- +86-0533-2185556
- WhatsApp: +86 15965530500
- admin@hangyubiotech.com
Your Location:Home >Products >API >22112-77-2
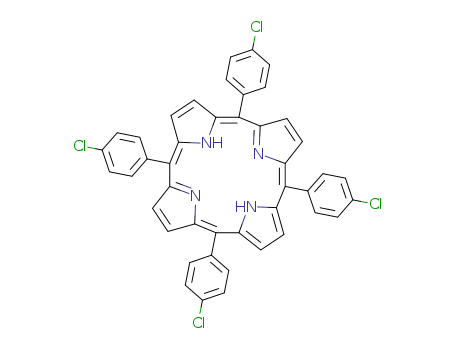

pd_meltingpoint:>27 °C
Purity:99%
|
General Description |
Meso-tetra(4-chlorophenyl)porphine is a chemical compound that belongs to the group of porphyrins, which are heterocyclic macrocycles containing four pyrrole rings. meso-Tetra (4-chlorophenyl) porphine is a type of porphyrin that contains four 4-chlorophenyl groups attached to the pyrrole rings. It has been studied for its potential applications in photodynamic therapy, a treatment that uses light-sensitive compounds to selectively destroy cancer cells. Meso-tetra(4-chlorophenyl)porphine has also been investigated for its use in chemical sensors, catalysis, and as a model system for studying the properties of natural porphyrin compounds. Furthermore, it has been utilized in the synthesis of other porphyrin derivatives for various research and industrial purposes. |
InChI:InChI=1/C44H26Cl4N4/c45-29-9-1-25(2-10-29)41-33-17-19-35(49-33)42(26-3-11-30(46)12-4-26)37-21-23-39(51-37)44(28-7-15-32(48)16-8-28)40-24-22-38(52-40)43(36-20-18-34(41)50-36)27-5-13-31(47)14-6-27/h1-24,49,52H/b41-33-,41-34-,42-35-,42-37-,43-36-,43-38-,44-39-,44-40-
In this work we report the synthesis of ...
The current study determines optical and...
The one-step synthesis of well-defined C...
Tin tetrakis(4-fluorophenyl)porphyrin, t...
5, 10, 15, 20-tetrakis-(4-chlorophenyl) ...
Main properties of 5,10,15,20-tetra(4-ch...
By combining 5,10,15,20-tetra(4-chlorine...
The direct sources of aliphatic acids in...
Two new cobaltous-porphyrin complexes, n...
The synthesis of dipyrromethanes from py...
pyrrole

