- +86-0533-2185556
- WhatsApp: +86 15965530500
- admin@hangyubiotech.com
Your Location:Home >Products >Pesticide >75983-36-7
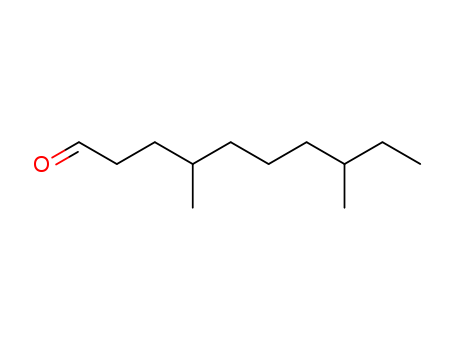

Purity:99%
|
General Description |
4,8-dimethyldecanal is a naturally occurring organic compound primarily found in citrus fruits, characterized by its strong, sweet, and citrus-like aroma. 4,8-dimethyldecanal belongs to the family of Aldehydes, specifically the Aliphatic Acyclic Aldehydes. 4,8-dimethyldecanal is used in the food and beverage industry to give a fruity and citrus flavor, while in the cosmetic industry, it's used as a fragrance ingredient. This substance is typically synthesized in a laboratory setting for industrial usage, but it is also naturally formed during the ripening process of certain citrus fruits, including oranges and mandarins. Despite its common usage, there are concerns about its potential toxicity and allergenic effects, and therefore its usage is regulated in cosmetics and food products. |
InChI:InChI=1/C12H24O/c1-4-11(2)7-5-8-12(3)9-6-10-13/h10-12H,4-9H2,1-3H3
10-Hydroxy-4,8-dimethyldeca-4E,8E-dienoi...
Seven new methyl-branched hydrocarbons w...
To elucidate the deuterium isotope effec...
A convenient synthesis of racemic 4,8-di...
The partial ozonolysis of (E)-2,7-octadi...
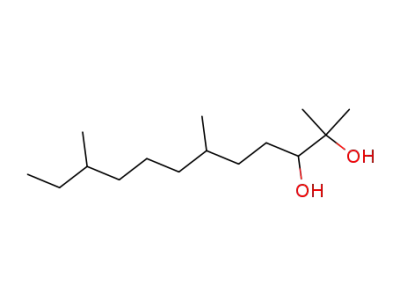
2,6,10-trimethyldodecane-2,3-diol

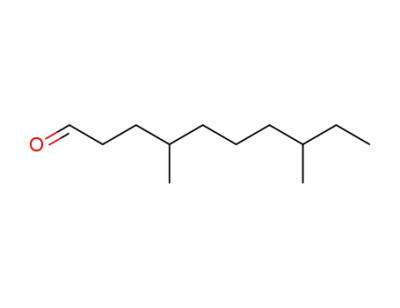
4,8-dimethyldecan-1-al
| Conditions | Yield |
|---|---|
|
With
periodic acid dihydrate;
In
tetrahydrofuran;
at 0 - 5 ℃;
for 0.333333h;
|
73% |

4,8-Dimethyldecan-1-ol


4,8-dimethyldecan-1-al
| Conditions | Yield |
|---|---|
|
With
pyridinium chlorochromate;
In
dichloromethane;
for 1h;
Ambient temperature;
|
68% |
|
With
pyridinium chlorochromate;
In
dichloromethane;
at 20 - 22 ℃;
for 3h;
|
67% |
|
With
pyridinium chlorochromate;
In
dichloromethane;
for 1h;
Yield given;
Ambient temperature;
|
|
|
With
pyridinium chlorochromate;
In
dichloromethane;
|

4,8-Dimethyldecan-1-ol
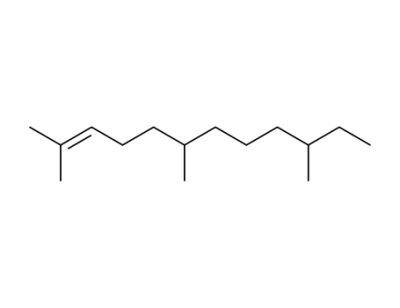
(6R/S,10R/S)-2,6,10-trimethyl-2-dodecene
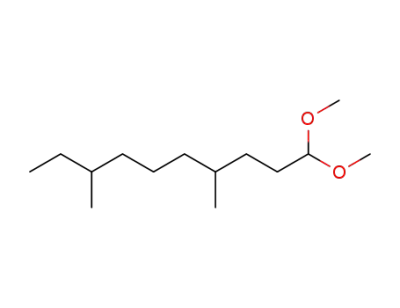
4,8-dimethyl-1,1-dimethoxydecane
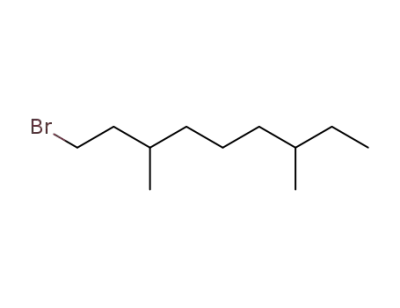
1-bromo-3,7-dimethylnonane
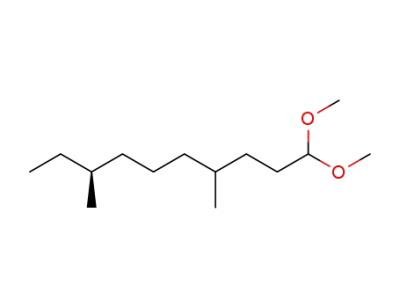
4(RS),8(S)-dimethyldecanal dimethyl acetal
CAS:112163-33-4
CAS:112-34-5
CAS:955371-84-3
CAS:161735-79-1