- +86-0533-2185556
- WhatsApp: +86 15965530500
- admin@hangyubiotech.com
Your Location:Home >Products >API >90729-43-4
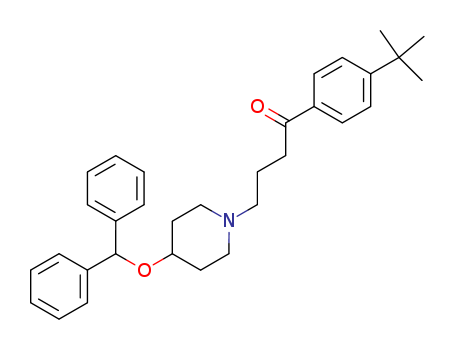

pd_meltingpoint:80-82 °C
Appearance:White to off-white powder
Purity:99%
|
Manufacturing Process |
(a) A mixture of 4-hydroxypiperidine (40.4 g; 0.4 moles), p-tert-butyl-ω- chlorobutyrophenone (105 g, 0.44 moles), sodium bicarbonate (67.2 g; 0.8 moles) and a crystal of potassium iodide in methyl isobutyl ketone (1 liter) was boiled under reflux for 24 hours. After cooling, the reaction mixture waswashed with water, dried (Na2SO4) and the solvent removed in vacuum. The residue was salified with the stoichiometric amount of fumaric acid in a mixture of acetone and ethanol to give 1-[3-(4-tert-butylbenzoyl)propyl]-4- hydroxypiperidine fumarate (148 g), melting point 163-165°C. Ebastine was converted into the free base, and 1-[3-(4-tert-butylbenzoyl)propyl]-4- hydroxypiperidine was obtained and recrystallized from a mixture of diethyl ether and petroleum ether (boiling point 50-70°C). 102 g were obtained (yield 84%), melting point 63-65°C.(b) A mixture of 1-[3-(tert-butylbenzoyl)propyl]-4-hydroxypiperidine (60.68 g; 0.2 moles) and sodium carbonate (42.4 g; 0.4 moles) in methyl isobutyl ketone (500 ml) was heated to the boiling point and a solution of diphenylmethyl bromide (49.42 g; 0.2 moles) in methyl isobutyl ketone (75 ml) was slowly added in 1.5 hours. The resulting mixture was boiled under reflux for another 12 hours, and then another solution of diphenylmethyl bromide (24.71 g; 0.1 moles) in methyl isobutyl ketone (50 ml) was added and the mixture boiled under reflux again for 12 hours. Another solution of diphenylmethyl bromide in the same quantity was added and after refluxing for 12 additional hours the reaction mixture was cooled, washed with water, dried (Na2SO4) and the solvent removed in vacuum.The residual oil was treated with the stoichiometric amount of fumaric acid in ethanol and 4-diphenylmethoxy-1-[3-(4-tert-butylbenzoyl)propyl]piperidine fumarate crystallized. After recrystallisation from ethanol the pure compound was obtained (88 g; yield 75%), melting point 197-198°C. |
|
Therapeutic Function |
Antihistaminic, Antiallergic, Calcium entry blocker |
|
Biochem/physiol Actions |
Ebastine is a non-sedating histamine H1 receptor antagonist, which inhibits allergen-induced bronchospasm in conscious guinea pigs. Unlike other compounds in this category, ebastine does not prolong the QT interval at up to five times the recommended therapeutic dose. |
|
Brand name |
Kestine (Rhone-Poulenc Rorer);Ebastel. |
|
General Description |
Ebastine is metabolised by cytochrome P450 3A (CYP3A4) to carebastine. It is used to treat allergic rhinitis and chronic idiopathic urticaria. |
InChI:InChI=1/C32H39NO2/c1-32(2,3)28-18-16-25(17-19-28)30(34)15-10-22-33-23-20-29(21-24-33)35-31(26-11-6-4-7-12-26)27-13-8-5-9-14-27/h4-9,11-14,16-19,29,31H,10,15,20-24H2,1-3H3
The invention provides a method for prep...
The present invention relates to a novel...
The present invention describes processe...
The present invention describes processe...
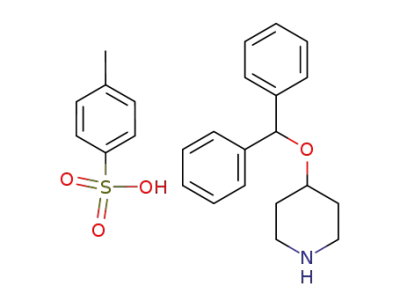
4-benzhydryloxypiperidine toluenesulfonate

