- +86-0533-2185556
- WhatsApp: +86 15965530500
- admin@hangyubiotech.com

Purity:99%
|
General Description |
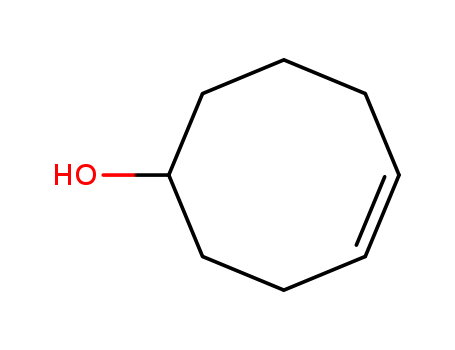
Cyclooct-4-en-1-ol is a chemical compound known for its strong woody, minty, herbal, and camphoraceous odor. This chemical, characterized by a cyclooctene backbone with a hydroxyl group (-OH) at its first carbon and a double bond between its fourth and fifth carbons, primarily functions in creating scents and flavors in product manufacturing. It is considerably utilized in the flavoring and fragrance industry due to its unique aromatic features. However, like any other chemical substance, it may pose risks and hazards when mishandled or used inappropriately. Thus, safety measures, including wearing personal protective types of equipment, should be observed when handling cyclooct-4-en-1-ol. |
InChI:InChI=1/C8H14O/c9-8-6-4-2-1-3-5-7-8/h1-2,8-9H,3-7H2/b2-1-
A simple, cost effective, and readily ac...
trans-Cyclooctenes (TCOs) are essential ...
Bioorthogonal chemistry holds great pote...
The antibody pretargeting approach for r...
cis-1,4-dihydroxycyclooctane

cis-Cyclooctene

3-cyclohexenol

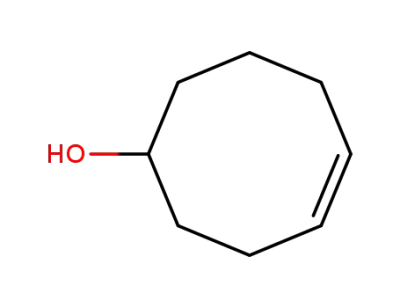
(Z)-cyclooct-4-enol


(E)-cyclooctanol-4-d
| Conditions | Yield |
|---|---|
|
With
lithium triethyl borodeuteride; p-toluenesulfonyl chloride;
Yield given. Multistep reaction. Further byproducts given. Yields of byproduct given;
1.) pyridine, -5 deg C, 24 h, 2.) THF;
|
![(Z)-9-oxabicyclo[6.1.0]non-4-ene](/upload/2026/5/c7125512-f8fc-414d-9689-2cc5ba0711e2.png)
(Z)-9-oxabicyclo[6.1.0]non-4-ene


