- +86-0533-2185556
- WhatsApp: +86 15965530500
- admin@hangyubiotech.com
Your Location:Home >Products >Inhibitor >202821-81-6
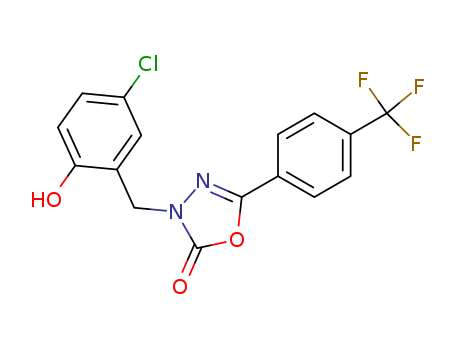

Purity:99%
|
Biological Activity |
Potent Maxi-K channel opener (large-conductance Ca 2+ -activated potassium channel, K Ca 1.1, BK Ca ). Neuroprotectant in two distinct animal models of stroke (MCAO in the SHR rat and a normotensive model of focal stroke). |
|
references |
[1]. hewawasam p, ding m, chen n, et al. synthesis of water-soluble prodrugs of bms-191011: a maxi-k channel opener targeted for post-stroke neuroprotection. bioorganic & medicinal chemistry letters, 2003, 13(10): 1695-1698.[2]. valverde ma, rojas p, amigo j, et al. acute activation of maxi-k channels (hslo) by estradiol binding to the β subunit. science, 1999, 285(5435): 1929-1931.[3]. zhang y, bonnan a, bony g, et al. dendritic channelopathies contribute to neocortical and sensory hyperexcitability in fmr1-/y mice. nature neuroscience, 2014, 17(12): 1701-1709.[4]. gu xq, pamenter me, siemen d, et al. mitochondrial but not plasmalemmal bk channels are hypoxia-sensitive in human glioma. glia, 2014, 62(4): 504-513.[5]. mori a, suzuki s, sakamoto k, et al. bms-191011, an opener of large-conductance ca2+-activated potassium channels, dilates rat retinal arterioles in vivo. biological and pharmaceutical bulletin, 2011, 34(1): 150-152.[6]. romine jl, martin sw, meanwell na, et al. 3-[(5-chloro-2-hydroxyphenyl) methyl]-5-[4-(trifluoromethyl) phenyl]-1, 3, 4-oxadiazol-2 (3 h)-one, bms-191011: opener of large-conductance ca2+-activated potassium (maxi-k) channels, identification, solubility, and sar. journal of medicinal chemistry, 2007, 50(3): 528-542. |
InChI:InChI=1/C16H10ClF3N2O3/c17-12-5-6-13(23)10(7-12)8-22-15(24)25-14(21-22)9-1-3-11(4-2-9)16(18,19)20/h1-7,23H,8H2
The invention relates to a synthetic met...
A novel palladium-catalyzed oxidative an...
Compound 8a (BMS-191011), an opener of t...
The present invention provides novel pho...
