- +86-0533-2185556
- WhatsApp: +86 15965530500
- admin@hangyubiotech.com
Your Location:Home >Products >Supplement >110958-19-5
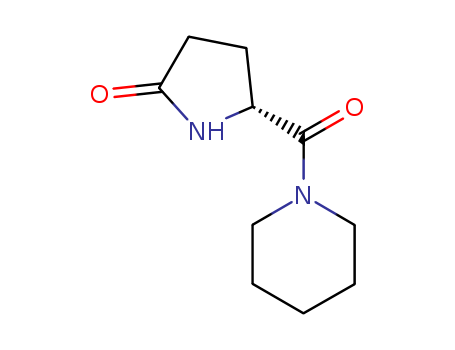

Purity:99%
|
General Description |
Fasoracetam is a synthetic compound that belongs to the racetam family of nootropics, which are known for their cognitive enhancing properties. It is often used as a study aid and to improve memory and learning. Fasoracetam works by modulating the activity of neurotransmitters in the brain, specifically by targeting the glutamate and GABA systems. It is believed to have potential for treating conditions such as ADHD and anxiety, and has shown promise in animal studies for its potential neuroprotective and antidepressant effects. While more research is needed to fully understand its mechanisms and potential therapeutic uses, fasoracetam is considered to be a safe and well-tolerated compound when used at appropriate doses. |
InChI:InChI=1/C10H16N2O2/c13-9-5-4-8(11-9)10(14)12-6-2-1-3-7-12/h8H,1-7H2,(H,11,13)/t8-/m1/s1
The disclosure is directed to cocrystals...
Amidation of unprotected amino acids has...
Efficient syntheses of chiral vicinal di...
Pyroglutamide derivatives have been foun...
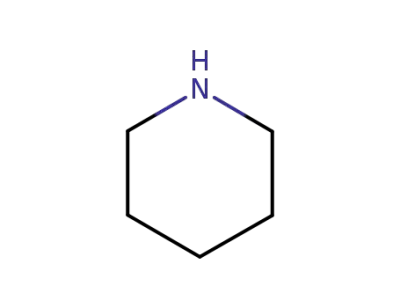
piperidine

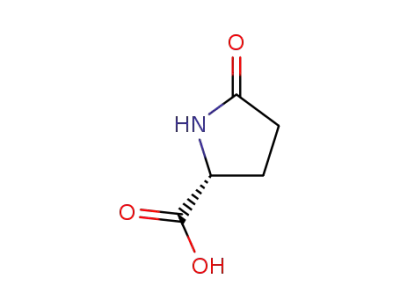
D-pyrrolidone-5-carboxylic acid

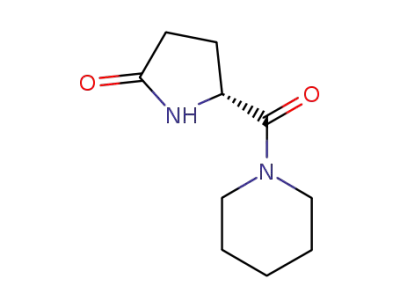
Fasoracetam
| Conditions | Yield |
|---|---|
|
In
acetonitrile;
|
|
|
In
ethyl acetate;
|

D-pyrrolidone-5-carboxylic acid


Fasoracetam
| Conditions | Yield |
|---|---|
|
Multi-step reaction with 2 steps
1: PTSA / toluene / 16 h / Heating
2: acetonitrile / 0.17 h / 20 °C
With
toluene-4-sulfonic acid;
In
toluene; acetonitrile;
|

piperidine

D-pyrrolidone-5-carboxylic acid
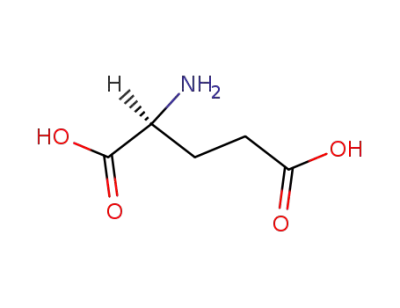
L-glutamic acid
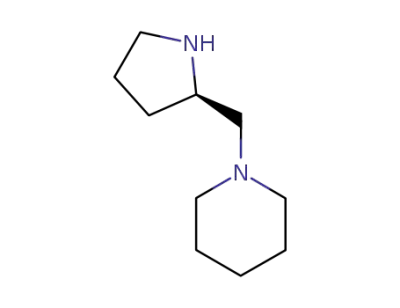
(R)-2-[(piperidin-1-yl)methyl]pyrrolidine
CAS:112163-33-4
CAS:112-84-5
CAS:17696-69-4
CAS:116-31-4