- +86-0533-2185556
- WhatsApp: +86 15965530500
- admin@hangyubiotech.com

pd_meltingpoint:-66.5 °C(lit.)
Appearance:colourless liquid
Purity:99%
|
Synthesis |
An efficient method for the preparation of IODOMETHANE-D3, using METHANOL-D3 and iodine monomers as reaction materials, in a hydrogen atmosphere, with the addition of transition metal catalysts and ligands to produce IODOMETHANE-D3 in situ at 0°C-120°C. |
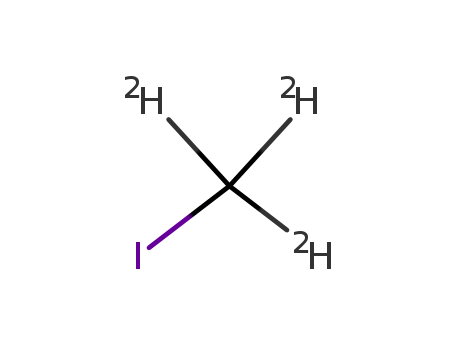
InChI:InChI=1/CH3I/c1-2/h1H3/i1D3
-
The invention discloses a method for eff...
The frustrated Lewis pair (FLP) derived ...
Trialkylammonium (most notably N,N,N-tri...
Herein, a novel approach for the trideut...
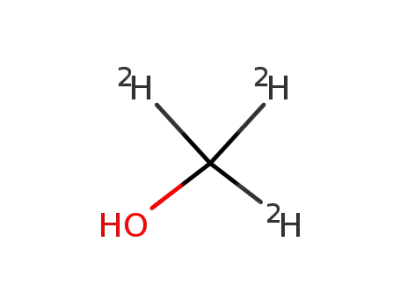
1,1,1-trideuteromethanol

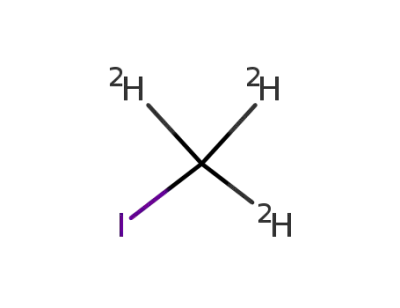
iodomethane-d3
| Conditions | Yield |
|---|---|
|
With
bis(1,5-cyclooctadiene)diiridium(I) dichloride; hydrogen; iodine; triphenylphosphine;
In
tetrahydrofuran;
at 20 ℃;
under 15001.5 Torr;
Reagent/catalyst;
Solvent;
Pressure;
Temperature;
Autoclave;
|
88% |
|
With
hydrogen iodide;
In
water;
at 20 - 55 ℃;
|
83.8% |
|
With
phosphorus; iodine;
|
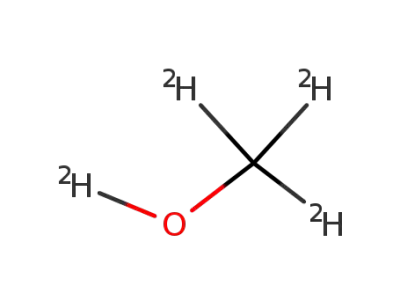
d(4)-methanol


iodomethane-d3
| Conditions | Yield |
|---|---|
|
With
phosphorus; iodine;
In
water;
Heating;
|
85% |
|
With
hydrogen iodide;
In
water;
at 40 - 50 ℃;
for 4h;
|
83.8% |
|
With
phosphorus; iodine;
Heating;
|
69.8% |
|
With
iodine;
|
62% |
|
With
phosphorus; iodine;
Heating;
|
|
|
With
hydrogen iodide;
at 60 - 70 ℃;
for 1h;
|
17.35 g |
|
With
phosphorus; iodine;
Reflux;
|
|
|
With
phosphorus; iodine;
at 0 ℃;
|

1,1,1-trideuteromethanol
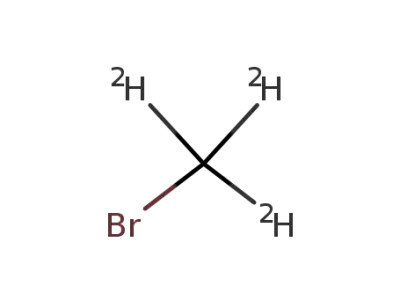
Trideutero-methylbromid
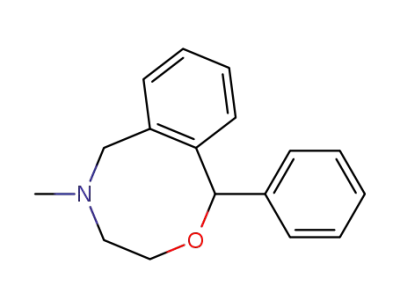
nefopam

d(4)-methanol
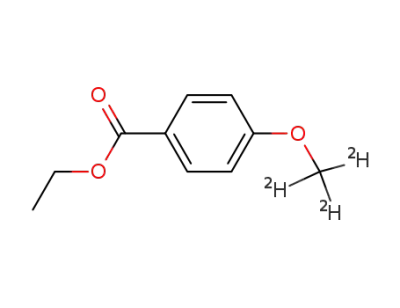
p-(trideuteromethoxy)benzoic acid ethyl ester
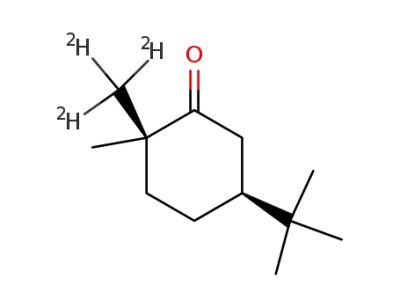
5-tert-butyl-2-methyl-d3-2-methylcyclohexanone
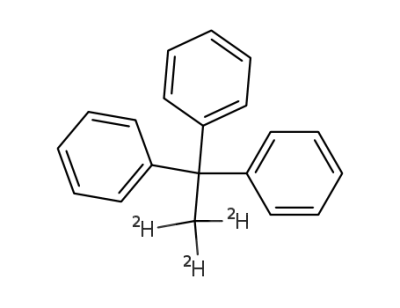
Trideutero-1.1.1-triphenyl-aethan
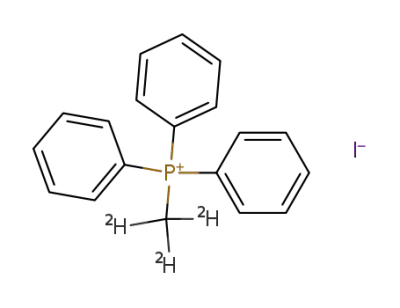
(methyl-d3)triphenylphosphonium iodide
CAS:7436-22-8
CAS:1859-09-2
CAS:1601496-05-2
CAS:25070-79-5