- +86-0533-2185556
- WhatsApp: +86 15965530500
- admin@hangyubiotech.com
Your Location:Home >Products >Pharmaceutical intermediates >6531-38-0
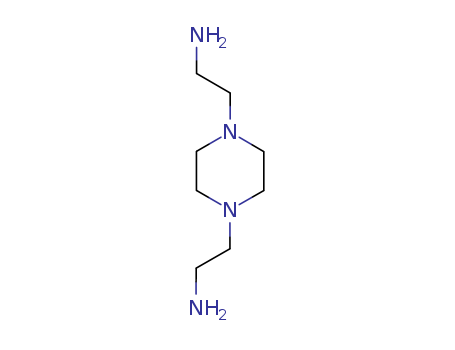

pd_meltingpoint:40 °C
Purity:99%
|
General Description |
Piperazine-1,4-diethylamine, also known as 1,4-bis(2-ethylhexyl)piperazine, is a chemical compound that is often used in the synthesis of other chemicals, including pharmaceutical products. It has a molecular formula of C14H32N2 and it exists in a liquid state at room temperature. Piperazine-1,4-diethylamine is colorless, possesses a mild odor, and is soluble in most organic solvents. It should be handled with care as it is corrosive and can cause significant eye, skin, and respiratory irritation. It may also have harmful effects if swallowed or inhaled. |
InChI:InChI=1/C8H20N4/c9-1-3-11-5-7-12(4-2-10)8-6-11/h1-10H2
The kinetic and thermodynamic aspects of...
Two new tetranuclear CuII complexes [Cu4...
Three pyridine-based macrocyclic ligands...
In alternative embodiments, provided are...
[Figure not available: see fulltext.] An...
![2‐[4‐(cyanomethyl)piperazin‐1‐yl]acetonitrile](/upload/2026/5/5b5dc8cf-a5f9-4af3-95fd-e4da7f05545d.png)
2‐[4‐(cyanomethyl)piperazin‐1‐yl]acetonitrile

1,4-bis(2-aminoethyl)piperazine
| Conditions | Yield |
|---|---|
|
With
Raney nickel;
In
methanol; water;
|
90% |
|
With
lithium aluminium tetrahydride;
In
tetrahydrofuran;
for 4h;
Reflux;
|
87% |
|
2‐[4‐(cyanomethyl)piperazin‐1‐yl]acetonitrile;
With
lithium aluminium tetrahydride;
In
tetrahydrofuran;
for 3h;
Heating / reflux;
With
potassium hydroxide;
In
tetrahydrofuran; water;
|
80% |
|
With
ammonia; hydrogen;
nickel;
In
ethanol;
at 20 ℃;
|
73% |
|
2‐[4‐(cyanomethyl)piperazin‐1‐yl]acetonitrile;
With
lithium aluminium tetrahydride;
In
tetrahydrofuran;
at 0 ℃;
for 3.25h;
Reflux;
With
potassium hydroxide;
In
tetrahydrofuran;
pH=8 - 9;
|
41% |
|
With
lithium aluminium tetrahydride;
In
various solvent(s);
for 1h;
Heating;
|
|
|
With
lithium aluminium tetrahydride;
In
tetrahydrofuran; diethyl ether;
at 90 ℃;
for 4h;
Inert atmosphere;
|
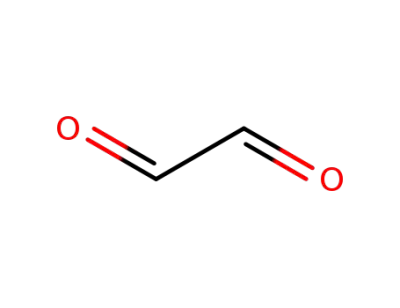
Glyoxal

