- +86-0533-2185556
- WhatsApp: +86 15965530500
- admin@hangyubiotech.com
Your Location:Home >Products >API >161735-79-1
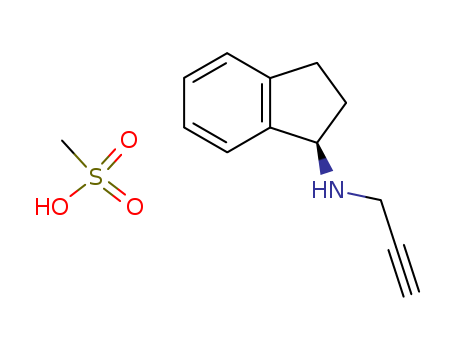

pd_meltingpoint:155-158 °C
Appearance:white to off-white crystalline solid
Purity:99%
|
Indications |
Rasagiline mesylate is a novel, potent, second-generation, selective, irreversible MAO-B inhibitor that blocks the breakdown of dopamine. It is approved for the treatment of PD. Indications for use of once-daily rasagiline are as a monotherapy in early PD and as an adjunct to levodopa in moderate to advanced disease. Rasagiline significantly improves symptoms during initial monotherapy in patients with early PD and as an adjunct treatment to levodopa in moderate-to-advanced patients. Rasagiline is well tolerated up to doses as high as 20 mg/day. Evidence for neuroprotective effect of rasagiline is as follows (Jain 2010c): Structure activity studies have shown that the neuroprotective activity is associated with the propargyl moiety of rasagiline, which protects mitochondrial viability. Experimental evidence supports rasagiline's neuroprotective efficacy, showing that neuronal survival is related to the anti-apoptotic properties of its propargyl moiety. Aminoindan metabolite of rasagiline has been shown to have neuroprotective properties (Bar-Am et al. 2010). |
|
Biochem/physiol Actions |
Rasagiline mesylate is an irreversible inhibitor of monoamine oxidase selective for MAO type B over type A by a factor of fourteen. It has anti-apoptotic and neuroprotectant activity and has been used as a treatment for Parkinson′s disease. |
|
Synthesis |
1-Indanone (122) was condensed with benzyl amine to give corresponding enamine which was reduced with sodium borohydride in ethanol to give racemic Nbenzyl- 1-inda-namine (123) in 82% yield. The racemic benzylamine 123 was resolved with L-tartaric acid and recrystallized from boiling water to give optical pure Rbenzylamine 124 as a tartarate salt. The recovered S-isomer 125 can be racemized under basic condition to give back as the starting racemic 123. Compound 124 was hydrogenated and basified to give free amine 126 in 72 % yield which was alkylated with propargyl chloride and K2CO3 in hot acetonitrile to yield free resagiline. Finally resagiline mesilate (XVII) was obtained by treating resagiline with methanesulfonic acid in refluxing IPA. |
|
General Description |
Rasagiline mesylate, (R)-N-(prop-2-ynyl)-2,3-dihydro-1H-inden-1-amine methanesulfonate(Azilect), belongs to the propargylamine family and is a whiteto off-white powder, soluble in water or ethanol, slightly solublein isopropanol. Rasagiline is rapidly absorbed. Plasmaprotein binding for rasagiline ranges from 88% to 94%, withspecific binding to serum albumin being 61% to 63%. It undergoescomplete biotransformation before excretion, mainlyvia N-dealkylation and hydroxylation, to yield three majormetabolites: 1(R)-aminoindan, 3-hydroxy-N-propargyl-1-aminoindan, and 3-hydroxy-1-aminoindan. Both oxidativepathways are catalyzed by cytochrome P450 (CYP) enzymes,mainly the 1A2 isozyme. Rasagiline and its metabolites undergoglucuronide conjugation with subsequent urinary excretion.Inhibitors of the CYP1A2 may increase plasmaconcentrations of rasagiline up to twofold. Because rasagilineis a selective and irreversible inhibitor of MAO-B, itsduration of action is independent of the drug’s half-life and isinstead determined by the regeneration rate of MAO-B. Thischaracteristic is potentially beneficial in PD, where rasagiline’sprolonged effect may be able to limit the fluctuating responsesthat are characteristic of long-term drug treatmentwith levodopa. |
InChI:InChI=1/C12H13N.CH4O3S/c1-2-9-13-12-8-7-10-5-3-4-6-11(10)12;1-5(2,3)4/h1,3-6,12-13H,7-9H2;1H3,(H,2,3,4)/t12-;/m1./s1
The invention discloses an improved prep...
The invention discloses a preparation me...
A straightforward chemoenzymatic synthes...
The present invention relates to a proce...
(S)-indanol

