- +86-0533-2185556
- WhatsApp: +86 15965530500
- admin@hangyubiotech.com
Your Location:Home >Products >API >1187595-90-9

Purity:99%
The invention relates to synthesis of ba...
The present disclosure provides a Barici...
Nine compounds potentially related to th...
Method and intermediates are disclosed f...
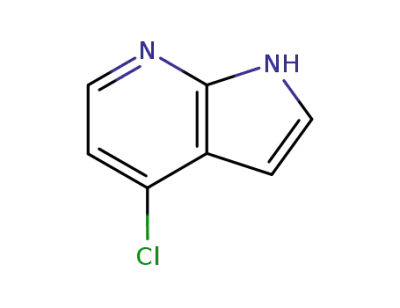
4-chloro-7-azaindole

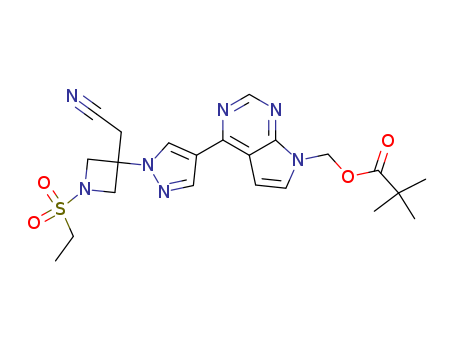
![(4-{1-[3-(cyanomethyl)-1-(ethylsulphonyl)azetidin-3-yl]-1H-pyrazol-4-yl}-7H-pyrrolo[2,3-d]pyrimidin-7-yl)methyl 2,2-dimethylpropanoate](/upload/2026/5/68166035-71e9-4e1e-8194-ad9b737beec7.png)
(4-{1-[3-(cyanomethyl)-1-(ethylsulphonyl)azetidin-3-yl]-1H-pyrazol-4-yl}-7H-pyrrolo[2,3-d]pyrimidin-7-yl)methyl 2,2-dimethylpropanoate
| Conditions | Yield |
|---|---|
|
Multi-step reaction with 4 steps
1: potassium carbonate / N,N-dimethyl-formamide / 16 h / 20 °C
2: bis-triphenylphosphine-palladium(II) chloride; potassium carbonate / 1,4-dioxane; water / 3 h / 72 °C / Inert atmosphere
3: acetyl chloride / methanol / 6 h / 5 - 20 °C
4: 1,8-diazabicyclo[5.4.0]undec-7-ene / N,N-dimethyl-formamide / 3 h / 5 - 10 °C
With
bis-triphenylphosphine-palladium(II) chloride; potassium carbonate; 1,8-diazabicyclo[5.4.0]undec-7-ene; acetyl chloride;
In
1,4-dioxane; methanol; water; N,N-dimethyl-formamide;
|
|
|
Multi-step reaction with 4 steps
1: potassium carbonate / N,N-dimethyl-formamide / 16 h / 20 °C
2: bis(1,5-cyclooctadiene)nickel (0); tricyclohexylphosphine; potassium carbonate / water; tert-butyl alcohol / 3 h / 40 °C / Glovebox
3: hydrogenchloride / water; tetrahydrofuran / 6 h / 5 - 20 °C
4: 1,8-diazabicyclo[5.4.0]undec-7-ene / N,N-dimethyl-formamide / 3 h / 5 - 10 °C
With
hydrogenchloride; bis(1,5-cyclooctadiene)nickel (0); potassium carbonate; 1,8-diazabicyclo[5.4.0]undec-7-ene; tricyclohexylphosphine;
In
tetrahydrofuran; water; N,N-dimethyl-formamide; tert-butyl alcohol;
|
![(4-{1-[3-(cyanomethyl)azetidin-3-yl]-1H-pyrazol-4-yl}-7H-pyrrolo[2,3-d]pyrimidin-7-yl)methyl 2,2-dimethylpropanoate](/upload/2026/5/6a990e17-e016-4dd4-8c56-703ff79c64a5.png)
(4-{1-[3-(cyanomethyl)azetidin-3-yl]-1H-pyrazol-4-yl}-7H-pyrrolo[2,3-d]pyrimidin-7-yl)methyl 2,2-dimethylpropanoate

