- +86-0533-2185556
- WhatsApp: +86 15965530500
- admin@hangyubiotech.com
Your Location:Home >Products >Pharmaceutical intermediates >149376-69-2
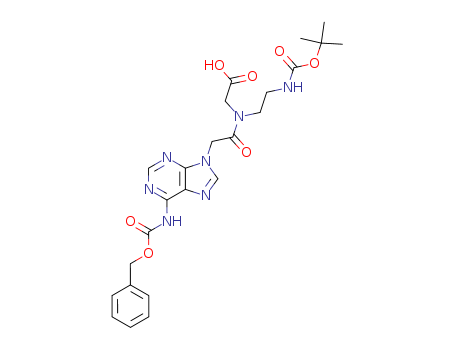

Purity:99%
The preparation of mixed-sequence PNAs (...
Peptide nucleic acid (PNA) monomer build...
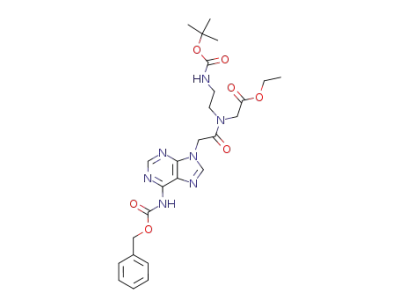
ethyl N-((N6-(benzyloxycarbonyl)adenin-9-yl)acetyl)-N-(2-Boc-aminoethyl)glycinate

N-((N6-(benzyloxycarbonyl)adenin-9-yl)acetyl)-N-(2-Boc-aminoethyl)glycine
| Conditions | Yield |
|---|---|
|
With
lithium hydroxide;
In
tetrahydrofuran;
at 0 ℃;
for 0.25h;
|
55% |
|
With
sodium hydroxide; water;
In
methanol;
|
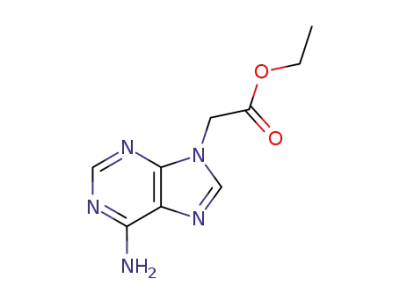
ethyl 2-(9H-adenin-9-yl)acetate

N-((N6-(benzyloxycarbonyl)adenin-9-yl)acetyl)-N-(2-Boc-aminoethyl)glycine
| Conditions | Yield |
|---|---|
|
Multi-step reaction with 4 steps
1: 63 percent / dimethylformamide; CH2Cl2 / 24 h
2: aq. NaOH / methanol / 0.5 h / 0 °C
3: 3,4-dihydro-3-hydroxy-4-oxo-1,2,3-benzotriazine, DCC / CH2Cl2; dimethylformamide / 1.) 0 deg C, 2.5 h, 2.) r.t., 1.5 h
4: 55 percent / 1 M aq. LiOH / tetrahydrofuran / 0.25 h / 0 °C
With
lithium hydroxide; sodium hydroxide; 3-hydroxy-3,4-dihydrobenzotriazine-4-one; dicyclohexyl-carbodiimide;
In
tetrahydrofuran; methanol; dichloromethane; N,N-dimethyl-formamide;
|
|
|
Multi-step reaction with 4 steps
1: dimethylformamide; CH2Cl2
2: LiOH, H2O / tetrahydrofuran
3: dicyclohexylcarbodiimide (DCC), DhbtOH / dimethylformamide; CH2Cl2
4: NaOH, H2O / methanol
With
lithium hydroxide; sodium hydroxide; water; dicyclohexyl-carbodiimide;
In
tetrahydrofuran; methanol; dichloromethane; N,N-dimethyl-formamide;
|

ethyl N-((N6-(benzyloxycarbonyl)adenin-9-yl)acetyl)-N-(2-Boc-aminoethyl)glycinate

ethyl 2-(9H-adenin-9-yl)acetate
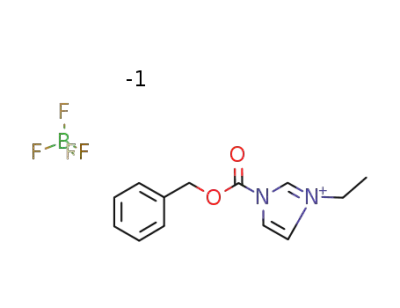
1-(benzyloxycarbonyl)-3-ethylimidazolium tetrafluoroborate
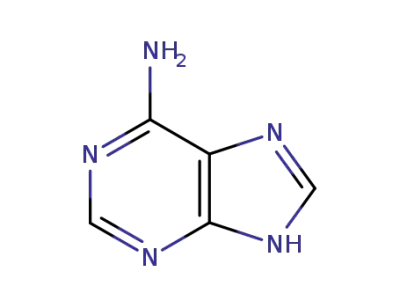
adenine
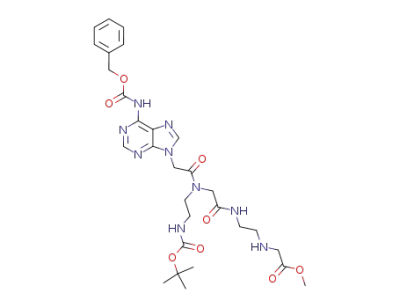
(2-{2-[[(6-benzyloxycarbonylamino-purin-9-yl)-acetyl]-(2-tert-butoxycarbonylamino-ethyl)-amino]-acetylamino}-ethylamino)-acetic acid methyl ester
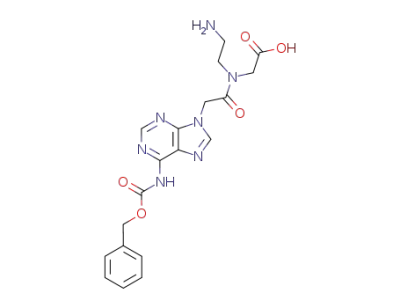
{(2-Amino-ethyl)-[2-(6-benzyloxycarbonylamino-purin-9-yl)-acetyl]-amino}-acetic acid
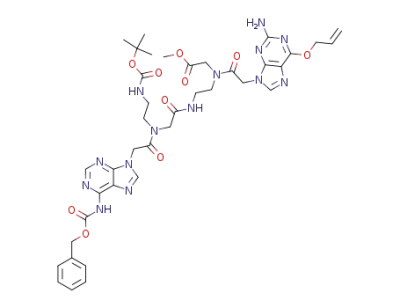
[[(6-allyloxy-2-amino-purin-9-yl)-acetyl]-(2-{2-[[(6-benzyloxycarbonylamino-purin-9-yl)-acetyl]-(2-tert-butoxycarbonylamino-ethyl)-amino]-acetylamino}-ethyl)-amino]-acetic acid methyl ester
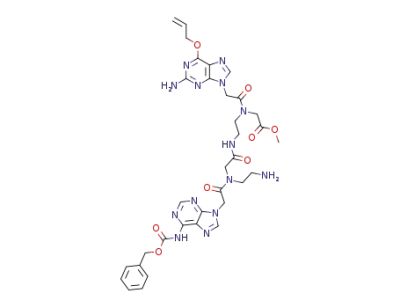
{[2-(6-Allyloxy-2-amino-purin-9-yl)-acetyl]-[2-(2-{(2-amino-ethyl)-[2-(6-benzyloxycarbonylamino-purin-9-yl)-acetyl]-amino}-acetylamino)-ethyl]-amino}-acetic acid methyl ester
CAS:112163-33-4
CAS:112-84-5
CAS:1220022-06-9
CAS:1260393-98-3