- +86-0533-2185556
- WhatsApp: +86 15965530500
- admin@hangyubiotech.com
Your Location:Home >Products >Customization >10147-11-2
Purity:99%
|
Synthesis Reference(s) |
The Journal of Organic Chemistry, 47, p. 1837, 1982 DOI: 10.1021/jo00349a007 |
|
General Description |
3-Phenyl-1-propyne is a 3-aryl-1-propyne. The electronic transition of the resonance-stabilized 1-phenylpropargyl radical, produced in a jet-cooled discharge of 3-phenyl-1-propyne, has been studied by laser-induced fluorescence excitation and dispersed single vibronic level fluorescence (SVLF) spectroscopy. The microwave rotational spectrum of 3-phenyl-1-propyne (propargyl benzene) has been studied and its stable conformation is reported to have coplanar carbon atoms. Reaction of N-methyl-N-phenylhydrazine or N-phenylhydrazine with 3-phenyl-1-propyne is reported to yield indoles. 3-Phenyl-1-propyne is reported to react with styrene oxide and sodium azide, to afford β-hydroxytriazoles. |
InChI:InChI=1/C9H8/c1-2-6-9-7-4-3-5-8-9/h1,3-5,7-8H,6H2
"Hits" with high quantum yields: The scr...
Organovanadium compounds generated in di...
The reaction of alkynylzinc bromides wit...
Phase-transfer catalyzed preparation of ...
-
-
Hydroboration of conjugated enyne alcoho...
Alkenylsilanes, on treatment with iodosy...
Three complementary methods for altering...
An efficient approach for the synthesis ...
(Chemical Equation Presented) Ultimate c...
Terminal alkynes RC≡CH [R = PhCO, PhCH2O...
-
The invention discloses a compound with ...
In the presence of trimethylsilyl triflu...
Ruthenium-catalyzed propargylic reductio...
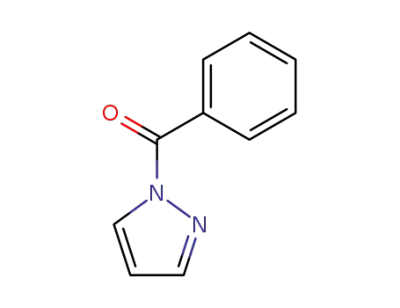
benzoylpyrazole

