- +86-0533-2185556
- WhatsApp: +86 15965530500
- admin@hangyubiotech.com
Your Location:Home >Products >API >198481-33-3

pd_meltingpoint:174-178°
Purity:99%
|
Biological Activity |
bazedoxifene, a novel selective estrogen receptor modulator (serm), has been developed to have favorable effects on bone and the lipid profile while minimizing stimulation of uterine or breast tissues. two large phase iii |
|
Biochem/physiol Actions |
Bazedoxifene is a third generation nonsteroidal selective estrogen receptor modulator (SERM), used clinically to treat postmenopausal osteoporosis. Bazedoxifene binds to estrogen receptor-α with IC50 = 26 nM, similar to that of raloxifene, but lower affinity than 17-β estradiol. Bazedoxifene did not stimulate proliferation of MCF-7 cells, instead inhibited 17β -estradiol-induced proliferation with IC50 = 0.19 nM, exhibiting a desirable profile of agonist/antagonist activity. |
|
Synthesis |
Among many syntheses reported for this drug, the most recent process scale synthesis (multi-kg scale) is highlighted and involves the union of azepane ether 9 and indole 12. 4-Hydroxybenzyl alcohol (6) was converted in two steps to chloride 9 (the Scheme). The reaction of 6 with 2-chloroethyl azepane hydrochloride (7) in a biphasic mixture of sodium hydroxide and toluene in the presence of tetrabutylammonium bromide (TBAB) gave the desired intermediate alcohol 8 in 61% yield. Treatment of 8 with thionyl chloride (SOCl2) gave the requisite chloride 9 in 61% yield. The reaction of 2-bromopropiophenone (10) with an excess of 4-benzyloxy aniline hydrochloride (11) in the presence of triethylamine (TEA) in N,N-dimethylformamide (DMF) at elevated temperatures resulted in indole 12 in 65% yield. Alkylation of 12 with benzylchloride 9 in the presence of sodium hydride (NaH) afforded N-alkylated compound 13. The benzyl ether functionalities from compound 13 were removed via hydrogenolysis and subsequently subjected to acidic conditions, providing diol 14 as the hydrochloride salt in 91% yield. The hydrochloride was then exchanged for the acetate via free base preparation with 5% sodium bicarbonate or triethylamine, followed by treatment with acetic acid giving bazedoxifene acetate (II) in 73–85% yield. |
InChI:InChI=1/C30H34N2O3.C2H4O2/c1-22-28-20-26(34)12-15-29(28)32(30(22)24-8-10-25(33)11-9-24)21-23-6-13-27(14-7-23)35-19-18-31-16-4-2-3-5-17-31;1-2(3)4/h6-15,20,33-34H,2-5,16-19,21H2,1H3;1H3,(H,3,4)
The invention discloses a preparation me...
The invention belongs to the field of me...
The invention relates to a process for p...
The invention discloses a bazedoxifene a...
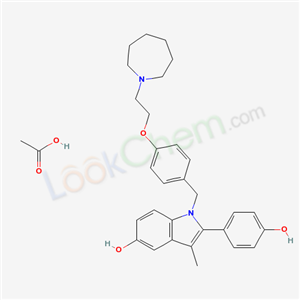
acetic acid

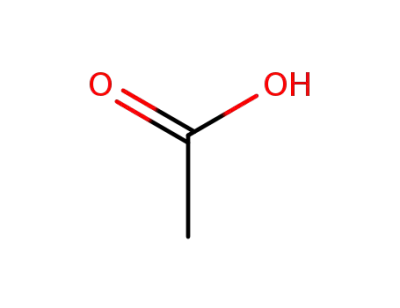
bazedoxifene

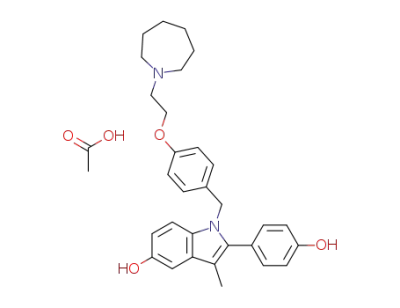
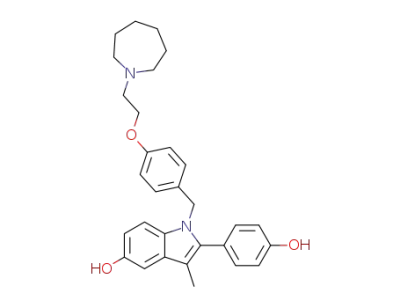
bazedoxifene acetate
| Conditions | Yield |
|---|---|
|
for 6h;
Heating;
|
98.5% |
|
In
ethanol; ethyl acetate;
at 20 - 30 ℃;
|
95.5% |
|
In
acetone;
at 20 - 55 ℃;
|
95% |
|
With
ascorbic acid;
In
isopropyl alcohol;
at 60 ℃;
for 3h;
Solvent;
Inert atmosphere;
Reflux;
|
90% |
|
In
ethanol;
at 40 - 60 ℃;
Large scale;
|
88.3% |
|
In
ethyl acetate;
at 15 - 45 ℃;
Temperature;
Solvent;
Inert atmosphere;
|
84.9% |
|
In
ethyl acetate;
at 50 ℃;
|
74.6% |
|
With
vitamin C;
In
ethanol; ethyl acetate;
at 0 - 20 ℃;
for 4h;
pH=5;
|
|
|
In
acetone;
at 20 - 35 ℃;
|
1.4 g |
|
With
ascorbic acid;
In
ethanol; cyclohexane; acetone;
Inert atmosphere;
|
|
|
With
ascorbic acid;
In
ethanol; ethyl acetate;
at 0 - 20 ℃;
for 4h;
Reagent/catalyst;
|