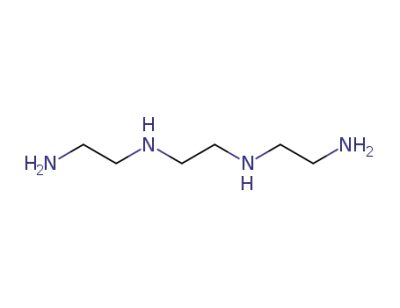
triethylentetramine


1,4-bis(2-aminoethyl)piperazine
| Conditions | Yield |
|---|---|
|
With
sodium tetrahydroborate;
In
ethanol;
at -10 ℃;
for 2h;
|
|
|
With
sodium tetrahydroborate;
|
|
|
With
sodium tetrahydroborate;
In
ethanol;
at 0 ℃;
for 2h;
|
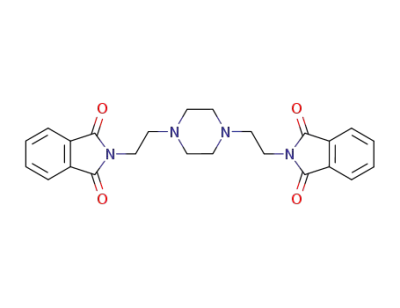
N,N'-bis(2-phthalimidoethyl)piperazine
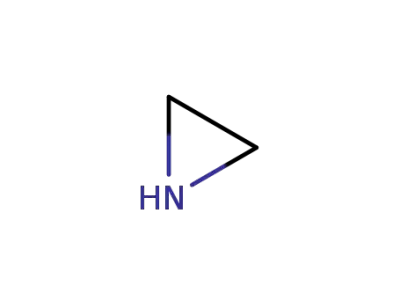
ethyleneimine
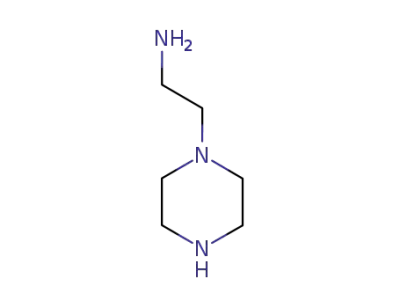
aminoethylpiperazine
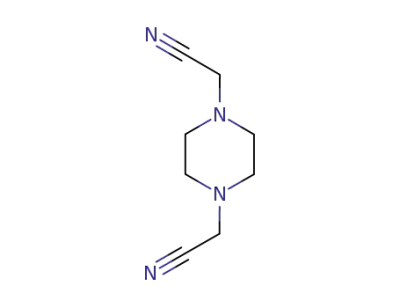
2‐[4‐(cyanomethyl)piperazin‐1‐yl]acetonitrile

N,N'-bis(2-phthalimidoethyl)piperazine
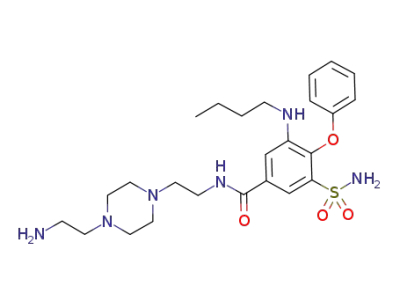
C25H38N6O4S
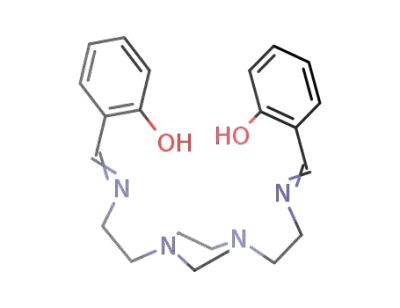
N,N'-bis{2-[(2-ethylimino)methyl]phenol}piperazine
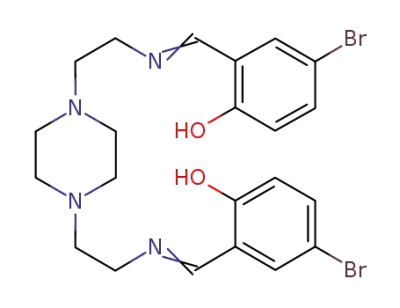
N,N'-bis{2-[(2-ethylimino)methyl]-5-bromophenol}piperazine
CAS:112163-33-4
CAS:112-84-5
CAS:533-45-9