- +86-0533-2185556
- WhatsApp: +86 15965530500
- admin@hangyubiotech.com
Your Location:Home >Products >API >10589-94-3
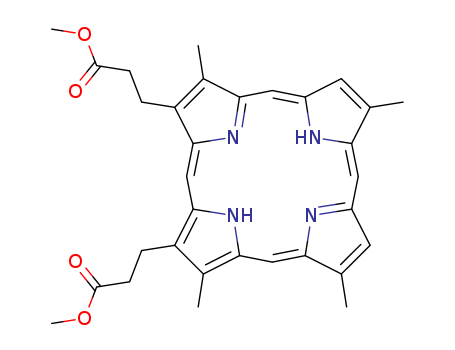

pd_meltingpoint:217-220 °C(lit.)
Purity:99%
InChI:InChI=1/C32H34N4O4/c1-17-11-22-14-27-19(3)23(7-9-31(37)39-5)29(35-27)16-30-24(8-10-32(38)40-6)20(4)28(36-30)15-26-18(2)12-21(34-26)13-25(17)33-22/h11-16,34-35H,7-10H2,1-6H3/b21-13-,22-14-,25-13-,26-15-,27-14-,28-15-,29-16-,30-16-
Deuteroporphyrin-niacin dyads with diffe...
-
A novel thiol-derivative porphyrin [2,7,...
A facile, efficient and general method f...
The vanadyl porphyrins from Boscan oil (...
We present a broad study of the effect o...
Eight porphyrin nitrogen mustards and si...
By short treatment of zinc(II) deuteropo...
-
-
-
The N-protonations in the deutero, hemat...
A series of cobalt porphyrins derived fr...
A facile biomimetic method for rapid oxi...
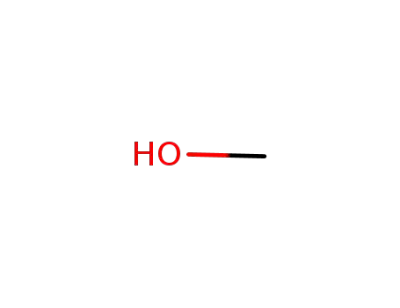
methanol

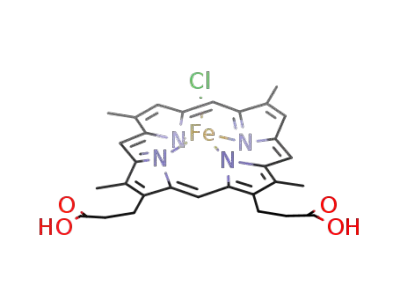
deuterohemin chloride

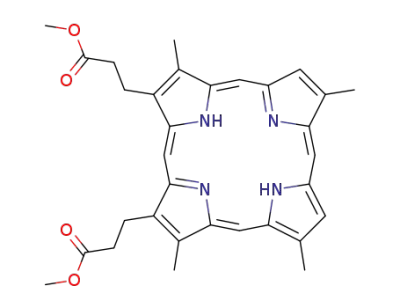
deuteroporphyrin IX dimethyl ester
| Conditions | Yield |
|---|---|
|
With
sulfuric acid;
at 20 ℃;
Sonication;
|
97% |

methanol

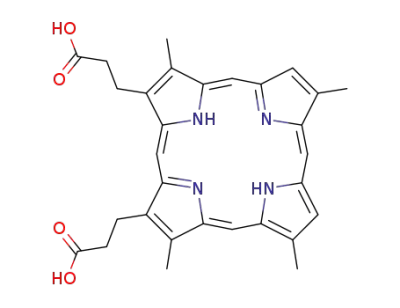
deuteroporphyrin IX


deuteroporphyrin IX dimethyl ester
| Conditions | Yield |
|---|---|
|
With
sulfuric acid;
for 18h;
Ambient temperature;
|
93.4% |
|
With
hydrogenchloride;
|
|
|
With
hydrogenchloride;
|
|
|
With
sulfuric acid;
for 1h;
Irradiation;
|

diazomethane

methanol
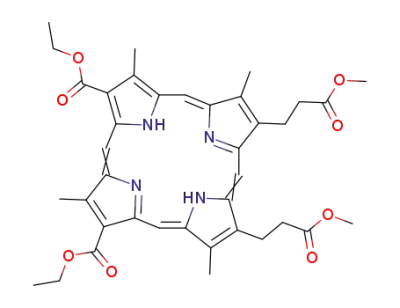
diethyl 2,7,12,18-tetramethyl-13,17-bis<β-(methoxycarbonyl)ethyl>porphyrin-3,8-dicarboxylate

deuterohemin chloride
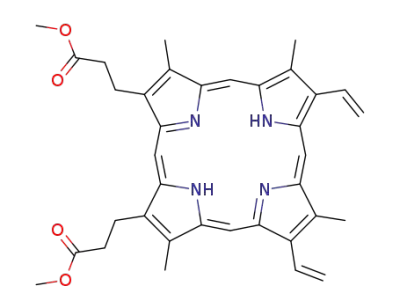
3,3'-(3,7,12,17-tetramethyl-8,13-divinyl-21H,23H-porphine-2,18-diyl)-bis-propionic acid dimethyl ester
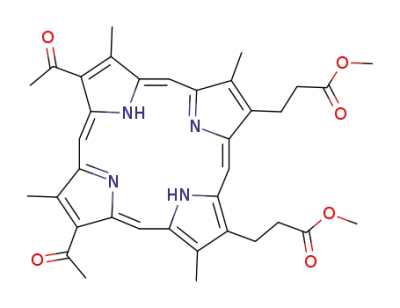
2,4-diacetyl-6,7-bis<2-(methoxycarbonyl)ethyl>-1,3,5,8-tetramethylporphyrin
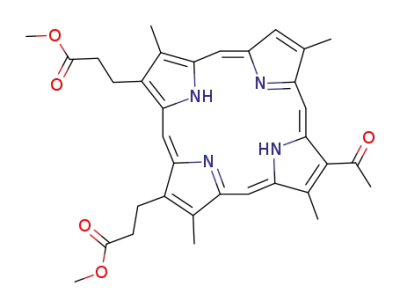
3,3'-<3-Acetyl-2,7,12,18-tetramethyl-21H,23H-porphyrin-13,17-diyl>dipropionsaeure-dimethylester
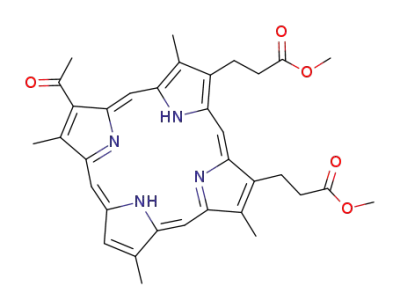
3,3'-<8-Acetyl-2,7,12,18-tetramethyl-21H,23H-porphyrin-13,17-diyl>dipropionsaeure-dimethylester
CAS:112163-33-4
CAS:112-84-5
CAS:313-06-4
CAS:4617-99-6