- +86-0533-2185556
- WhatsApp: +86 15965530500
- admin@hangyubiotech.com
Your Location:Home >Products >Pharmaceutical intermediates >87443-64-9
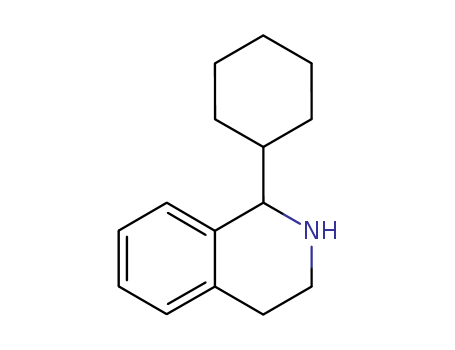

Purity:99%
Histone deacetylase 8 (HDAC8) is a promi...
The present invention relates to an irid...
A cyclometallated iridium complex contai...
Cyclometallated Cp*Ir(N∧C)Cl complexes d...
C15H19N

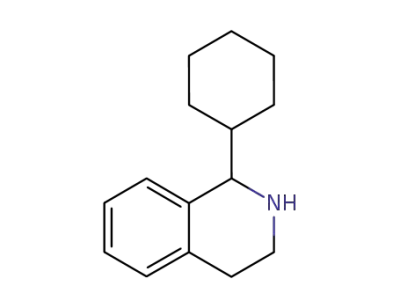
1-cyclohexyl-1,2,3,4-tetrahydroisoquinoline
| Conditions | Yield |
|---|---|
|
With
C25H29ClIrN2O3; hydrogen;
In
2,2,2-trifluoroethanol;
at 75 ℃;
for 2h;
under 15001.5 Torr;
|
95% |
|
With
C25H28ClIrN2O3; hydrogen;
In
2,2,2-trifluoroethanol;
at 75 ℃;
for 2h;
under 15001.5 Torr;
|
95% |
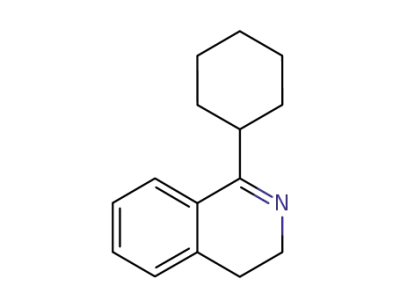
1-cyclohexyl-3,4-dihydroisoquinoline


1-cyclohexyl-1,2,3,4-tetrahydroisoquinoline
| Conditions | Yield |
|---|---|
|
With
C25H28ClIrN2O3; hydrogen;
In
2,2,2-trifluoroethanol;
at 75 ℃;
for 2h;
under 15001.5 Torr;
chemoselective reaction;
Autoclave;
|
93% |
|
With
C26H29ClIrNO3; hydrogen;
In
2,2,2-trifluoroethanol;
at 20 ℃;
for 3h;
under 760.051 Torr;
|
89% |
|
With
sodium tetrahydroborate;
In
ethanol;
for 16h;
Yield given;
Heating;
|
|
|
With
sodium tetrahydroborate;
In
ethanol;
at 20 ℃;
for 60h;
|
|
|
With
platinum(IV) oxide; hydrogen;
In
ethanol;
at 20 ℃;
for 12h;
|

1-cyclohexyl-3,4-dihydroisoquinoline
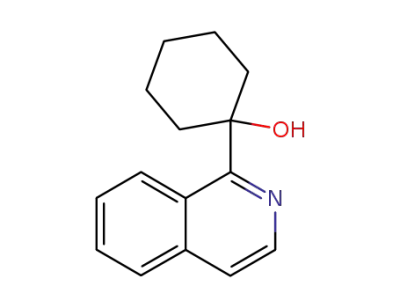
1-[1]isoquinolyl-cyclohexanol
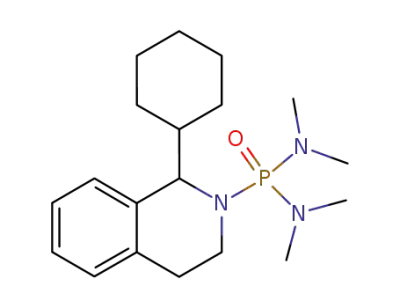
2-bis(dimethylamino)phosphinoyl-1-cyclohexyl-1,2,3,4-tetrahydroisoquinoline
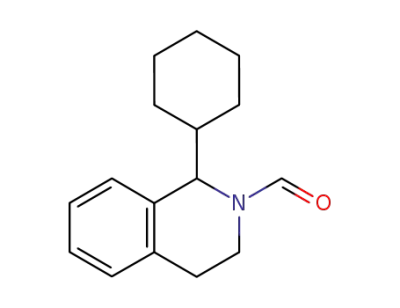
N-formyl-1-cyclohexyl-1,2,3,4-tetrahydroisoquinoline
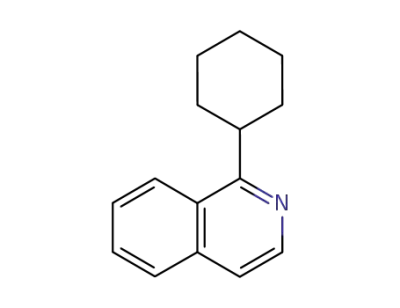
1‐cyclohexylisoquinoline
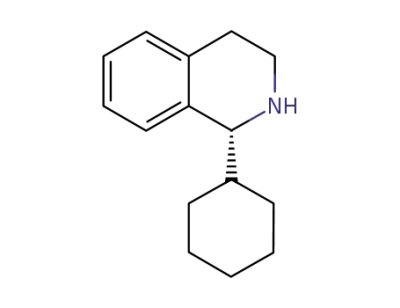
(R)-1-cyclohexyl-1,2,3,4-tetrahydroisoquinoline

1-cyclohexyl-3,4-dihydroisoquinoline
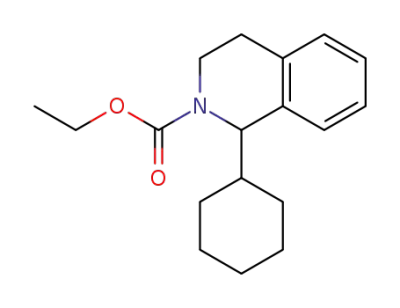
ethyl 1-cyclohexyl-1,2,3,4-tetrahydro-2-isoquinolinecarboxylate
CAS:112163-33-4
CAS:112-84-5
CAS:51-24-1
CAS:637-90-1