- +86-0533-2185556
- WhatsApp: +86 15965530500
- admin@hangyubiotech.com

pd_meltingpoint:212-214°C
Purity:99%
|
Purification Methods |
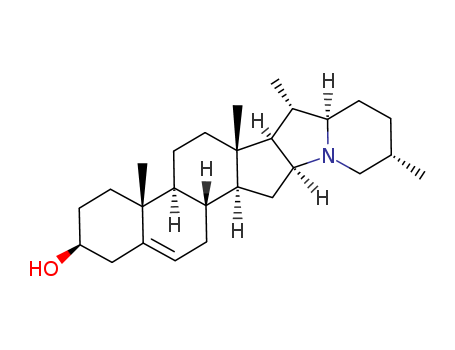
Solanidine crystallises from CHCl3/MeOH, aqueous EtOH or aqueous MeOH as needles. TLC on Al2O3 plates using CH2Cl2/MeOH (98:2) gives a spot at RF 0.47. The hydrochloride crystallises from aqueous EtOH with m 345o(dec). The acetate crystallises from EtOH with m 208o. [Schreiber & Roensch Tetrahedron 21 6451965, Kessar et al. Tetrahedron 27 2153 1971, Reichestein & Reich Ann Rev Biochem 15 155 1946, Beilstein 21 III/IV 1398, 27 III/IV 2000.] |
InChI:InChI=1/C27H43NO/c1-16-5-8-23-17(2)25-24(28(23)15-16)14-22-20-7-6-18-13-19(29)9-11-26(18,3)21(20)10-12-27(22,25)4/h6,16-17,19-25,29H,5,7-15H2,1-4H3
-
A divergent synthesis of solanidine and ...
A concise asymmetric synthesis of two na...
Demissidine and solanidine, two steroida...
Potato tubers accumulate varying amounts...
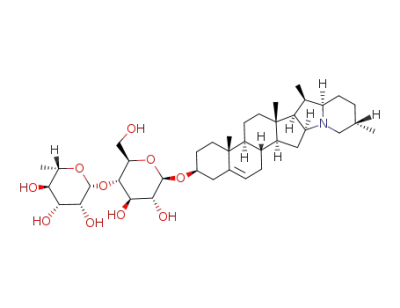
β-Chaconine

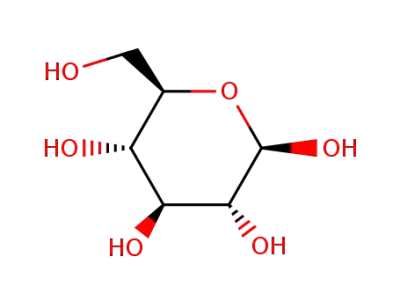
β-D-glucose

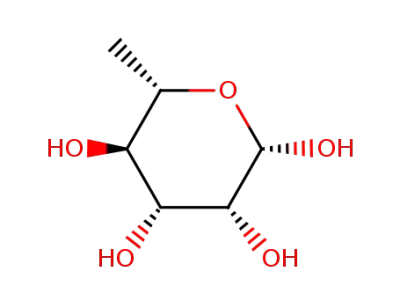
β-L-rhamnose

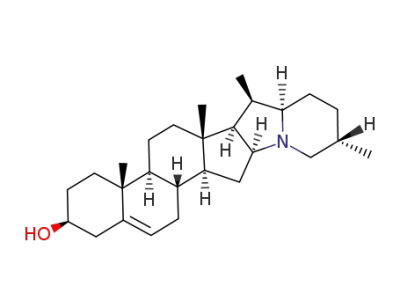
Solanidine
| Conditions | Yield |
|---|---|
|
With
hydrogenchloride;
In
ethanol;
at 100 ℃;
for 3h;
Product distribution;
|
|
|
With
hydrogenchloride;
In
ethanol;
at 100 ℃;
for 3h;
|
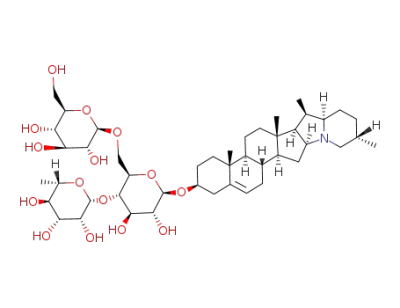
Stenanthine