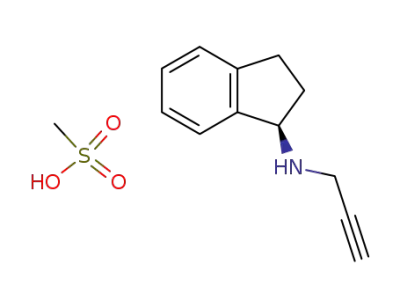
rasagiline mesylate
| Conditions | Yield |
|---|---|
|
Multi-step reaction with 4 steps
1: diphenyl phosphoryl azide; 1,8-diazabicyclo[5.4.0]undec-7-ene / toluene / 22 h / 0 - 20 °C / Inert atmosphere
2: triphenylphosphine; potassium hydroxide; water / tetrahydrofuran / 24 h / 20 °C
3: potassium carbonate / acetonitrile / 16 h / 60 °C
4: isopropyl alcohol / 1 h / Reflux
With
diphenyl phosphoryl azide; water; potassium carbonate; 1,8-diazabicyclo[5.4.0]undec-7-ene; triphenylphosphine; potassium hydroxide;
In
tetrahydrofuran; isopropyl alcohol; toluene; acetonitrile;
1: |Mitsunobu Displacement / 2: |Staudinger Azide Reduction;
|
methanesulfonic acid

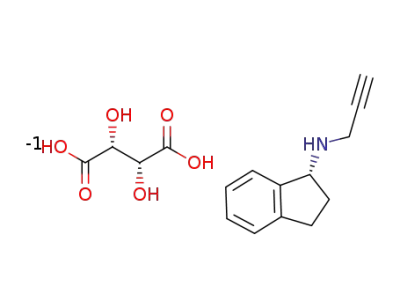
(1R)-N-prop-2-ynyl-2,3-dihydro-1H-inden-1-amine L-(+)-tartrate


rasagiline mesylate
| Conditions | Yield |
|---|---|
|
(1R)-N-prop-2-ynyl-2,3-dihydro-1H-inden-1-amine L-(+)-tartrate;
With
sodium hydrogencarbonate;
In
water; ethyl acetate;
at 20 - 25 ℃;
Inert atmosphere;
methanesulfonic acid;
In
isopropyl alcohol;
Reflux;
|
89% |

methanesulfonic acid

rasagiline
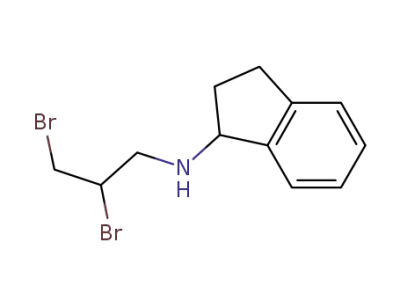
N-(2,3 dibromopropyl)-1-aminoindan
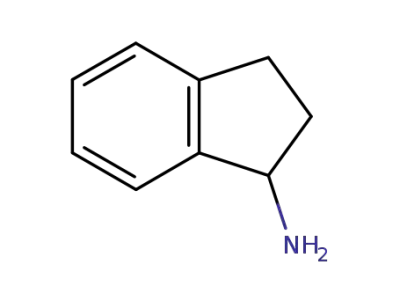
2,3-dihydro-1H-inden-1-amine

rasagiline
CAS:118685-33-9
CAS:66104-23-2
CAS:75983-36-7
CAS:9001-62-1