![3-[(5-Chloro-2-methoxyphenyl)methyl]-5-[4-(trifluoromethyl)-phenyl]-1,3,4-oxadiazol-2(3H)-one](/upload/2026/5/cc62d978-e27e-4199-908d-377579494e18.png)
3-[(5-Chloro-2-methoxyphenyl)methyl]-5-[4-(trifluoromethyl)-phenyl]-1,3,4-oxadiazol-2(3H)-one

![3-[(5-Chloro-2-hydroxyphenyl)methyl]-5-[4-(trifluoromethyl)-phenyl]-1,3,4-oxadiazol-2(3H)-one](/upload/2026/5/6a8e935f-9399-4b6f-bec8-9bd555d0548a.png)
3-[(5-Chloro-2-hydroxyphenyl)methyl]-5-[4-(trifluoromethyl)-phenyl]-1,3,4-oxadiazol-2(3H)-one
| Conditions | Yield |
|---|---|
|
With
methyl bromide;
In
dichloromethane;
at 0 - 20 ℃;
for 18h;
Inert atmosphere;
|
90% |
|
With
pyridine hydrochloride;
for 1h;
Heating;
|
86% |
|
With
hydrogenchloride; pyridine hydrochloride;
|
|
|
With
hydrogenchloride; pyridine hydrochloride;
|
|
|
With
hydrogenchloride; pyridine hydrochloride;
|
|
|
With
hydrogenchloride; pyridine hydrochloride;
|
|
|
With
hydrogenchloride; pyridine hydrochloride;
|
|
|
With
pyridine hydrochloride;
|
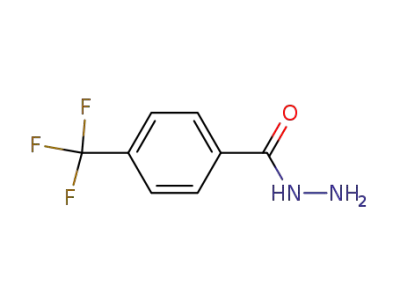
4-(trifluoromethyl)benzoic acid hydrazide

![3-[(5-Chloro-2-hydroxyphenyl)methyl]-5-[4-(trifluoromethyl)-phenyl]-1,3,4-oxadiazol-2(3H)-one](/upload/2026/5/6a8e935f-9399-4b6f-bec8-9bd555d0548a.png)
3-[(5-Chloro-2-hydroxyphenyl)methyl]-5-[4-(trifluoromethyl)-phenyl]-1,3,4-oxadiazol-2(3H)-one
| Conditions | Yield |
|---|---|
|
Multi-step reaction with 3 steps
1: 89 percent / Et3N / tetrahydrofuran / 18 h / 24 °C
2: 92 percent / NaH / dimethylformamide / 18 h / 60 °C
3: 86 percent / pyridine hydrochloride / 1 h / Heating
With
pyridine hydrochloride; sodium hydride; triethylamine;
In
tetrahydrofuran; N,N-dimethyl-formamide;
|
|
|
Multi-step reaction with 3 steps
1: palladium(II) trifluoroacetate; trifluoroacetic acid / acetonitrile / 12 h / 60 °C
2: sodium hydride / N,N-dimethyl-formamide; mineral oil / 18 h / 60 °C / Inert atmosphere
3: methyl bromide / dichloromethane / 18 h / 0 - 20 °C / Inert atmosphere
With
methyl bromide; palladium(II) trifluoroacetate; sodium hydride; trifluoroacetic acid;
In
dichloromethane; N,N-dimethyl-formamide; acetonitrile; mineral oil;
|
|
|
Multi-step reaction with 3 steps
1: palladium(II) trifluoroacetate; trifluoroacetic acid / acetonitrile / 12 h / 60 °C