acetic acid

![5-benzyloxy-2-(4-benzyloxy-phenyl)-3-methyl-1-[4-(2-azepan-1-yl-ethoxy)-benzyl]-1H-indole](/upload/2026/5/1f3a582b-6bdc-43fc-a2a5-e0424fb46d0e.png)
5-benzyloxy-2-(4-benzyloxy-phenyl)-3-methyl-1-[4-(2-azepan-1-yl-ethoxy)-benzyl]-1H-indole


bazedoxifene acetate
| Conditions | Yield |
|---|---|
|
5-benzyloxy-2-(4-benzyloxy-phenyl)-3-methyl-1-[4-(2-azepan-1-yl-ethoxy)-benzyl]-1H-indole;
With
hydrogen;
In
ethanol; ethyl acetate;
at 40 ℃;
under 2250.23 - 3000.3 Torr;
acetic acid;
Temperature;
Pressure;
Reagent/catalyst;
Solvent;
|
96.7% |
|
5-benzyloxy-2-(4-benzyloxy-phenyl)-3-methyl-1-[4-(2-azepan-1-yl-ethoxy)-benzyl]-1H-indole;
With
5%-palladium/activated carbon; ammonium formate;
In
tetrahydrofuran; ethanol;
at 65 ℃;
for 28h;
acetic acid;
at 0 - 5 ℃;
for 2.5h;
Solvent;
Reagent/catalyst;
|
90.5% |
|
5-benzyloxy-2-(4-benzyloxy-phenyl)-3-methyl-1-[4-(2-azepan-1-yl-ethoxy)-benzyl]-1H-indole;
With
palladium 10% on activated carbon; ammonium formate;
In
ethanol;
at 50 - 55 ℃;
for 1h;
acetic acid;
at 20 - 25 ℃;
for 2h;
Temperature;
|
85.3% |
|
5-benzyloxy-2-(4-benzyloxy-phenyl)-3-methyl-1-[4-(2-azepan-1-yl-ethoxy)-benzyl]-1H-indole;
With
hydrogen;
5% palladium over charcoal;
In
ethanol;
at 25 ℃;
for 20h;
under 2585.81 Torr;
acetic acid;
With
ascorbic acid;
at 0 - 20 ℃;
for 4h;
pH=5;
|
74.2% |
|
5-benzyloxy-2-(4-benzyloxy-phenyl)-3-methyl-1-[4-(2-azepan-1-yl-ethoxy)-benzyl]-1H-indole;
With
hydrogen;
palladium 10% on activated carbon;
In
ethanol; ethyl acetate;
at 25 ℃;
for 20h;
under 2585.81 Torr;
acetic acid;
With
ascorbic acid;
In
ethanol; ethyl acetate;
at 0 - 20 ℃;
for 4.16667h;
pH=~ 5;
|
74.2% |
|
5-benzyloxy-2-(4-benzyloxy-phenyl)-3-methyl-1-[4-(2-azepan-1-yl-ethoxy)-benzyl]-1H-indole;
With
palladium 10% on activated carbon; ammonium formate;
In
tetrahydrofuran;
at 45 - 50 ℃;
Large scale;
acetic acid;
With
tert-butyl methyl ether;
In
tetrahydrofuran;
at 10 - 15 ℃;
for 12h;
Large scale;
|
72% |
|
5-benzyloxy-2-(4-benzyloxy-phenyl)-3-methyl-1-[4-(2-azepan-1-yl-ethoxy)-benzyl]-1H-indole;
With
hydrogen;
2.34% Pd/C;
In
ethyl acetate;
for 24h;
acetic acid;
In
ethanol; ethyl acetate;
Cooling with ice;
|
41% |
|
5-benzyloxy-2-(4-benzyloxy-phenyl)-3-methyl-1-[4-(2-azepan-1-yl-ethoxy)-benzyl]-1H-indole;
With
hydrogen;
palladium on carbon;
In
methanol; ethyl acetate;
at 50 ℃;
for 24h;
under 3750.38 Torr;
acetic acid;
With
ascorbic acid;
In
methanol; ethyl acetate;
at 30 ℃;
for 4h;
Product distribution / selectivity;
|

acetic acid

bazedoxifene
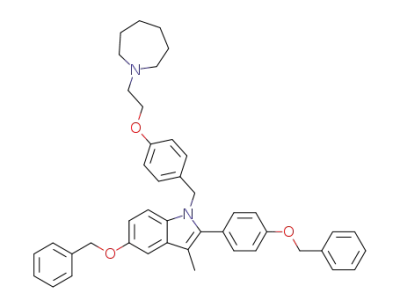
5-benzyloxy-2-(4-benzyloxy-phenyl)-3-methyl-1-[4-(2-azepan-1-yl-ethoxy)-benzyl]-1H-indole
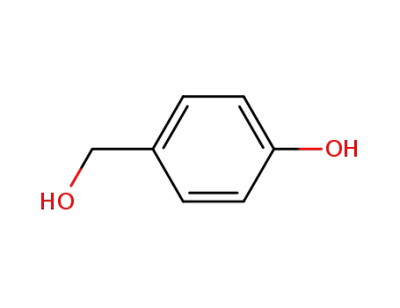
(4-hydroxyphenyl)methanol
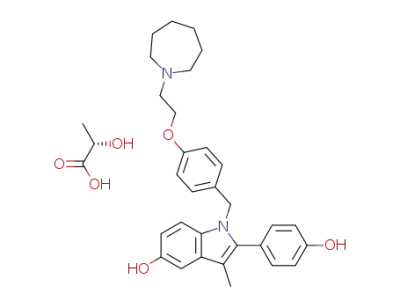
bazedoxifene L-lactate
CAS:112163-33-4
CAS:112-84-5
CAS:10147-11-2
CAS:5557-31-3