- +86-0533-2185556
- WhatsApp: +86 15965530500
- admin@hangyubiotech.com
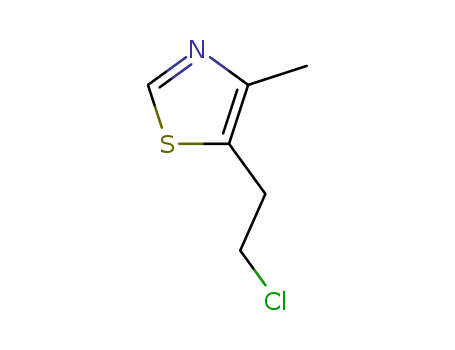

pd_meltingpoint:127-128 °C
Purity:99%
|
World Health Organization (WHO) |
Clomethiazole, which has sedative, anxiolytic and anticonvulsant activity, was introduced in 1960 for the treatment of acute alcohol withdrawal, delirium tremens, status epilepticus, eclamptic toxaemia, sleep disturbances in the elderly and agitation in psychogeriatic patients. It is also used as a sedative in certain anaesthetic procedures. There is little evidence of primary dependence in man but secondary dependence can occur in patients with a history of abuse of other substances, particularly alcohol. Dependence of this type has been reported as a result of inappropriate, long-term prescribing to outpatient alcoholics. Clomethiazole should not be prescribed to alcoholics who continue to drink. Adverse interactions with alcohol have been fatal. Although not controlled under the 1971 Convention on Psychotropic Substances, clomethiazole is subject to analogous controls in some countries. |
|
Biological Activity |
Sedative and anticonvulsant which is neuroprotective in a number of animal models. Prevents the degeneration of serotonergic nerve terminals induced by MDMA ('Ecstasy'). |
|
Brand name |
Clomiazin;Distraneurine;Emineurina;Gebriazol;Hemineurine;Heminevrin;Somnevrin. |
InChI:InChI=1/C6H8ClNS.ClH/c1-5-6(2-3-7)9-4-8-5;/h4H,2-3H2,1H3;1H
-
-
The present invention provides a heteroc...
Since our first discovery of a CD4-mimic...
Learning and memory deficits in Alzheime...
Tumor cells extensively utilize the pent...
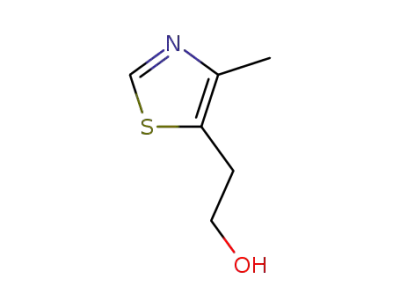
5-hydroxyethyl-4-methylthiazole

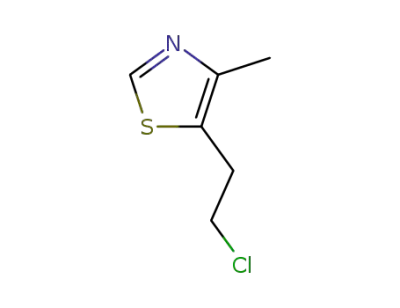
chlormethiazole
| Conditions | Yield |
|---|---|
|
With
thionyl chloride;
for 3h;
Reflux;
|
98% |
|
With
thionyl chloride;
for 2h;
Heating;
|
96% |
|
With
phosphorus pentachloride;
In
dichloromethane;
for 3h;
Heating;
|
95% |
|
With
thionyl chloride;
In
chloroform;
at 0 ℃;
for 1h;
|
88% |
|
With
thionyl chloride;
In
chloroform;
for 4h;
Reflux;
|
84% |
|
With
thionyl chloride;
In
tetrahydrofuran;
at 0 - 80 ℃;
for 1.5h;
|
83% |
|
With
thionyl chloride;
|
|
|
With
hydrogenchloride;
|
|
|
With
pyridine; thionyl chloride;
for 0.75h;
Ambient temperature;
|
|
|
With
methanesulfonyl chloride; N-ethyl-N,N-diisopropylamine;
In
acetonitrile;
|
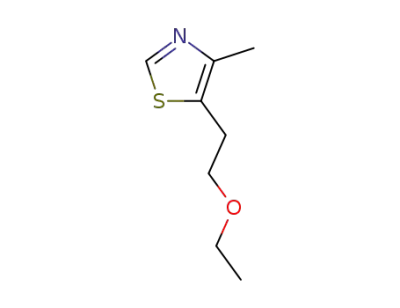
5-(2-ethoxyethyl)-4-methylthiazole