2: sodium hydride / N,N-dimethyl-formamide; mineral oil / 18 h / 60 °C / Inert atmosphere
3: methyl bromide / dichloromethane / 18 h / 0 - 20 °C / Inert atmosphere
With
methyl bromide; palladium(II) trifluoroacetate; sodium hydride; trifluoroacetic acid;
In
dichloromethane; N,N-dimethyl-formamide; acetonitrile; mineral oil;
|
|
|
Multi-step reaction with 3 steps
1: palladium(II) trifluoroacetate; trifluoroacetic acid / acetonitrile / 12 h / 60 °C
2: sodium hydride / N,N-dimethyl-formamide; mineral oil / 2 h / 0 °C / Inert atmosphere
3: methyl bromide / dichloromethane / 18 h / 0 - 20 °C / Inert atmosphere
With
methyl bromide; palladium(II) trifluoroacetate; sodium hydride; trifluoroacetic acid;
In
dichloromethane; N,N-dimethyl-formamide; acetonitrile; mineral oil;
|
|
|
Multi-step reaction with 3 steps
1: palladium(II) trifluoroacetate; trifluoroacetic acid / acetonitrile / 12 h / 60 °C
2: triphenylphosphine; diethylazodicarboxylate / tetrahydrofuran / 24 h / 0 °C / Inert atmosphere
3: methyl bromide / dichloromethane / 18 h / 0 - 20 °C / Inert atmosphere
With
methyl bromide; palladium(II) trifluoroacetate; triphenylphosphine; trifluoroacetic acid; diethylazodicarboxylate;
In
tetrahydrofuran; dichloromethane; acetonitrile;
2: |Mitsunobu Displacement;
|
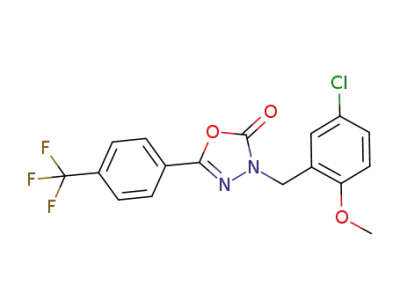
3-[(5-Chloro-2-methoxyphenyl)methyl]-5-[4-(trifluoromethyl)-phenyl]-1,3,4-oxadiazol-2(3H)-one

4-(trifluoromethyl)benzoic acid hydrazide
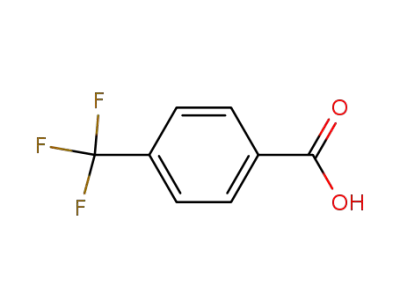
4-trifluoromethylbenzoic acid
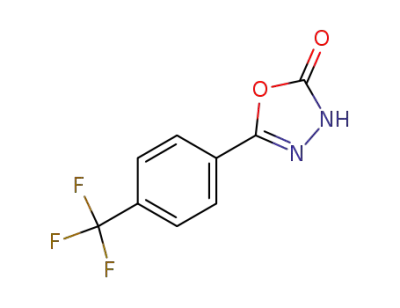
5-[4-(trifluoromethyl)phenyl]-1,3,4-oxadiazol-2-(3H)-one
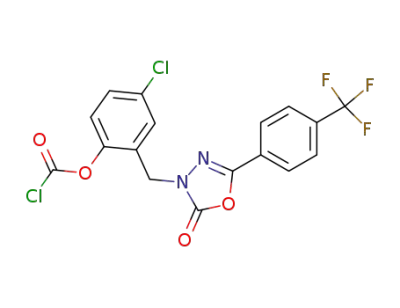
4-Chloro-2-[[5-[4-(trifluoromethyl)phenyl]-2,3-dihydro-2-oxo-1,3,4-oxadiazol-3-yl]methyl]phenyl chloroformate
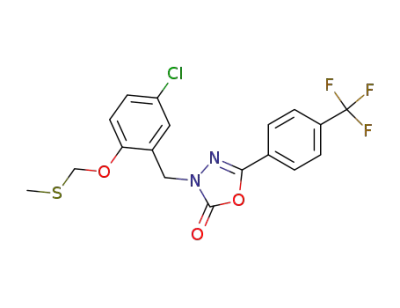
3-[(5-chloro-2-[(methylthiomethyl)oxy]-phenyl)methyl]-5-[4-(trifluoromethyl)phenyl]-1,3,4-oxadiazol-2(3H)-one
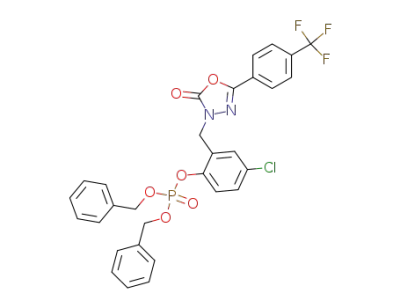
3-[(5-Chloro-2-[[bis[phenylmethyl]phosphono]oxy]phenyl)-methyl]-5-[4-(trifluoromethyl)phenyl]-1,3,4-oxadiazol-2(3H)-one
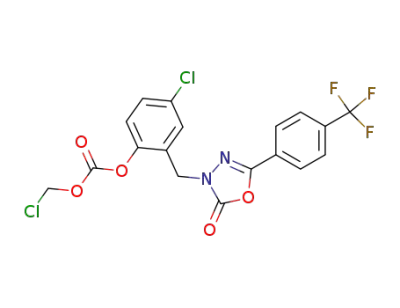
3-[(5-Chloro-2-[[(chloromethyl)oxy]carbonyloxy]phenyl)methyl]-5-[4-(trifluoromethyl)phenyl]-1,3,4-oxadiazol-2(3 H)-one
CAS:755037-03-7
CAS:80565-30-6
CAS:8047-67-4