(Z)-cyclooct-4-enol
| Conditions | Yield |
|---|---|
|
With
lithium aluminium tetrahydride;
In
diethyl ether;
at 20 ℃;
for 4h;
|
96.1% |
|
With
lithium aluminium tetrahydride;
In
tetrahydrofuran;
at 0 ℃;
for 4h;
Reflux;
|
95% |
|
With
lithium aluminium tetrahydride;
In
tetrahydrofuran;
at 0 ℃;
for 4h;
Reflux;
|
95% |
|
With
lithium aluminium tetrahydride;
In
tetrahydrofuran;
at 20 ℃;
for 24h;
|
89% |
|
With
lithium aluminium tetrahydride;
In
tetrahydrofuran;
at 0 - 20 ℃;
|
88% |
|
With
ammonia; sodium;
|
87% |
|
With
lithium aluminium tetrahydride;
In
diethyl ether;
for 24h;
Heating;
|
70% |
|
With
lithium aluminium tetrahydride;
In
diethyl ether;
Yield given;
|
|
|
With
lithium aluminium tetrahydride;
In
diethyl ether;
at 0 - 20 ℃;
|
1.03 g |
|
With
lithium aluminium tetrahydride;
In
tetrahydrofuran;
at 0 - 20 ℃;
|
|
|
With
lithium aluminium tetrahydride;
In
tetrahydrofuran;
at 65 - 70 ℃;
for 4h;
|
|
|
(Z)-9-oxabicyclo[6.1.0]non-4-ene;
With
lithium aluminium tetrahydride;
In
tetrahydrofuran;
at 0 ℃;
Inert atmosphere;
Reflux;
With
water; sodium hydroxide;
In
tetrahydrofuran;
at 0 - 20 ℃;
|
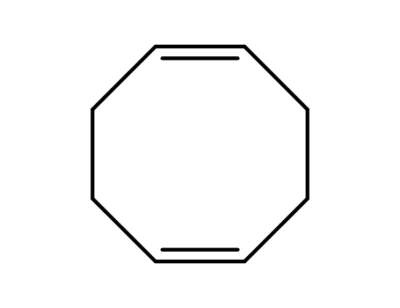
1,5-cis,cis-cyclooctadiene
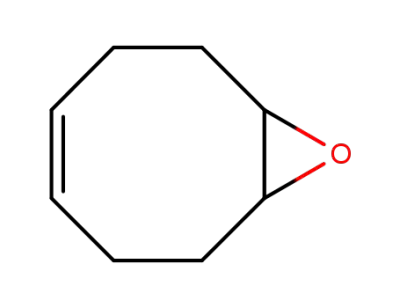
(Z)-9-oxabicyclo[6.1.0]non-4-ene
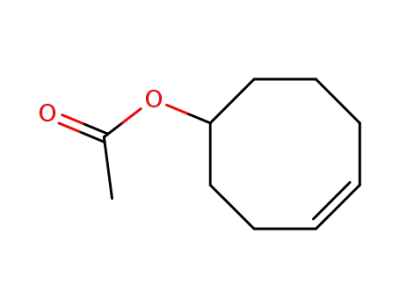
4-cycloocten-1-yl acetate

cis-1,4-dihydroxycyclooctane
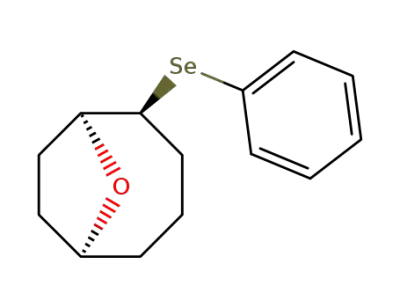
2-(Phenylseleno)-9-oxabicyclo<4.2.1>nonane
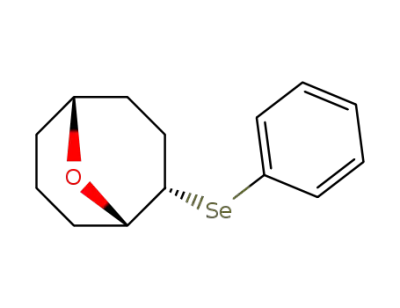
2-(Phenylseleno)-9-oxabicyclo<3.3.1>nonane
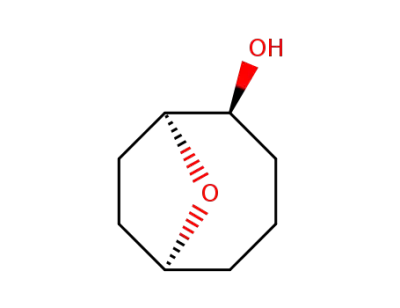
(1S,2S,6S)-9-Oxa-bicyclo[4.2.1]nonan-2-ol
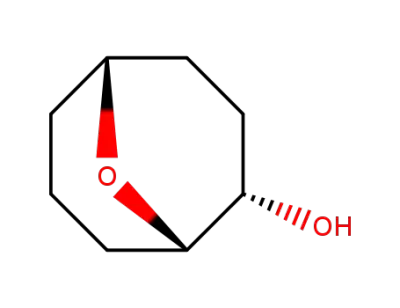
(1S,2S,5R)-9-Oxa-bicyclo[3.3.1]nonan-2-ol
CAS:112163-33-4
CAS:112-34-5
CAS:121776-33-8
CAS:30482-25-8