- +86-0533-2185556
- WhatsApp: +86 15965530500
- admin@hangyubiotech.com

pd_meltingpoint:35339ºC
Appearance:Off-white to pale yellow solid
Purity:99%
|
Manufacturing Process |
A mixture of 155 parts of 2-chloro-10-(γ-chloropropyl)phenothiazine, 76 partsof sodium iodide, 216 parts of piperazine and 2,000 parts of butanone is refluxed for 8 hours, concentrated and extracted with dilute hydrochloric acid. The extract is rendered alkaline by addition of dilute potassium carbonate and benzene or chloroform extracted. This extract is washed with water, dried over anhydrous potassium carbonate, filtered and evaporated. Vacuum distillation at 0.1 mm pressure yields 2-chloro-10-[γ-(N-piperazino)propyl]phenothiazine at about 214°-218°C.A stirred mixture of 5 parts of 2-chloro-10-[γ-(Npiperazino)propyl]phenothiazine, 1.92 parts of 2-bromoethanol, 2.11 parts of potassium carbonate and 35 parts of toluene is refluxed for 5 hours. The mixture is treated with water and benzene and the organic layer is separated, washed with water, dried over anhydrous potassium carbonate, filtered and evaporated. The residue is distilled at about 240°-244°C and 0.15 mm pressure to yield 2-chloro-10-[γ-(N'-β-hydroxyethyl-N-piperazino)propyl]phenothiazine according to US Patent 2,838,507.The 2-chloro-10-(γ-chloropropyl)phenothiazine starting material is produced from 2-chlorophenothiazine and 1-bromo-3-chloropropane. |
|
Therapeutic Function |
Tranquilizer |
|
Biochem/physiol Actions |
D2 dopamine receptor antagonist; α-adrenergic receptor antagonist and σ-receptor agonist; phenothiazine antipsychotic. Inhibits glutamate dehydrogenase in vitro. |
|
Safety Profile |
Poison by ingestion, intravenous, subcutaneous, intraperitoneal, and intramuscular routes. Human systemic effects by intramuscular route: muscle spasms. Experimental teratogenic and reproductive effects. Human mutation data reported. When heated to decomposition it emits very toxic fumes of SOx, NOx, and cl-. |
|
Definition |
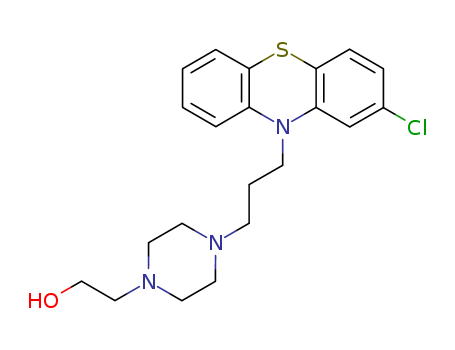
ChEBI: A phenothiazine derivative in which the phenothiazine tricycle carries a chloro substituent at the 2-position and a 3-[4-(2-hydroxyethyl)piperazin-1-yl]propyl group at N-10. |
|
Brand name |
Trilafon (Schering). |
|
General Description |
Perphenazine, 4-[3-(2-chlorophenothiazine-10-yl)propyl]piperazineethanol; 2-chloro-10-[3-[4-(2-hydroxyethyl)piperazinyl]propyl]phenothiazine(Trilafon), is an effective antipsychotic and antiemetic. |
InChI:InChI=1/C21H26ClN3OS/c22-17-6-7-21-19(16-17)25(18-4-1-2-5-20(18)27-21)9-3-8-23-10-12-24(13-11-23)14-15-26/h1-2,4-7,16,26H,3,8-15H2
Novel chemical conjugates of a psychotro...
-
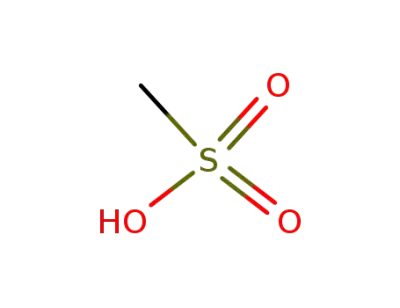
methanesulfonic acid

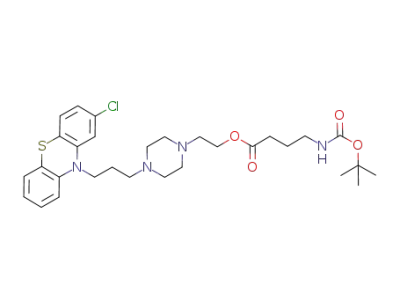
perphenazine N-Boc-4-aminobutyrate

![4-[3-(2-chloro-10H-phenothiazin-10-yl)propyl]-1-piperazineethyl 4-aminobutyryl ester trimesylate](/upload/2026/5/1877428d-35d1-4dd4-b68d-876378f3cf9d.png)
4-[3-(2-chloro-10H-phenothiazin-10-yl)propyl]-1-piperazineethyl 4-aminobutyryl ester trimesylate