![(4-{1-[3-(cyanomethyl)-1-(ethylsulphonyl)azetidin-3-yl]-1H-pyrazol-4-yl}-7H-pyrrolo[2,3-d]pyrimidin-7-yl)methyl 2,2-dimethylpropanoate](/upload/2026/5/68166035-71e9-4e1e-8194-ad9b737beec7.png)
(4-{1-[3-(cyanomethyl)-1-(ethylsulphonyl)azetidin-3-yl]-1H-pyrazol-4-yl}-7H-pyrrolo[2,3-d]pyrimidin-7-yl)methyl 2,2-dimethylpropanoate
| Conditions | Yield |
|---|---|
|
With
N-ethyl-N,N-diisopropylamine; Ethanesulfonyl chloride;
In
ethyl acetate;
at 0 - 20 ℃;
for 1.5h;
Solvent;
Temperature;
|
95.7% |
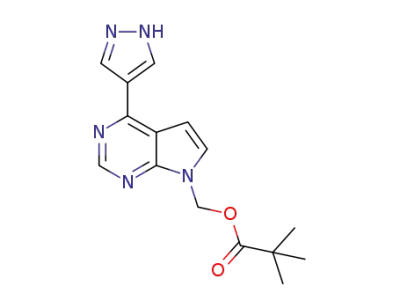
[4-(1H-pyrazol-4-yl)-7H-pyrrolo[2,3-d]pyrimidin-7-yl]methyl 2,2-dimethylpropanoate
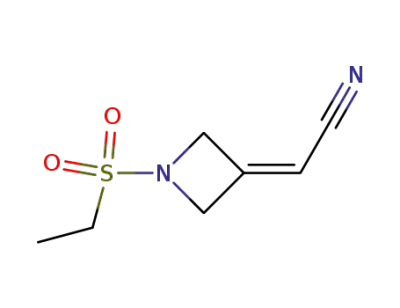
2-[1-(ethanesulfonyl)azetidin-3-ylidene]acetonitrile
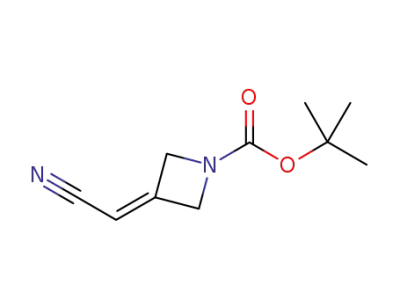
3-(cyanomethylene)azetidine-1-carboxylic acid tert-butyl ester
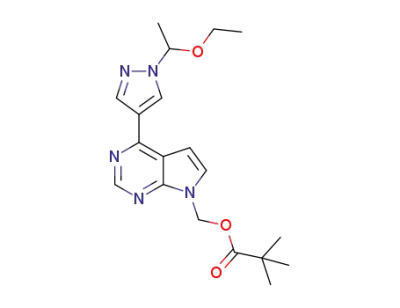
[4-[1-(1-ethoxyethyl)-1H-pyrazol-4-yl]-7H-pyrrolo[2,3-d]pyrimidin-7-yl]methyl pivalate
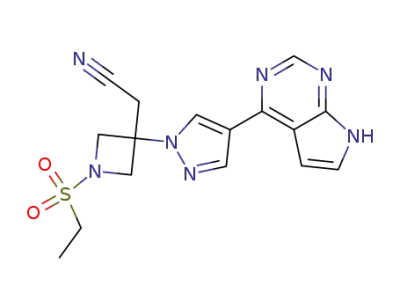
Baricitinib
CAS:11130-12-4
CAS:1445566-01-7
CAS:516-54-1
CAS:1044589-82-3