β-D-glucose


β-L-rhamnose


Solanidine
| Conditions | Yield |
|---|---|
|
With
hydrogenchloride;
In
ethanol;
at 100 ℃;
Product distribution;
|
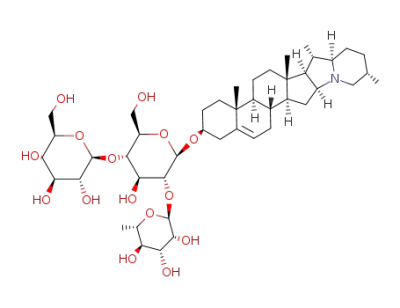
solanidine 3-O-α-L-rhamnopyranosyl-(1<*>2)-O-<β-D-glucopyranosyl-(1<*>4)>-β-D-glucopyranoside
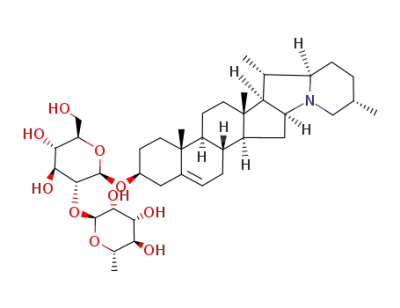
solanidine 3-O-α-L-rhamnopyranosyl-(1->2)-β-D-glucopyranoside
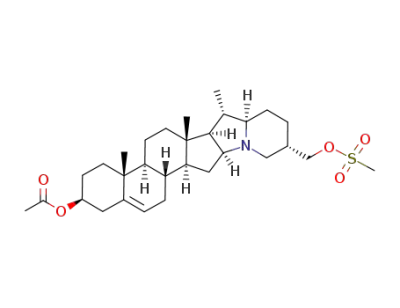
C30H47NO5S
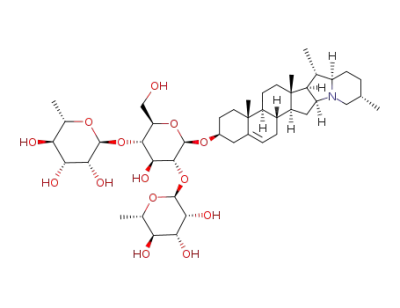
α-chaconine
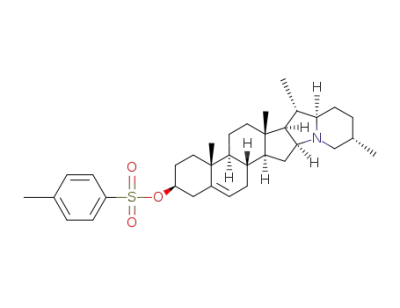
3β(p-toluenesulfonyloxy)-22αH,25βH-solanid-5-en-3-ol
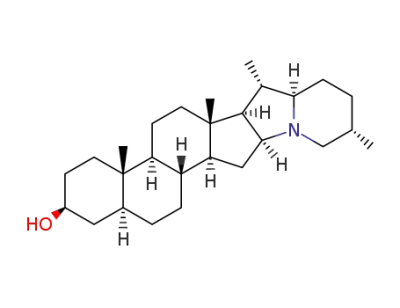
demissidine
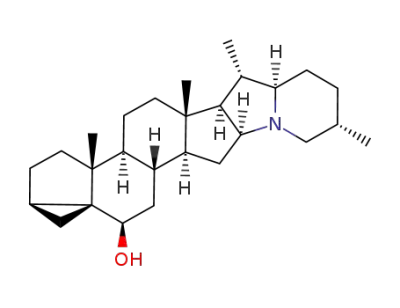
3α,5-cyclo-5α,22αH,25βH-solanidan-6β-ol
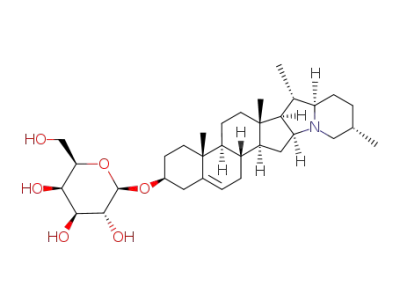
solanidine β-D-galactopyranoside
CAS:112163-33-4
CAS:112-84-5
CAS:75345-27-6
CAS:1189541-98-7