- +86-0533-2185556
- WhatsApp: +86 15965530500
- admin@hangyubiotech.com
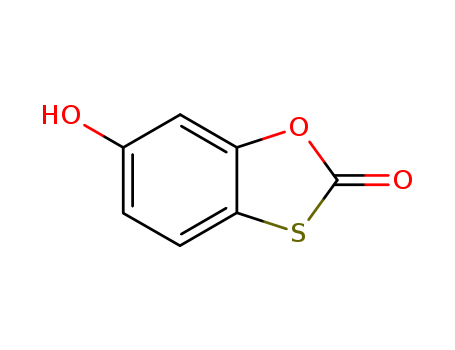

pd_meltingpoint:158-160 °C(lit.)
Appearance:light yellow to beige powder
Purity:99%
|
Manufacturing Process |
40 g potassium thiocyanate in 50 ml of water are added, while stirring at room temperature, to a solution of 11 g of resorcinol and 50 g of crystallized copper sulfate in 250 ml of water. The black cupric thiocyanate formed becomes colorless after a short time, which indicates that the introduction of thiocyanogen is terminated. The cuprous thiocyanate is removed by filtering with suction and then washed with water; the filtrate is mixed with 50 ml of a 2 N sodium carbonate solution, whereby the imino-thiocarbonate of resorcinol separates in the form of a colorless crystalline body. The yield amounts to 16 g. The new compound which melts at 149°C dissolves very easily in many organic solvents and in mineral acids. A 10% solution of the imino-thiocarbonate of resorcinol in 10% hydrochloric acid is heated for 15 min on the steam bath. The 6-hydroxy-1,3-benzoxathiol- 2-one (thiocarbonate free from) nitrogen separates, on cooling, in the form of fine crystals melting at 158°C. |
|
Therapeutic Function |
Antiseborreic, Antifungal, Keratolytic |
|
Definition |
ChEBI: A 1,3-benzoxathiole having a hydroxy substituent at the 6-position. |
|
General Description |
Antimicrobial and cytostatic properties of 6-hydroxy-1,3-benzoxathiol-2-one has been investigated. Supramolecular structure of 6-hydroxy-1,3-benzoxathiol-2-one has been studied. |
InChI:InChI=1/C7H4O3S/c8-4-1-2-6-5(3-4)10-7(9)11-6/h1-3,8H
The present research paper reports the s...
![2-iminobenzo[d][1,3]oxathiol-6-ol](/upload/2026/5/7622c5de-9416-4df2-863b-00f5cabaa70d.png)
2-iminobenzo[d][1,3]oxathiol-6-ol

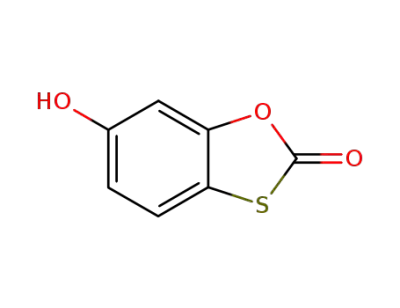
tioxolone
| Conditions | Yield |
|---|---|
|
With
hydrogenchloride;
In
water;
for 0.25h;
Heating;
|
89% |
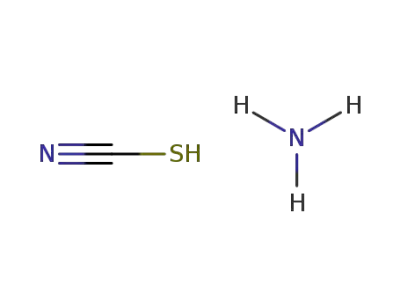
ammonium thiocyanate

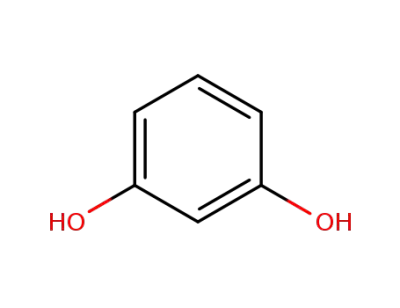
recorcinol


tioxolone
| Conditions | Yield |
|---|---|
|
With
water; copper(II) sulfate;
anschliessend Erwaermen;
|

ammonium thiocyanate

recorcinol
water
copper(II) sulfate
2,4-dihydroxythiophenol
(S)-2-(2-Hydroxy-4-methoxy-5-methylsulfanyl-benzylamino)-propionic acid
CAS:115473-15-9
CAS:1173-88-2
CAS:95-44-3
CAS:18835-32-0