- +86-0533-2185556
- WhatsApp: +86 15965530500
- admin@hangyubiotech.com
Your Location:Home >Products >Pharmaceutical intermediates >2716-23-6
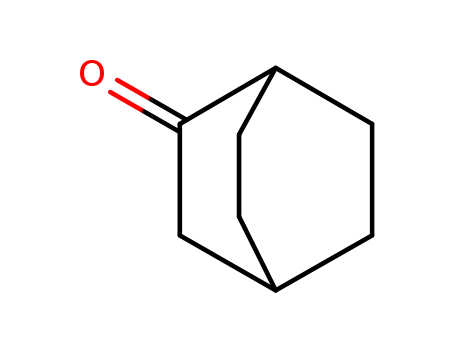

pd_meltingpoint:178-179 °C
Purity:99%
|
General Description |
Bicyclo[2.2.2]octan-2-one is a bicyclic ketone compound with a unique three-dimensional structure. It is often used as a building block in the synthesis of various organic compounds, including pharmaceuticals, agrochemicals, and fragrance ingredients. The compound's rigid and strained ring system confers unique reactivity and stereochemistry, making it a valuable precursor in organic chemistry. Bicyclo[2.2.2]octan-2-one can undergo various chemical reactions to functionalize the carbonyl group or modify the ring structure, allowing for the creation of diverse molecular structures with different properties and applications. |
InChI:InChI=1/C8H12O/c9-8-5-6-1-3-7(8)4-2-6/h6-7H,1-5H2
α-Sulfonyl carbanions undergo oxidative ...
-
-
-
A compound of formula (I) or formula (Ia...
At 300 °C, bicyclo[4.2.0]oct-2-ene (1) i...
The gas phase thermal reactions exhibite...
Several 2-carbalkoxy-2-cyclohepten-1-one...
![bicyclo[2.2.2]octan-2-ol](/upload/2026/5/15285be4-5fdb-4d1f-a537-e5471fcaf38b.png)
bicyclo[2.2.2]octan-2-ol

![bicyclo[2.2.2]octan-2-one](/upload/2026/5/01381920-221f-4045-a0dc-33c0cd76cbe2.png)
bicyclo[2.2.2]octan-2-one
| Conditions | Yield |
|---|---|
|
With
1-hydroxy-3H-benz[d][1,2]iodoxole-1,3-dione;
In
1,1-dichloroethane;
for 1h;
Reflux;
|
87% |
|
With
chromic acid;
In
acetone;
|
70% |
|
With
cycloheptanone;
aluminum isopropoxide;
In
benzene;
at 80 ℃;
Thermodynamic data;
Mechanism;
ΔGox and ΔGr;
|
|
|
With
chromium(VI) oxide;
|
|
|
With
cycloheptanone;
aluminum isopropoxide;
In
benzene;
at 80 ℃;
|
(1S,4S)-(-)-bicyclo<2.2.2>oct-5-en-2-one

![bicyclo[2.2.2]octan-2-one](/upload/2026/5/01381920-221f-4045-a0dc-33c0cd76cbe2.png)
bicyclo[2.2.2]octan-2-one
| Conditions | Yield |
|---|---|
|
With
hydrogen;
palladium on activated charcoal;
|
89% |
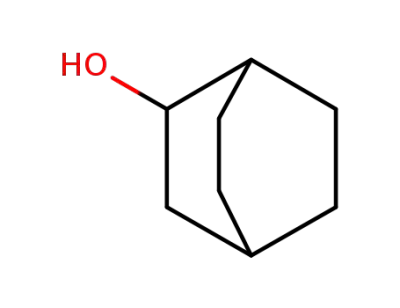
bicyclo[2.2.2]octan-2-ol
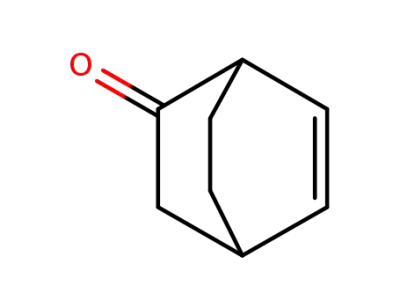
bicyclo[2.2.2]oct-5-en-2-one
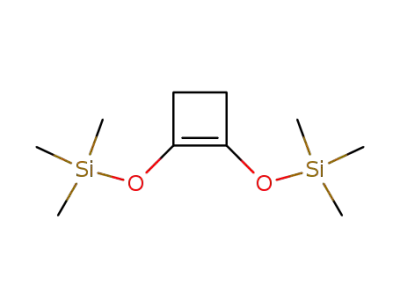
1,2-bis(trimethylsiloxy)cyclobutene
bicyclo[2,2,2]octane
3-benzylidene-bicyclo[2.2.2]octan-2-one
methyl cyclohexylacetate
2-Methyl-bicyclo-<2.2.2>-octanol-(2)

bicyclo[2.2.2]octan-2-ol
CAS:112163-33-4
CAS:112-34-5
CAS:1297538-32-9
CAS:2936625-34-0