester (1g) in CHCl3 (200 mL) is mixed with pet ether (b 70-90o, 600mL), and any porphyrin (m > 260o) which is insoluble in this mixture is filtered off. The filtrate is passed through a column of CaCO3 [from CaCO3 (130g) which is kept overnight in a mixture of CHCl3/pet ether (b 70-90o, 1:3), and the slurry is poured into a glass tube (2.5 x 26cm) to form the column]. After all the filtrate is applied, the column is eluted with a solution of CHCl3/pet ether (b 70-90o, 1:3). All the coloured eluates are collected, evaporated at room temperature in a vacuum to give a residue (0.8g), m 208-211o. The residue (0.8g) in CHCl3 (66mL) is heated briefly to its boiling point, then boiling MeOH (198mL) is added immediately to it. The mixture is allowed to cool to room temperature, refrigerated for 2days and the solid is filtered off. The solid is washed on the filter funnel with CHCl3/MeOH (1:9, 50mL) and dried at 50o/vacuum (yield 0.62-0.66g). It can also be recrystallised by dissolving in as little hot dry *C6H6 as possible and left overnight at 20o, m 228-230o. It has also been purified by dissolving (0.4g) in CHCl3 (33mL) by boiling for a few minutes, then diluting with boiling MeOH (100mL) and refrigerating for 2days. The crystals are collected, washed with CHCl3/MeOH (1:9) and dried at 50o in a vacuum (yield 0.3g). The UV has max at 631, 576, 541, 506 and 407nm in CHCl3 and 601, 556 and 406nm in 25% HCl. [Ramsey Biochemical Preparations 3 39 1953, Beilstein 26 III/IV 3052.] |
InChI:InChI=1/C36H38N4O4/c1-9-23-19(3)27-15-28-21(5)25(11-13-35(41)43-7)33(39-28)18-34-26(12-14-36(42)44-8)22(6)30(40-34)17-32-24(10-2)20(4)29(38-32)16-31(23)37-27/h9-10,15-18,37,40H,1-2,11-14H2,3-8H3/b27-15-,28-15-,29-16-,30-17-,31-16-,32-17-,33-18-,34-18-
-
Herein, we report the functionalization ...
The substitution pattern of the two majo...
The aim of this work is the synthesis of...
The first synthesis of α-amino phosphona...
Protoporphyrin upon its binding with ser...
This work is focused on the evaluation o...
-
-
-
We report the photosensitization of crys...
The first syntheses of a series of regio...
Poly-trans-[(2-carboxyethyl) germasesqui...
Protoporphyrin IX (PPIX) lipids were syn...
A novel series of boronated porphyrins f...
The presence of carbon linked oligomeric...
It was previously reported that photon u...
Iodination of protoporphyrin IX dimethyl...
methanol

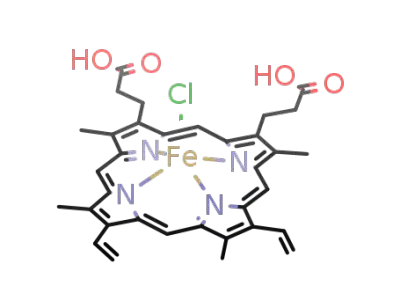
hemin

3,3'-(3,7,12,17-tetramethyl-8,13-divinyl-21H,23H-porphine-2,18-diyl)-bis-propionic acid dimethyl ester
| Conditions | Yield |
|---|---|
|
With
pyridine; diammonium iron(II) sulfate hexahydrate; acetyl chloride;
In
dichloromethane;
for 1h;
Cooling;
|
99% |

methanol

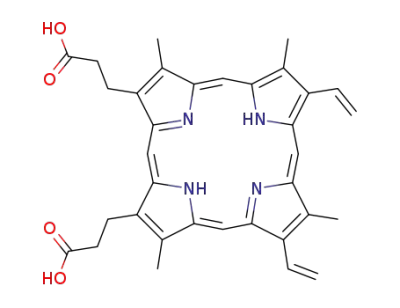
protoporphyrin IX

3,3'-(3,7,12,17-tetramethyl-8,13-divinyl-21H,23H-porphine-2,18-diyl)-bis-propionic acid dimethyl ester
| Conditions | Yield |
|---|---|
|
at 20 ℃;
for 6h;
|
98% |
|
With
sulfuric acid;
at -10 ℃;
for 18h;
Inert atmosphere;
Schlenk technique;
Darkness;
|
95% |
|
With
trimethyl orthoformate;
sulfuric acid;
for 0.75h;
Ambient temperature;
|
71 % Chromat. |
|
With
sulfuric acid;
|
|
|
With
sulfuric acid;
at -10 ℃;
for 18h;
Inert atmosphere;
Darkness;
|
|
|
With
sulfuric acid;
at 20 ℃;
for 12h;
Darkness;
|
1.4 g |
|
With
hydrogenchloride;
|

diazomethane

protoporphyrin IX
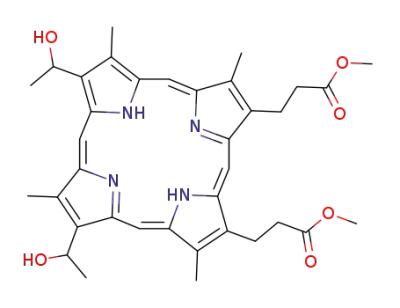
hematoporphyrin-dimethyl ester
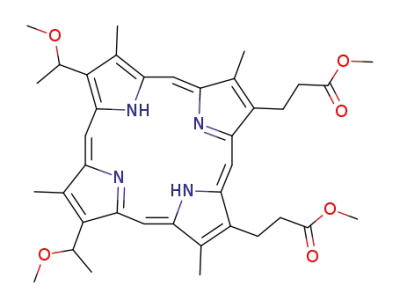
protoporphyrin

protoporphyrin IX
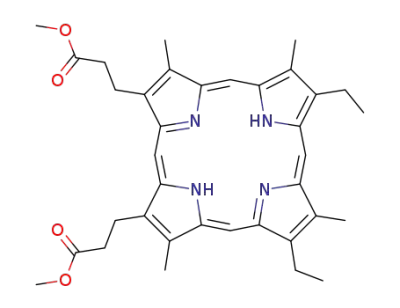
mesoporphyrin IX dimethyl ester
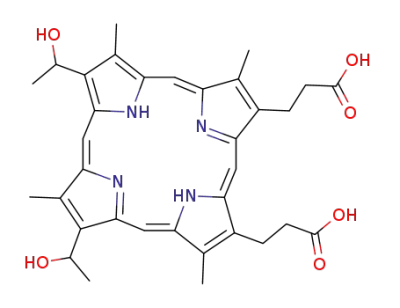
hematoporphyrin
mesoporphyrin IX
CAS:112163-33-4
CAS:112-84-5
CAS:144337-18-8
CAS:68929-05-5