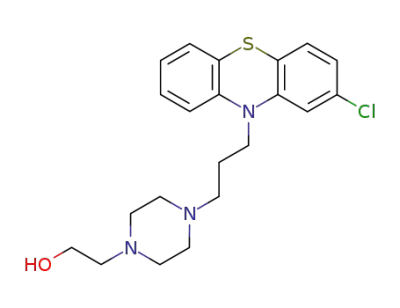
perphenazine
| Conditions | Yield |
|---|---|
|
In
acetonitrile;
at 0 - 40 ℃;
for 6h;
Product distribution / selectivity;
|
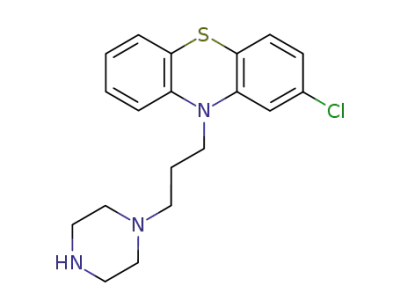
N-Demethyl prochlorperazine

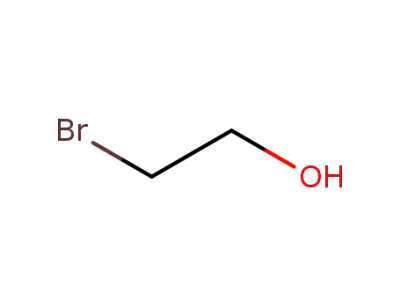
2-bromoethanol


perphenazine
| Conditions | Yield |
|---|---|
|
With
potassium carbonate; toluene;
|
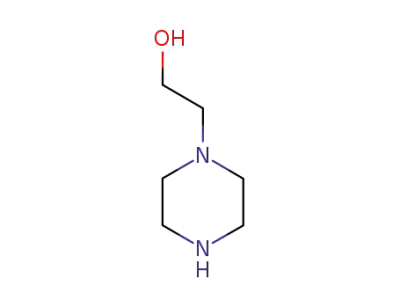
1-(2-hydroxyethyl)piperazine
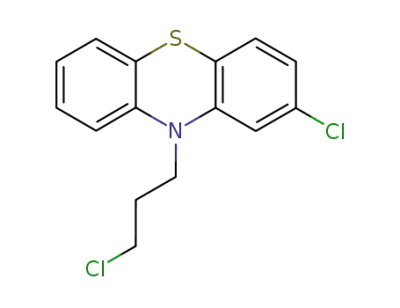
2-chloro-10-(3-chloropropyl)-10H-phenothiazine

N-Demethyl prochlorperazine

2-bromoethanol
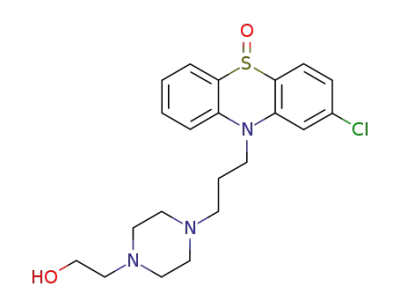
perphenazine sulfoxide
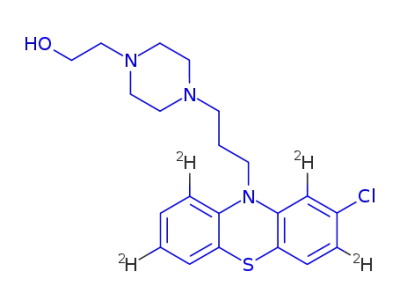
perphenazine-d4
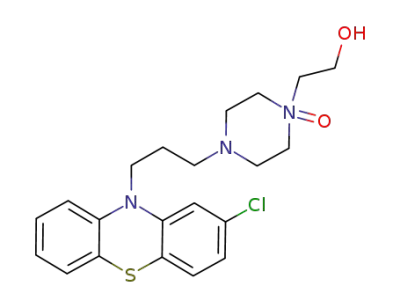
perphenazine 17-N-oxide
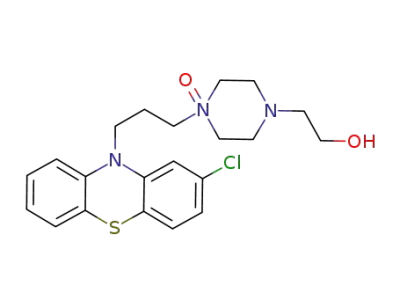
perphenazine 14-N-oxide
CAS:115473-15-9
CAS:118685-33-9
CAS:1700-10-3
CAS:1018448-65-1