4-chlorobenzaldehyde

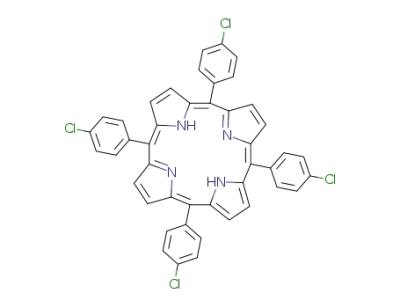
5,10,15,20-tetra-(4-chlorophenyl)porphyrin
| Conditions | Yield |
|---|---|
|
With
vanadium(V) oxychloride;
In
dichloromethane;
for 1h;
Ambient temperature;
other reagents: TiCl4, VO(OEt)Cl2, VO(OiPr)Cl2, Mn(OAc)3;
|
65% |
|
With
propionic acid;
Reflux;
|
65% |
|
pyrrole; 4-chlorobenzaldehyde;
With
boron trifluoride diethyl etherate;
In
chloroform;
at 61 ℃;
for 1h;
In
chloroform;
at 61 ℃;
for 1h;
|
64% |
|
With
air; nitrobenzene; valeric acid;
at 141 ℃;
for 1h;
|
56% |
|
With
propionic acid;
for 0.5h;
Reflux;
|
56% |
|
pyrrole; 4-chlorobenzaldehyde;
In
propionic acid; toluene;
at 152.4 ℃;
for 0.15h;
under 2280.15 Torr;
Large scale;
Green chemistry;
With
oxygen;
In
propionic acid; toluene;
for 0.3h;
under 1520.1 Torr;
Large scale;
Green chemistry;
|
52.2% |
|
pyrrole; 4-chlorobenzaldehyde;
With
propionic acid;
In
toluene;
at 152.4 ℃;
for 0.15h;
under 2280.15 Torr;
With
oxygen;
In
toluene;
for 0.3h;
Reflux;
|
51.8% |
|
With
1-methyl-3-nitrobenzene; propionic acid; valeric acid;
for 2h;
Reflux;
|
50.3% |
|
With
propionic acid;
for 0.5h;
Reflux;
|
41% |
|
With
hydrogenchloride;
In
N,N-dimethyl-formamide;
|
38% |
|
With
air; chloroacetic acid;
In
xylene;
for 1.5h;
Heating;
|
34.6% |
|
pyrrole; 4-chlorobenzaldehyde;
With
boron trifluoride diethyl etherate;
In
dichloromethane;
at 20 ℃;
for 0.5h;
With
selenium(IV) oxide;
In
dichloromethane;
at 20 ℃;
for 1h;
|
34% |
|
4-chlorobenzaldehyde;
With
benzoic acid;
at 200 ℃;
for 0.0833333h;
under 2736.18 Torr;
pyrrole;
at 200 ℃;
for 0.333333h;
|
25% |
|
pyrrole; 4-chlorobenzaldehyde;
With
boron trifluoride diethyl etherate;
In
dichloromethane;
at 20 ℃;
for 0.5h;
With
selenium(IV) oxide;
In
dichloromethane;
for 1h;
|
24% |
|
pyrrole; 4-chlorobenzaldehyde;
With
Tulsion-63 resin; Triethyl orthoacetate;
In
dichloromethane;
at 20 ℃;
for 17h;
With
chloranil;
In
dichloromethane;
for 2h;
Heating;
|
23% |
|
With
propionic acid;
at 165 ℃;
for 1h;
Inert atmosphere;
Schlenk technique;
Reflux;
|
23.1% |
|
With
acetic acid;
Reflux;
|
21% |
|
With
propionic acid;
for 2h;
Reflux;
|
20% |
|
pyrrole; 4-chlorobenzaldehyde;
With
trifluoroacetic acid;
In
dichloromethane;
at 25 ℃;
for 1h;
Inert atmosphere;
With
2,3-dicyano-5,6-dichloro-p-benzoquinone;
In
dichloromethane;
for 1h;
|
20.8% |
|
With
propionic acid;
for 0.5h;
Reflux;
|
20% |
|
at 200 ℃;
for 0.25h;
|
10% |
|
With
air; propionic acid;
for 0.0666667h;
microwave irradiation;
|
7% |
|
With
propionic acid;
In
nitrobenzene;
at 120 ℃;
for 1h;
|
|
|
In
propionic acid; nitrobenzene;
at 120 ℃;
for 1h;
|
|
|
With
propionic acid;
for 30h;
Heating;
|
|
|
|
|
|
With
chloranil; trifluoroacetic acid;
Yield given. Multistep reaction;
1) CH2Cl2, 2 h, r.t., 2) 1 h, reflux;
|
|
|
With
chloranil;
aluminum oxide; FSM-16(2.8 nm);
Yield given. Multistep reaction;
1) CH2Cl2, r.t., 1 h, 2) CH2Cl2, 45 deg C, 1 h;
|
|
|
In
propionic acid;
Heating;
|
|
|
|
|
|
With
trifluoroacetic acid;
In
dichloromethane; water;
for 2h;
|
|
|
With
propionic acid;
for 2h;
Reflux;
|
|
|
pyrrole; 4-chlorobenzaldehyde;
With
boron trifluoride diethyl etherate;
In
chloroform;
at 20 ℃;
for 1h;
Inert atmosphere;
With
2,3-dicyano-5,6-dichloro-p-benzoquinone;
|
|
|
pyrrole; 4-chlorobenzaldehyde;
With
trifluoroacetic acid;
In
dichloromethane;
for 1h;
Darkness;
Inert atmosphere;
With
2,3-dicyano-5,6-dichloro-p-benzoquinone;
In
dichloromethane;
for 1h;
Inert atmosphere;
|
|
|
In
propionic acid;
for 0.666667h;
Reflux;
|
|
|
With
propionic acid;
for 0.5h;
Reflux;
|
|
|
With
propionic acid;
for 0.5h;
Reflux;
|
|
|
With
propionic acid;
for 1h;
Reflux;
|
|
|
pyrrole; 4-chlorobenzaldehyde;
With
trifluoroacetic acid;
In
dichloromethane;
for 1h;
Inert atmosphere;
Schlenk technique;
Darkness;
With
2,3-dicyano-5,6-dichloro-p-benzoquinone;
In
dichloromethane;
for 1h;
Inert atmosphere;
Schlenk technique;
Darkness;
|
|
|
With
propionic acid;
Reflux;
|
|
|
In
propionic acid;
at 145 ℃;
for 2h;
Inert atmosphere;
Darkness;
|
|
|
With
propionic acid;
at 145 ℃;
for 2h;
Inert atmosphere;
Darkness;
|
|
|
With
propionic acid;
for 0.75h;
Reflux;
|
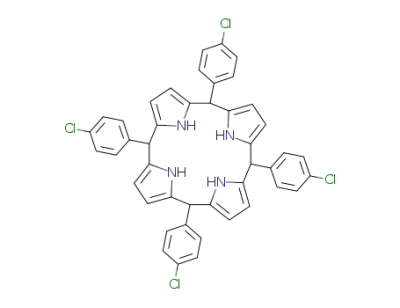
5,10,15,20-tetrakis(4-chlorophenyl)porphyrinogen


