- +86-0533-2185556
- WhatsApp: +86 15965530500
- admin@hangyubiotech.com
Your Location:Home >Products >API >24880-43-1
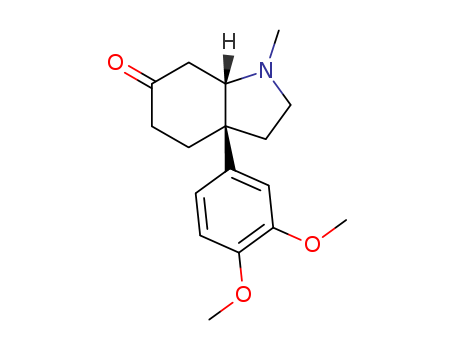

Purity:99%
|
Synthesis Reference(s) |
Tetrahedron, 24, p. 6583, 1968 DOI: 10.1016/S0040-4020(01)96872-3 |
InChI:InChI=1/C17H23NO3/c1-18-9-8-17(7-6-13(19)11-16(17)18)12-4-5-14(20-2)15(10-12)21-3/h4-5,10,16H,6-9,11H2,1-3H3/t16-,17-/m0/s1
We demonstrate that α-aryl cyclic vinylo...
A concise total synthesis of mesembrine ...
The direct oxidation of unprotected amin...
Joubertinamine and mesembrine are two re...
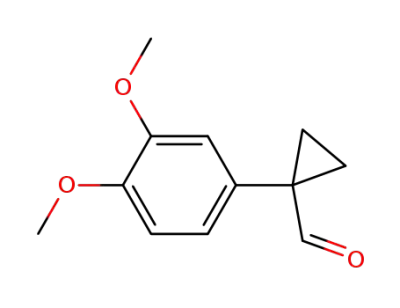
1-(3,4-dimethoxyphenyl)cyclopropanecarbaldehyde

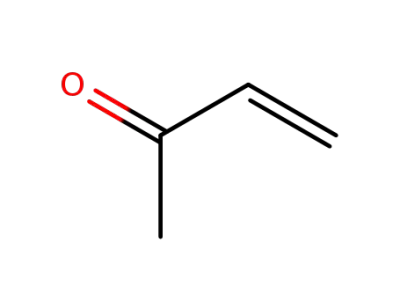
methyl vinyl ketone

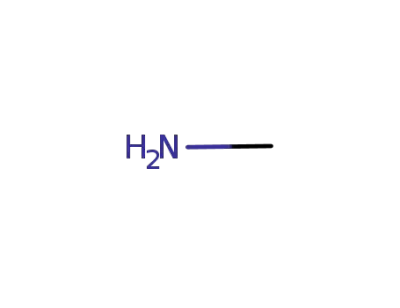
methylamine

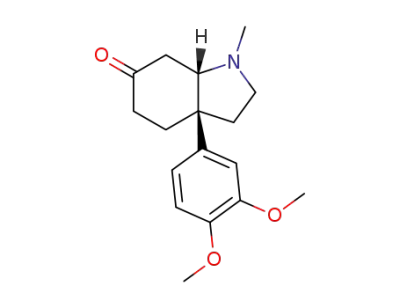
(-)-mesembrine
| Conditions | Yield |
|---|---|
|
1-(3,4-dimethoxyphenyl)cyclopropanecarbaldehyde; methylamine;
With
sodium sulfate;
In
1,2-dichloro-ethane;
at 20 ℃;
for 0.166667h;
With
chloro-trimethyl-silane; sodium iodide;
In
DMF (N,N-dimethyl-formamide);
at 90 ℃;
for 3h;
methyl vinyl ketone;
With
hydrogenchloride;
more than 3 stages;
|
40% |
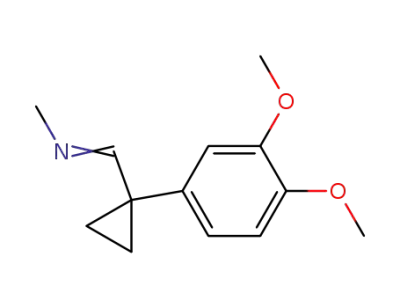
1-(3,4-Dimethoxy-phenyl)-1-methyl-iminomethyl-cyclopropan


methyl vinyl ketone

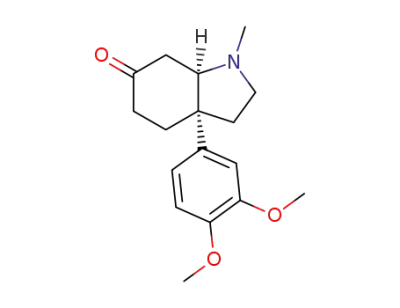
(3aR,7aR)-3a-(3,4-Dimethoxyphenyl)-1-methylhexahydro-1H-indol-6(2H)-one


(-)-mesembrine
| Conditions | Yield |
|---|---|
|
1-(3,4-Dimethoxy-phenyl)-1-methyl-iminomethyl-cyclopropan;
With
chloro-trimethyl-silane; sodium iodide;
In
DMF (N,N-dimethyl-formamide);
at 90 ℃;
for 3h;
With
hydrogenchloride;
In
diethyl ether; dichloromethane;
methyl vinyl ketone;
With
sodium hydroxide;
more than 3 stages;
|
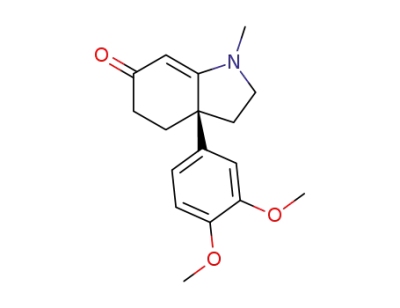
Δ7-mesembrenone
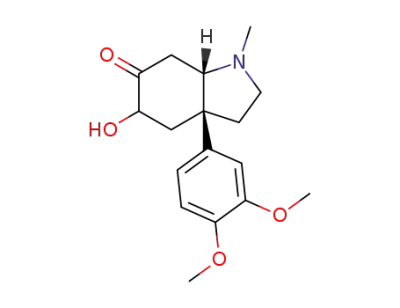
(3aR,7aS)-3a-(3,4-Dimethoxy-phenyl)-5-hydroxy-1-methyl-octahydro-indol-6-one
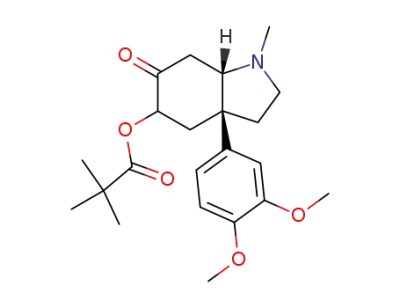
2,2-Dimethyl-propionic acid (3aR,7aS)-3a-(3,4-dimethoxy-phenyl)-1-methyl-6-oxo-octahydro-indol-5-yl ester
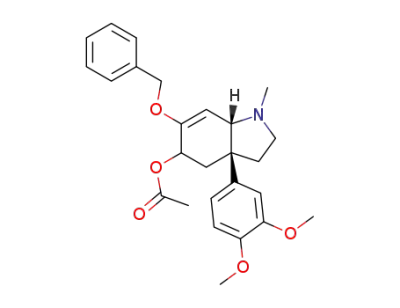
Acetic acid (3aR,7aS)-6-benzyloxy-3a-(3,4-dimethoxy-phenyl)-1-methyl-2,3,3a,4,5,7a-hexahydro-1H-indol-5-yl ester
CAS:112163-33-4
CAS:112-84-5
CAS:335-10-4
CAS:2521-07-5