chlormethiazole
| Conditions | Yield |
|---|---|
|
With
hydrogenchloride;
|

5-hydroxyethyl-4-methylthiazole

5-(2-ethoxyethyl)-4-methylthiazole
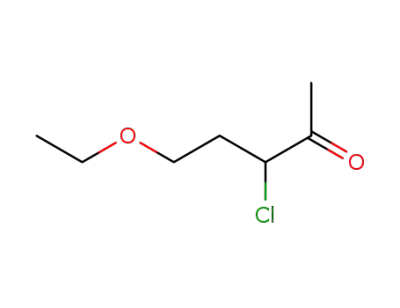
5-ethoxy-3-chloro-pentan-2-one
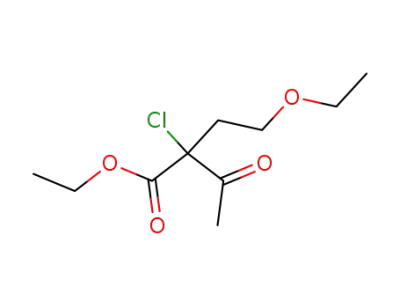
2-(2-ethoxy-ethyl)-2-chloro-acetoacetic acid ethyl ester
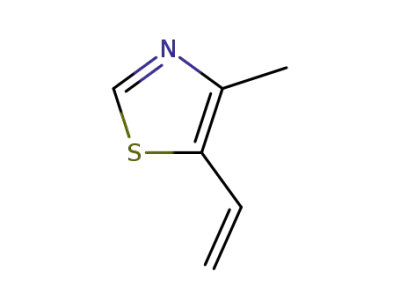
4-methyl-5-vinylthiazole
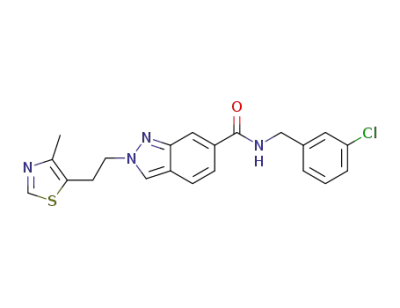
N-(3-Chlorobenzyl)-2-[2-(4-methyl-1,3-thiazol-5-yl)ethyl]-2H-indazole-6-carboxamide
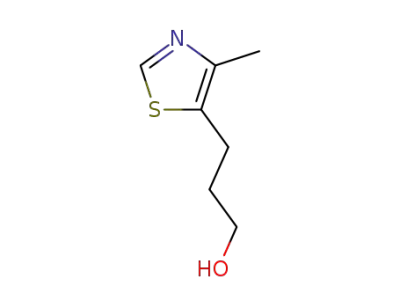
3-(4-methyl-thiazol-5-yl)-propan-1-ol
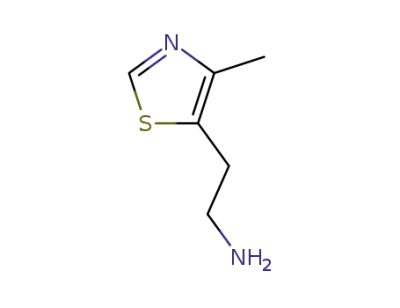
VUF 8952
CAS:115473-15-9
CAS:118685-33-9
CAS:6531-38-0
CAS:1421227-53-3