- +86-0533-2185556
- WhatsApp: +86 15965530500
- admin@hangyubiotech.com
Your Location:Home >Products >Inhibitor >1261576-81-1
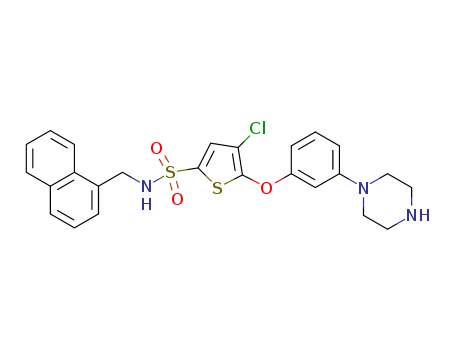

pd_meltingpoint:142-144 °C
Purity:99%
This invention relates generally to comp...
A compound, of formula (I): or a pharmac...
The synthesis of 4-chloro-N-(naphthalen-...
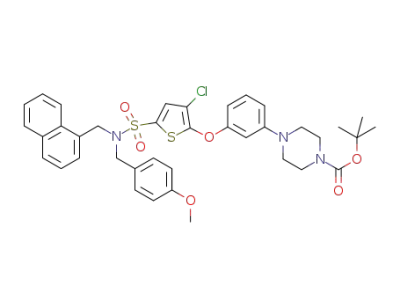
tert-butyl 4-(3-(3-chloro-5-(N-(4-methoxybenzyl)-N-(naphthalen-1-ylmethyl)sulfamoyl)thiophen-2-yloxy)phenyl)piperazine-1-carboxylate

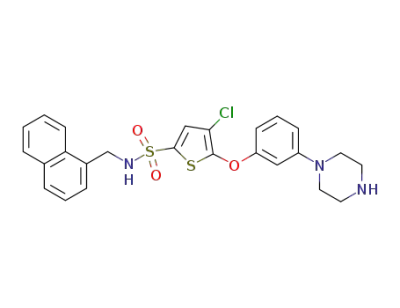
(4-chloro-N-(naphthalen-1-ylmethyl)-5-(3-(piperazin-1-yl) phenoxy)thiophene-2-sulfonamide)
| Conditions | Yield |
|---|---|
|
With
trifluoroacetic acid;
In
dichloromethane;
at 20 ℃;
for 2h;
Inert atmosphere;
|
97% |
|
With
trifluoroacetic acid;
In
dichloromethane;
at 20 ℃;
for 3h;
Inert atmosphere;
|
92% |
|
With
trifluoroacetic acid;
In
dichloromethane;
at 20 ℃;
for 3h;
Inert atmosphere;
|
92% |
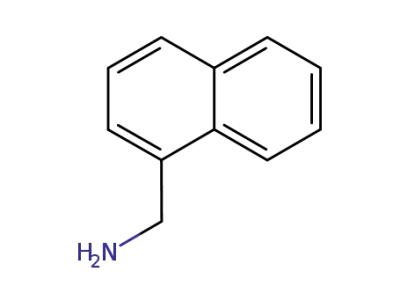
(naphth-1-yl)methylamine


(4-chloro-N-(naphthalen-1-ylmethyl)-5-(3-(piperazin-1-yl) phenoxy)thiophene-2-sulfonamide)
| Conditions | Yield |
|---|---|
|
Multi-step reaction with 4 steps
1.1: triethylamine / dichloromethane / 2 h / 20 °C / Inert atmosphere
2.1: sodium hydride / N,N-dimethyl-formamide / 0.25 h / 0 °C / Inert atmosphere
2.2: 2 h / 0 - 20 °C / Inert atmosphere
3.1: caesium carbonate / N,N-dimethyl-formamide / 2.5 h / 80 °C / Inert atmosphere
4.1: trifluoroacetic acid / dichloromethane / 3 h / 20 °C / Inert atmosphere
With
sodium hydride; caesium carbonate; triethylamine; trifluoroacetic acid;
In
dichloromethane; N,N-dimethyl-formamide;
|
|
|
Multi-step reaction with 4 steps
1.1: triethylamine / dichloromethane / 2 h / 20 °C / Inert atmosphere
2.1: sodium hydride / N,N-dimethyl-formamide / 0.25 h / 0 °C / Inert atmosphere
2.2: 2 h / 0 - 20 °C / Inert atmosphere
3.1: caesium carbonate / N,N-dimethyl-formamide / 2.5 h / 80 °C / Inert atmosphere
4.1: trifluoroacetic acid / dichloromethane / 3 h / 20 °C / Inert atmosphere
With
sodium hydride; caesium carbonate; triethylamine; trifluoroacetic acid;
In
dichloromethane; N,N-dimethyl-formamide;
|

tert-butyl 4-(3-(3-chloro-5-(N-(4-methoxybenzyl)-N-(naphthalen-1-ylmethyl)sulfamoyl)thiophen-2-yloxy)phenyl)piperazine-1-carboxylate

(naphth-1-yl)methylamine
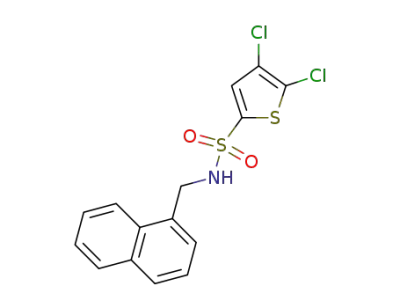
4,5-dichloro-N-(naphthalen-1-ylmethyl)thiophene-2-sulfonamide
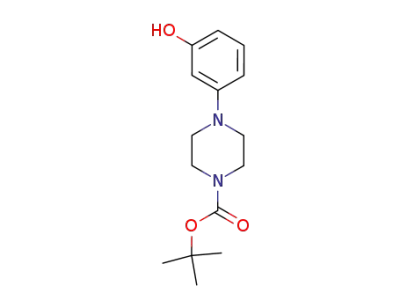
1,1-dimethylethyl 4-(3-hydroxyphenyl)-1-piperazinecarboxylate
CAS:85081-69-2
CAS:102397-69-3
CAS:152-58-9
CAS:54440-17-4