- +86-0533-2185556
- WhatsApp: +86 15965530500
- admin@hangyubiotech.com
Your Location:Home >Products >API >27519-02-4
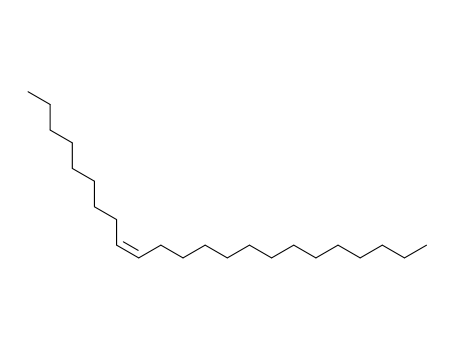

pd_meltingpoint:0°C
Appearance:clear colourless to slightly yellow liquid
Purity:99%
|
Safety Profile |
Moderately toxic by skin contact.When heated to decomposition it emits acrid smoke andirritating vapors. |
|
Metabolic pathway |
Muscalure is a long chain mono-unsaturated hydrocarbon and the 9,lO-double bond is a reactive site towards biologrcal and non-biological oxidative agents. |
|
Degradation |
Muscalure is stable to light and temperatures up to 50 °C for one year. |
|
General Description |
Muscalure is an insect pheromone utilized to attract houseflies. It is generally used as a coactive ingredient in fly-bait products. |
InChI:InChI=1/C23H46/c1-3-5-7-9-11-13-15-17-19-21-23-22-20-18-16-14-12-10-8-6-4-2/h17,19H,3-16,18,20-23H2,1-2H3/b19-17+
Long methylenic chain dialkylalkynes hav...
A new route to the synthesis of tricos-9...
Pure Z or E γ-iodoalkenes were prepared ...
The regioselection in the alkylation of ...
-
-
-
"Muscalure" (9-tricosene) was prepared v...
-
-
A cobalt-catalyzed decarboxylative methy...
The stereoselective hydrogenation of alk...
A practical and scalable synthesis of (Z...
-
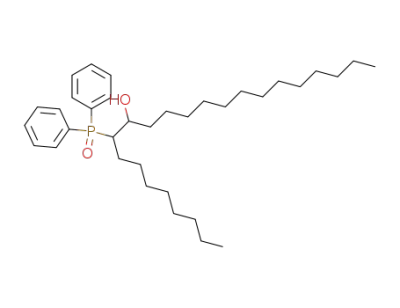
9-Diphenylphosphinoyltricosan-10-ol

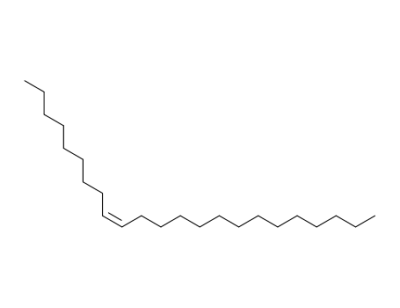
(Z)-tricos-9-ene


trans-9-tricosene
| Conditions | Yield |
|---|---|
|
With
potassium hydride;
In
N,N-dimethyl-formamide;
at 50 ℃;
for 0.5h;
Yield given. Yields of byproduct given. Title compound not separated from byproducts;
|
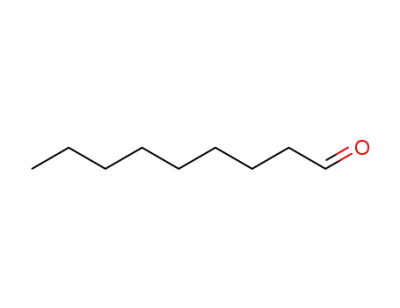
nonan-1-al


(n-tetradecyl)triphenylphosphonium bromide


(Z)-tricos-9-ene


trans-9-tricosene
| Conditions | Yield |
|---|---|
|
(n-tetradecyl)triphenylphosphonium bromide;
With
n-butyllithium;
In
tetrahydrofuran; dimethyl sulfoxide;
at 10 - 15 ℃;
for 0.0833333h;
nonan-1-al;
In
tetrahydrofuran; dimethyl sulfoxide;
at 20 ℃;
for 0.5h;
|
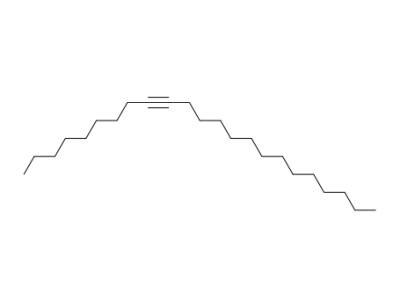
tricos-9-yne
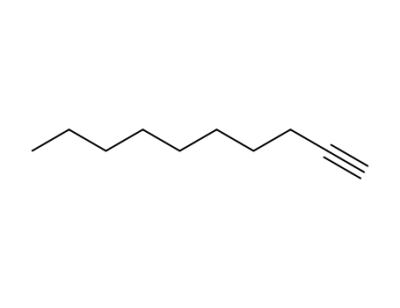
1-decyne
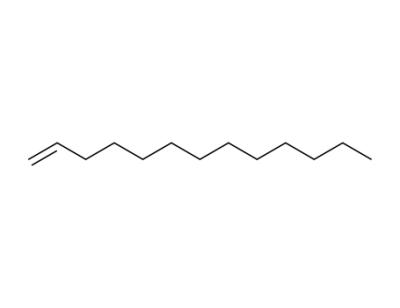
1-tridecene

nonan-1-al

nonan-1-al

myristylaldehyde

trans-9-tricosene
CAS:112163-33-4
CAS:112-34-5
CAS:27113-22-0
CAS:13870-90-1