- +86-0533-2185556
- WhatsApp: +86 15965530500
- admin@hangyubiotech.com
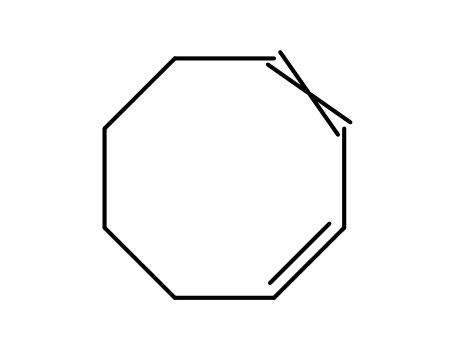

pd_meltingpoint:-53 – -51 °C
Appearance:colourless liquid
Purity:99%
|
General Description |
1,3-Cyclooctadiene, particularly its (Z,Z)-isomer, can undergo enantiodifferentiating photoisomerization to produce chiral (E,Z)-1,3-cyclooctadiene with significant enantiomeric excess (up to 18% ee) when sensitized by chiral aromatic carboxylates, such as (-)-menthyl benzenehexacarboxylate, in nonpolar solvents. The process is highly sensitive to solvent polarity, with polar solvents diminishing enantioselectivity, likely due to radical ionic intermediates. The resulting optically active (E,Z)-isomer exhibits exceptionally high specific rotation and circular dichroism, confirming theoretical predictions about its chiroptical properties. Supramolecular systems, such as γ-cyclodextrin-based rotaxanes, further enhance enantioselectivity (up to 15.3% ee) by providing chiral binding sites for sensitization. |
InChI:InChI=1/C8H12/c1-2-4-6-8-7-5-3-1/h1-4H,5-8H2
A biphenyl photosensitizer axle was impl...
Enantiodifferentiating photoisomerizatio...
-
6-O-(2-Naphthoyl)curdlan was newly synth...
tetrahydrofuran

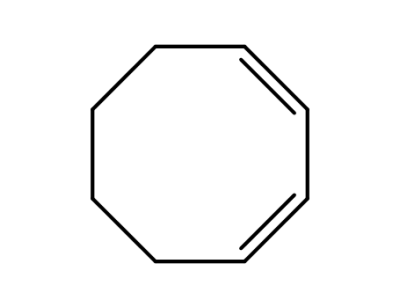
1,3-cyclooctadiene

![bicyclo[3.3.0]oct-2-ene](/upload/2026/5/e1d6899f-43b9-49a4-9df7-ac4103d18ef3.png)
bicyclo[3.3.0]oct-2-ene

cyclooctadienyl anion

4-Ethylbicyclo<3.3.0>oct-2-en
| Conditions | Yield |
|---|---|
|
With
[(trimethylsilyl)methyl]potassium;
at 0 ℃;
Mechanism;
Product distribution;
|
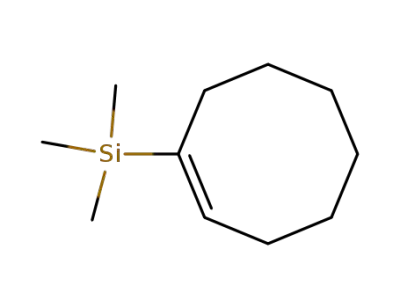
1-trimethylsilyl-cis-cyclo-octene

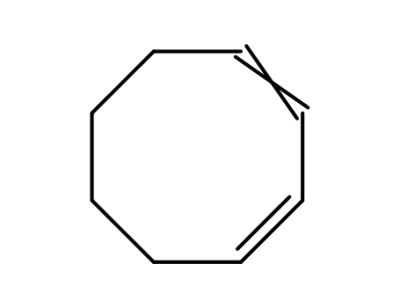
1,3-cyclooctadiene
| Conditions | Yield |
|---|---|
|
Multi-step reaction with 2 steps
1: 84.7 percent / m-chloroperbenzoic acid, disodium hydrogen phosphate / CH2Cl2 / 1 h
2: conc. H2SO4, MeOH / 0.33 h / 80 °C / 3 and 4 not separated
With
methanol; disodium hydrogenphosphate; sulfuric acid; 3-chloro-benzenecarboperoxoic acid;
In
dichloromethane;
|
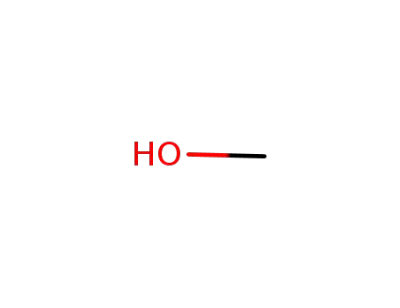
methanol
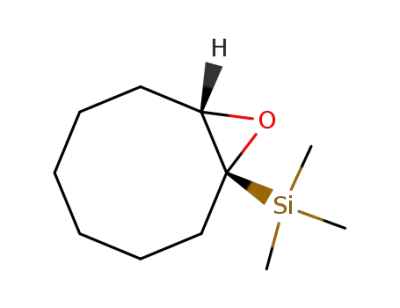
(+/-)-trimethyl(9-oxabicyclo[6.1.0]non-1-yl)silane
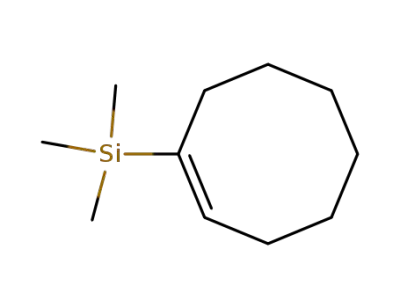
1-trimethylsilyl-cis-cyclo-octene

tetrahydrofuran
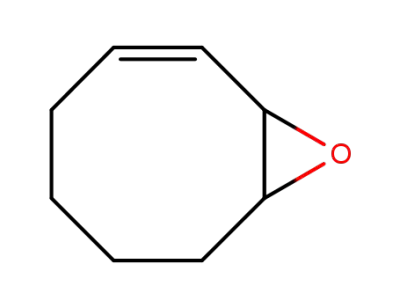
(Z)-9-oxabicyclo[6.1.0]non-2-ene
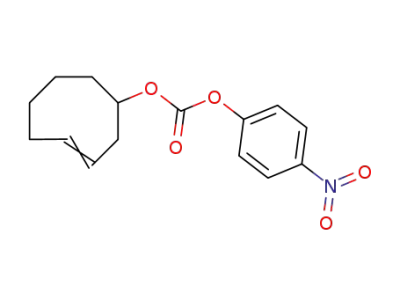
C15H17NO5
CAS:112163-33-4
CAS:112-34-5
CAS:8049-47-6
CAS:58-39-9