5,10,15,20-tetra-(4-chlorophenyl)porphyrin
| Conditions | Yield |
|---|---|
|
With
air;
In
dichloromethane;
at 39 ℃;
for 4h;
|
|
|
With
air;
In
dichloromethane;
at 39 ℃;
|
|
|
With
air;
In
dichloromethane;
at 39 ℃;
for 3h;
|
|
|
for 2h;
UV-irradiation;
|
|
|
With
2,3-dicyano-5,6-dichloro-p-benzoquinone;
In
dichloromethane;
for 2h;
Inert atmosphere;
Darkness;
|
117 mg |
|
With
1-hydroxy-3H-benz[d][1,2]iodoxole-1,3-dione;
In
dichloromethane;
at 39 ℃;
|
|
|
With
ammonium cerium (IV) nitrate;
In
dichloromethane;
at 20 ℃;
for 0.333333h;
Green chemistry;
|
|
|
With
2,3-dicyano-5,6-dichloro-p-benzoquinone;
In
dichloromethane;
for 0.0833333h;
Inert atmosphere;
|

pyrrole

4-chlorobenzaldehyde
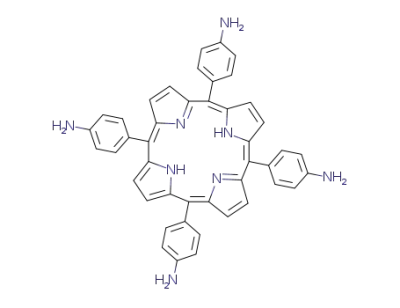
tetrakis(4-aminophenyl)porphyrin
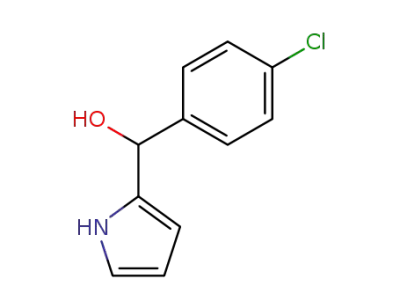
(4-Chloro-phenyl)-(1H-pyrrol-2-yl)-methanol
CAS:115473-15-9
CAS:118685-33-9
CAS:102029-73-2
CAS:1401242-74-7