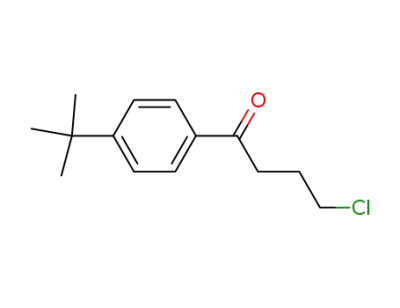
4'-tert-butyl-4-chlorobutyrophenone

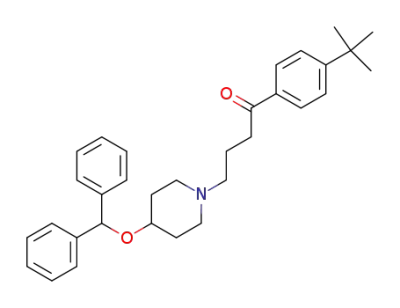
ebastine
| Conditions | Yield |
|---|---|
|
With
potassium carbonate;
In
4-methyl-2-pentanone;
at 105 - 116 ℃;
for 2.25h;
Inert atmosphere;
|
55.27% |
|
With
potassium carbonate;
In
4-methyl-2-pentanone;
at 105 - 116 ℃;
Inert atmosphere;
|
55.27% |
![4-diphenylmethoxy-1-[3-[2-(4-tert-butylphenyl)-1,3-dioxolan-2-yl]propyl]piperidine](/upload/2026/5/1a1f70cb-c2f0-4c79-8b89-be2ffcbb1f91.png)
4-diphenylmethoxy-1-[3-[2-(4-tert-butylphenyl)-1,3-dioxolan-2-yl]propyl]piperidine


ebastine
| Conditions | Yield |
|---|---|
|
In
hydrogenchloride; ethanol;
|
87.3% |
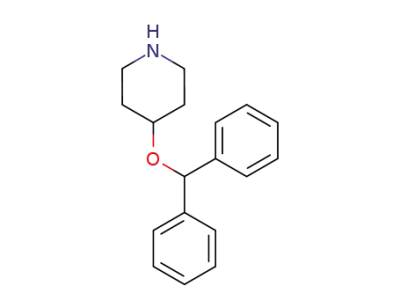
4-diphenylmethoxypiperidine

4'-tert-butyl-4-chlorobutyrophenone
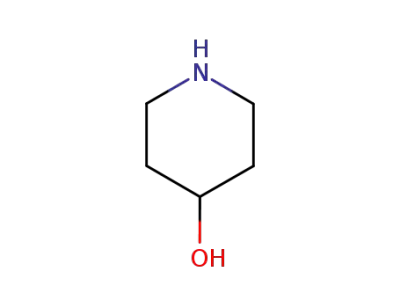
4-HYDROXYPIPERIDINE
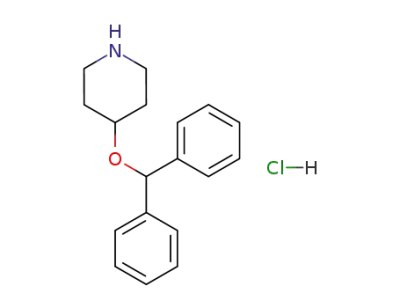
4-(diphenylmethoxy)piperidine hydrochloride
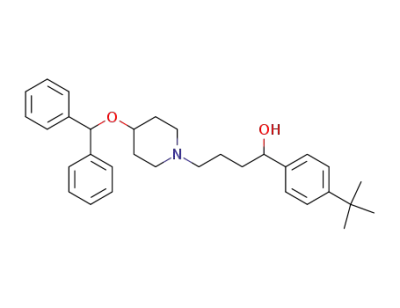
1-(4-tert-butylphenyl)-4-(4-(benzhydryloxy)piperidin-1-yl)butan-1-ol
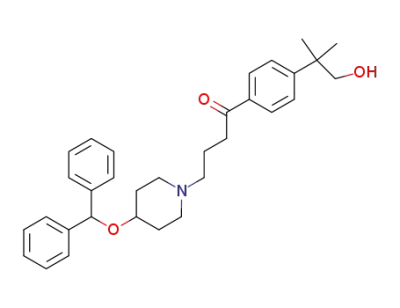
Hydroxyebastine
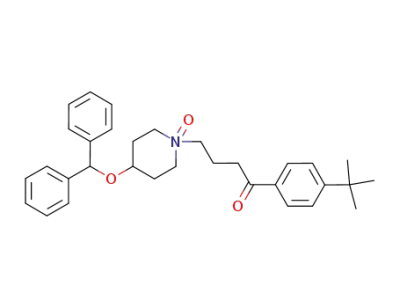
ebastine N-oxide
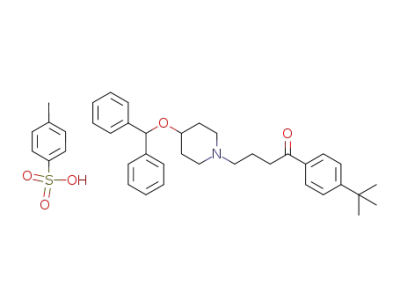
1-[4-(1,1-dimethylethyl)phenyl]-4-[4-(diphenylmethoxy)-1-piperidinyl]-1-butanone tosylate
CAS:112163-33-4
CAS:112-84-5
CAS:68929-05-5
CAS:6138-79-0