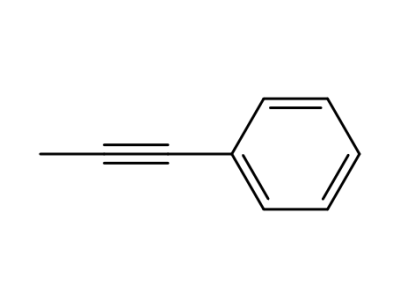
1-Phenylprop-1-yne

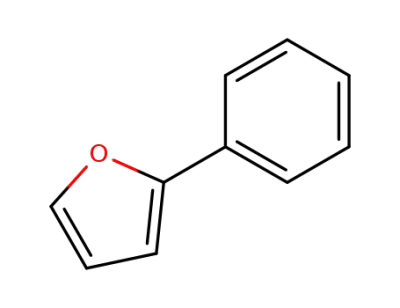
2-phenylfuran

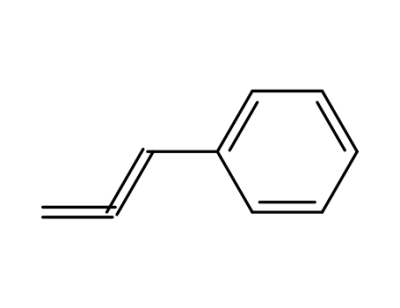
1-phenylpropadiene

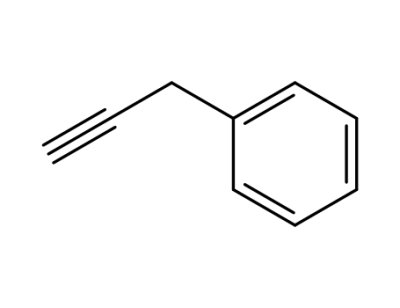
propargyl benzene

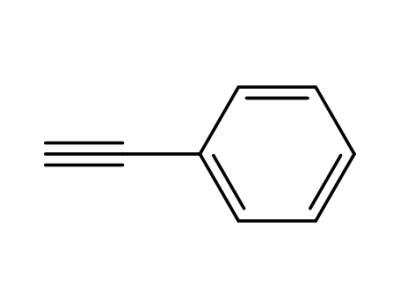
phenylacetylene

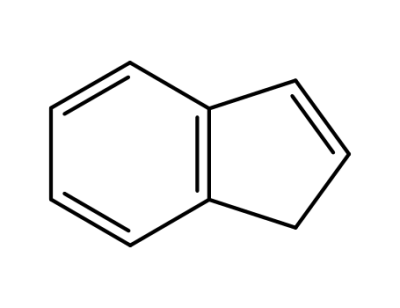
1-indene
| Conditions | Yield |
|---|---|
|
at 900 ℃;
flash-vacuum pyrolysis under low pressure;
|
56% |

1-(trimethylsilyl)-3-phenylprop-1-yne


propargyl benzene
| Conditions | Yield |
|---|---|
|
With
potassium carbonate;
In
methanol;
at 0 ℃;
|
93.5% |
|
With
potassium carbonate;
In
methanol; water;
at 0 - 20 ℃;
for 2h;
|
|
|
With
potassium carbonate;
In
methanol;
at 20 ℃;
for 2h;
Cooling with ice;
|
5 g |
|
With
trimethylsilyl trifluoromethanesulfonate; water;
In
dichloromethane;
at 20 ℃;
for 2h;
|

1-Phenylprop-1-yne
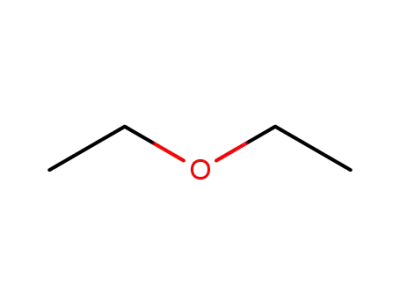
diethyl ether
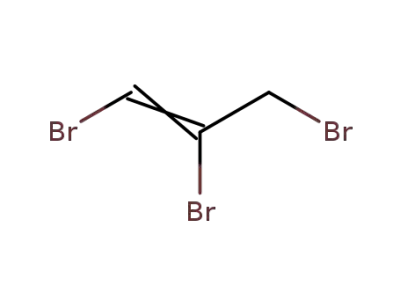
1,2,3-tribromopropene
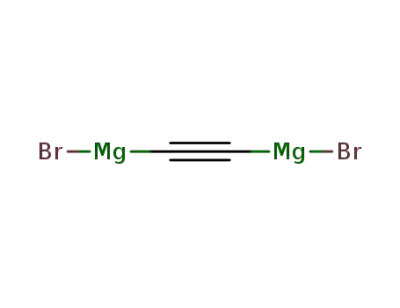
ethynyldimagnesium dibromide
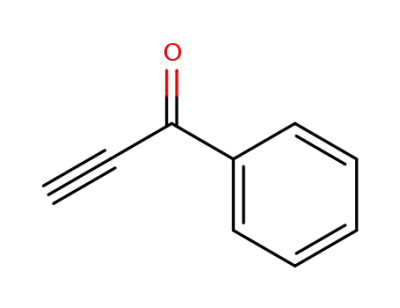
phenyl propargyl ketone

1-Diaethylamino-6-phenylhexadiin-(2,4)
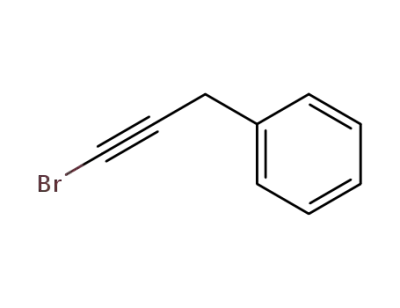
(3-bromoprop-2-yn-1-yl)benzene
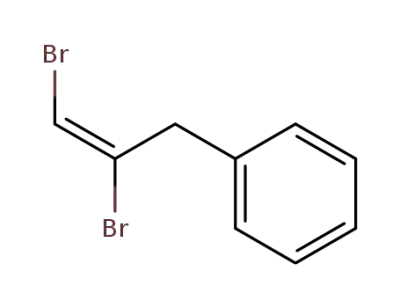
(E)-(2,3-dibromoallyl)benzene
CAS:112163-33-4
CAS:112-34-5
CAS:198481-33-3