- +86-0533-2185556
- WhatsApp: +86 15965530500
- admin@hangyubiotech.com
Your Location:Home >Products >API >24533-72-0

Purity:99%
|
General Description |
Pyropheophorbide a is a chlorophyll derivative that has been identified as a potential photosensitizer in photodynamic therapy, a treatment for various types of cancer. pyropheophorbide a is known for its ability to generate reactive oxygen species when exposed to light, which can induce cell death in cancerous cells. Pyropheophorbide a has been studied for its potential use in targeting and destroying cancer cells while sparing healthy tissues. Its unique chemical properties make it a promising candidate for further research and development in the field of cancer treatment. Additionally, pyropheophorbide a has also been investigated for its antimicrobial and antioxidant properties, suggesting potential applications in other medical and industrial fields. |
InChI:InChI=1/C33H34N4O3/c1-7-19-15(3)23-12-25-17(5)21(9-10-30(39)40)32(36-25)22-11-29(38)31-18(6)26(37-33(22)31)14-28-20(8-2)16(4)24(35-28)13-27(19)34-23/h7,12-14,17,21,34,37H,1,8-11H2,2-6H3,(H,39,40)/b23-12-,24-13-,25-12-,26-14-,27-13-,28-14-,32-22-/t17-,21-/m0/s1
Chlorophyll a derivatives were integrate...
A series of zinc 31-hydroxymethyl chlori...
Two heterodyads were prepared from the c...
Developing new strategies for controllin...
Self-assembly in a droplet: A supramolec...
Regioselective reduction of the 13-carbo...
The (22R)-30a,30b-dihomohopan-30b-yl and...
Regioselective reduction of the 13-carbo...
Manganese-substituted chlorophyll a deri...
Conjugates containing chlorine and isobo...
A series of carotenoid pyropheophorbide ...
Synthesis, spectral properties, and phot...
A new and simplified method of preparing...
Photodynamic therapy (PDT) is a promisin...
A cobalt chlorin complex (CoII(Ch)) effi...
Pyropheophorbide-a methyl ester (PPa-OMe...
Zinc complex of pyropheophorbide-b, a de...
In the present work, a number of chlorop...

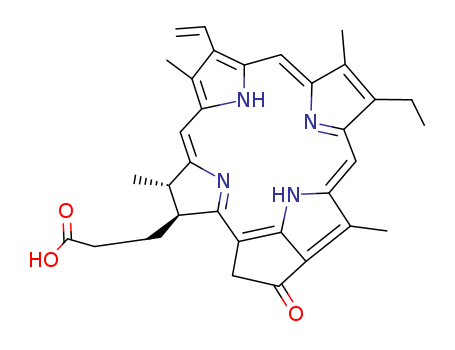
fucoxanthin pyropheophorbide A ester

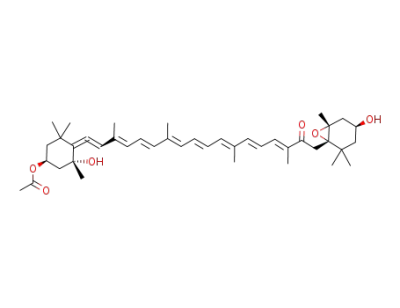
fucoxanthin

pyropheophorbide A
| Conditions | Yield |
|---|---|
|
With
4-oxo-2,2,6,6-tetramethylpiperidin-oxyl;
In
ethanol;
pH=7.5;
aq. phosphate buffer;
|
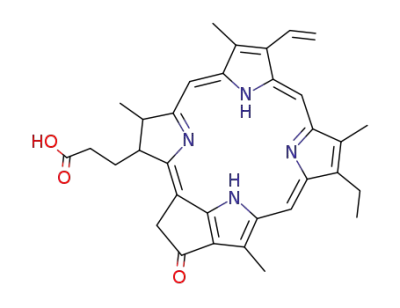
pyrochlorophyll a

