- +86-0533-2185556
- WhatsApp: +86 15965530500
- admin@hangyubiotech.com
Your Location:Home >Products >API >80565-30-6
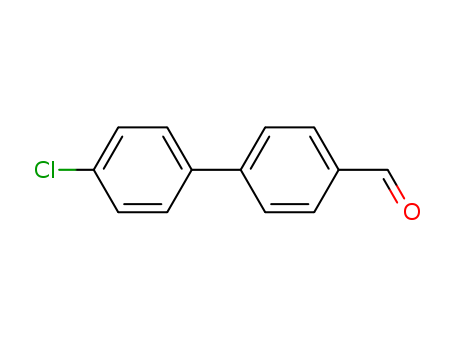
pd_meltingpoint:115-118oC
Purity:99%
Gold-catalyzed reactions of cycloheptatr...
Monoamine oxidase-B (MAO-B) is a well-es...
The Suzuki-Miyaura coupling reaction of ...
The synthesis and characterization of a ...

7-(4-chlorophenyl)-cyclohepta-1,3,5-triene

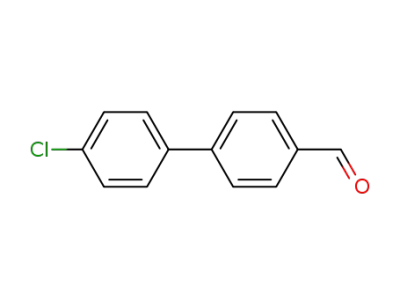
4'-chlorobiphenyl-4-carbaldehyde

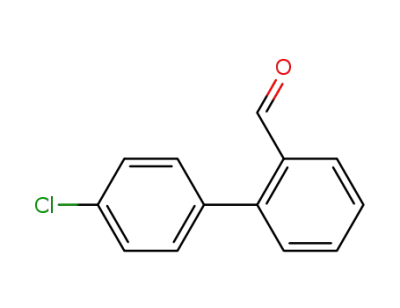
4'-chlorobiphenyl-2-carboxaldehyde
| Conditions | Yield |
|---|---|
|
With
2-(di-tert-butylphosphino)-1,1'-biphenylgold(I) chloride; oxygen; sodium tetrakis[(3,5-di-trifluoromethyl)phenyl]borate; Nitrosobenzene; copper(l) chloride;
In
dichloromethane;
at 23 ℃;
Inert atmosphere;
Molecular sieve;
|
38% 19% |
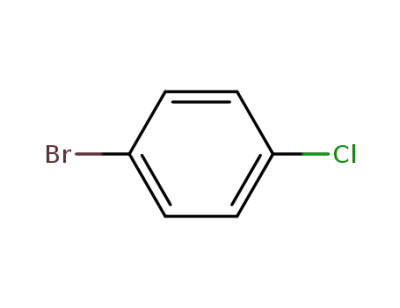
bromochlorobenzene

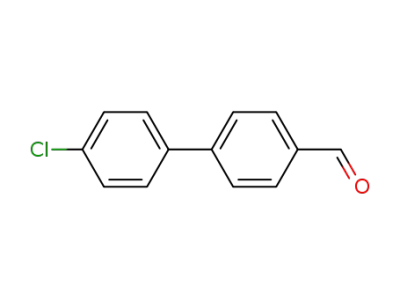
4'-chlorobiphenyl-4-carbaldehyde


4'-chlorobiphenyl-2-carboxaldehyde
| Conditions | Yield |
|---|---|
|
Multi-step reaction with 2 steps
1.1: n-butyllithium / hexane; tetrahydrofuran / 0.5 h / -78 °C / Inert atmosphere
1.2: 12 h / -78 - 23 °C / Inert atmosphere
2.1: sodium tetrakis[(3,5-di-trifluoromethyl)phenyl]borate; copper(l) chloride; oxygen; 2-(di-tert-butylphosphino)-1,1'-biphenylgold(I) chloride; Nitrosobenzene / dichloromethane / 23 °C / Inert atmosphere; Molecular sieve
With
n-butyllithium; 2-(di-tert-butylphosphino)-1,1'-biphenylgold(I) chloride; oxygen; sodium tetrakis[(3,5-di-trifluoromethyl)phenyl]borate; Nitrosobenzene; copper(l) chloride;
In
tetrahydrofuran; hexane; dichloromethane;
|
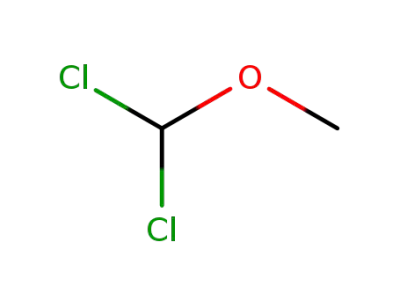
Dichloromethyl methyl ether

4'-biphenyl chloride
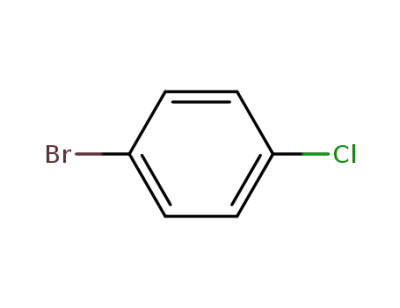
bromochlorobenzene
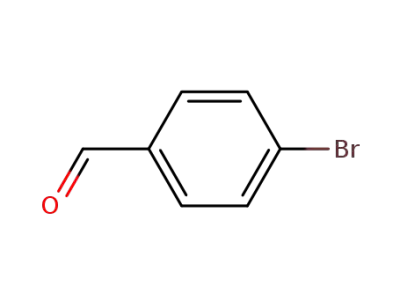
4-bromo-benzaldehyde
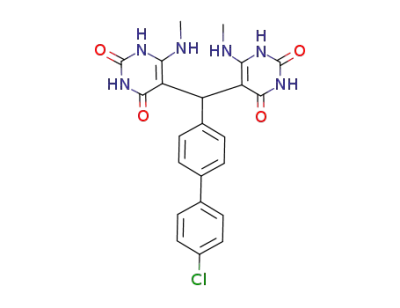
5,5'-<4'-chloro-1,1'-biphenyl>-4-ylmethylenebis(6-methylaminouracil)
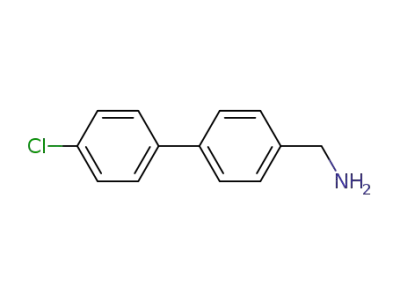
(4'-chloro-[1,1'-biphenyl]-4-yl)methanamine
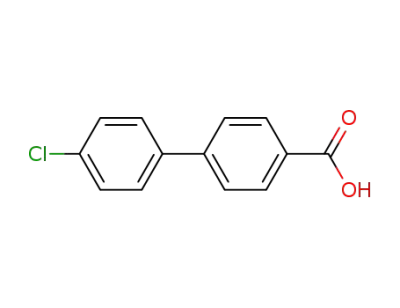
4'-chlorobiphenyl-4-carboxylic acid
CAS:115473-15-9
CAS:118685-33-9
CAS:41294-56-8
CAS:202821-81-6