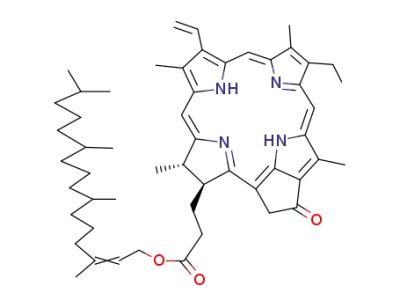
pyropheophytin a

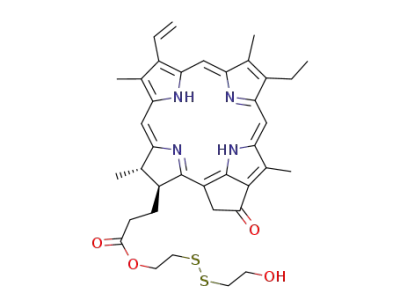
2-hydroxyethyl disulfide pyropheophorbide

pyropheophorbide a
| Conditions | Yield |
|---|---|
|
With
bis(2-hydroxyethyl) disulfide; water;
chlorophyll chlorophyllido-hydrolase; EC 3.1.1.14;
In
acetone;
for 24h;
|
41 % Chromat. 6 % Chromat. 10 % Chromat. |
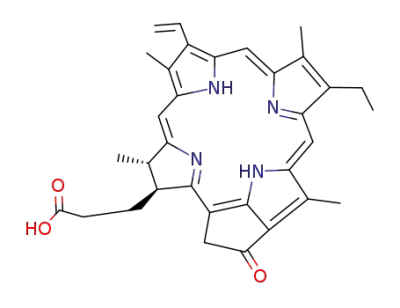
pheophorbide-a
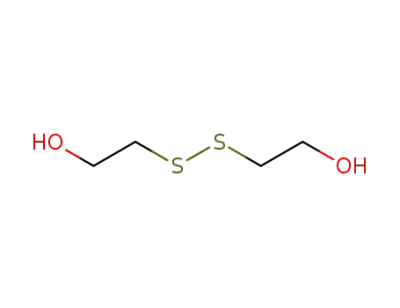
bis(2-hydroxyethyl) disulfide

pyropheophytin a
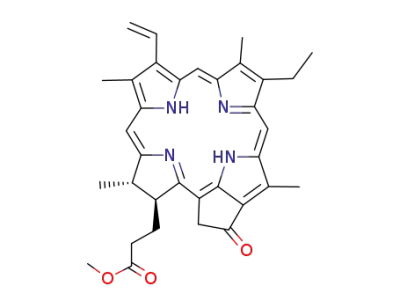
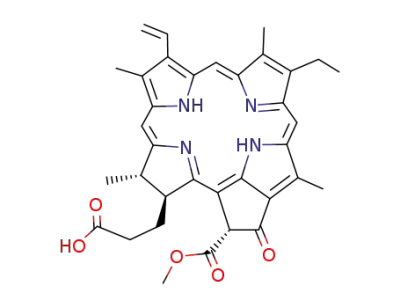
methyl pyropheophorbide a
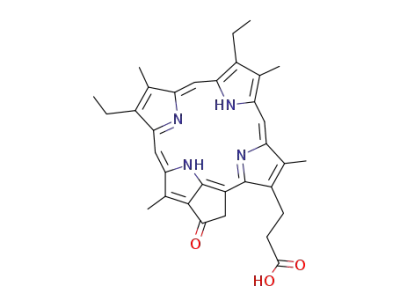
Phytoporphyrin

methyl pyropheophorbide a
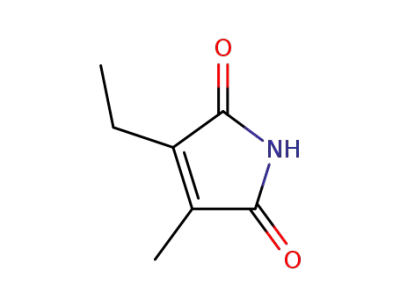
3-ethyl-4-methyl-pyrrole-2,5-dione
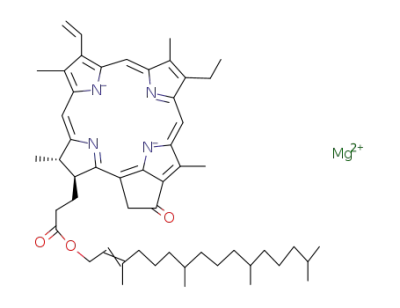
C66H92N4O19Zn
CAS:112163-33-4
CAS:112-84-5
CAS:1624306-20-2
CAS:24445-44-1