- +86-0533-2185556
- WhatsApp: +86 15965530500
- admin@hangyubiotech.com
Your Location:Home >Products >API >247068-85-5

pd_meltingpoint:120 - 123° C
Purity:99%
The synthesis of three acetylene functio...
Previously, we reported that immunoprote...
The invention relates to a preparation m...
The ubiquitin proteasome pathway (UPP) p...
Antibody–drug conjugates (ADCs) represen...
![tert-butyl [(1S)-3-methyl-1-(2-methylpropyl)-2-oxobut-3-en-1-yl]carbamate](/upload/2026/5/d385d748-d552-410d-9a3c-edf27888ed03.png)
tert-butyl [(1S)-3-methyl-1-(2-methylpropyl)-2-oxobut-3-en-1-yl]carbamate

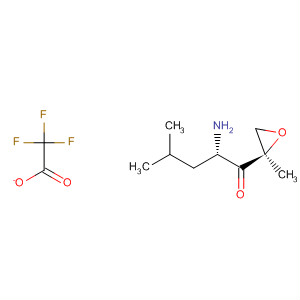
(S)-2-amino-4-methyl-1-((R)-2-methyloxiran-2-yl)pentan-1-one trifluoroacetic acid salt
| Conditions | Yield |
|---|---|
|
Multi-step reaction with 2 steps
1: N-ethyl-N,N-diisopropylamine; dihydrogen peroxide / methanol / 2 h
2: dichloromethane
With
dihydrogen peroxide; N-ethyl-N,N-diisopropylamine;
In
methanol; dichloromethane;
|
|
|
Multi-step reaction with 4 steps
1: sodium tetrahydroborate; cerium(III) chloride heptahydrate / methanol / 0.08 h / 0 °C / Inert atmosphere
2: bis(acetylacetonate)oxovanadium; tert.-butylhydroperoxide / dichloromethane / 1 h / 0 °C / Inert atmosphere
3: Dess-Martin periodane / dichloromethane / 3 h / 0 °C / Inert atmosphere
4: dichloromethane / 0.5 h / 20 °C / Inert atmosphere
With
tert.-butylhydroperoxide; sodium tetrahydroborate; bis(acetylacetonate)oxovanadium; cerium(III) chloride heptahydrate; Dess-Martin periodane;
In
methanol; dichloromethane;
2: |Sharpless Asymmetric Epoxidation / 3: |Dess-Martin Oxidation;
|
|
|
Multi-step reaction with 2 steps
1: calcium hypochlorite / water; 1-methyl-pyrrolidin-2-one / 0.33 h / -15 - 0 °C
2: dichloromethane / 2 h / 0 - 20 °C / Inert atmosphere
With
calcium hypochlorite;
In
1-methyl-pyrrolidin-2-one; dichloromethane; water;
|
|
|
Multi-step reaction with 2 steps
1: sodium hypochlorite; pyridine / water / 2 h / -5 - 0 °C
2: dichloromethane / 2 h / 0 - 20 °C / Inert atmosphere
With
pyridine; sodium hypochlorite;
In
dichloromethane; water;
|
|
|
Multi-step reaction with 4 steps
1: cerium(III) chloride heptahydrate; sodium tetrahydroborate / methanol / 1 h / 0 - 5 °C
2: 3-chloro-benzenecarboperoxoic acid / dichloromethane / 0 - 30 °C
3: Dess-Martin periodane / acetonitrile / 3 h / 0 - 30 °C
4: dichloromethane / 2 h / 2 - 30 °C
With
sodium tetrahydroborate; cerium(III) chloride heptahydrate; Dess-Martin periodane; 3-chloro-benzenecarboperoxoic acid;
In
methanol; dichloromethane; acetonitrile;
|
|
|
Multi-step reaction with 2 steps
1: acetic acid; dihydrogen peroxide; C28H34F6MnN6O6S2 / acetonitrile / 2 h / -20 °C
2: dichloromethane / 4 h / 20 °C
With
C28H34F6MnN6O6S2; dihydrogen peroxide; acetic acid;
In
dichloromethane; acetonitrile;
|
|
|
Multi-step reaction with 4 steps
1: sodium tetrahydroborate; cerium(III) chloride heptahydrate / methanol / 0.42 h / 0 °C
2: bis(acetylacetonate)oxovanadium; tert.-butylhydroperoxide / dichloromethane / 2 h / 0 °C
3: Dess-Martin periodane / dichloromethane / 20 °C
4: dichloromethane / 0.5 h
With
tert.-butylhydroperoxide; sodium tetrahydroborate; bis(acetylacetonate)oxovanadium; cerium(III) chloride heptahydrate; Dess-Martin periodane;
In
methanol; dichloromethane;
|
|
|
Multi-step reaction with 4 steps
1: aluminum isopropoxide; isopropyl alcohol / toluene / 3 h / 50 °C
2: tert.-butylhydroperoxide; tris(acetylacetonato)vanadium(III) / dichloromethane / 12 h / 0 - 20 °C
3: N-ethyl-N,N-diisopropylamine; sulfur trioxide pyridine complex / dimethyl sulfoxide / 6 h
4: dichloromethane / 2 h / 0 - 20 °C
With
tert.-butylhydroperoxide; tris(acetylacetonato)vanadium(III); sulfur trioxide pyridine complex; aluminum isopropoxide; N-ethyl-N,N-diisopropylamine; isopropyl alcohol;
In
dichloromethane; dimethyl sulfoxide; toluene;
|
![tert-butyl [(1S)-3-methyl-1-(2-methylpropyl)-2-oxobut-3-en-1-yl]carbamate](/upload/2026/5/d385d748-d552-410d-9a3c-edf27888ed03.png)
tert-butyl [(1S)-3-methyl-1-(2-methylpropyl)-2-oxobut-3-en-1-yl]carbamate

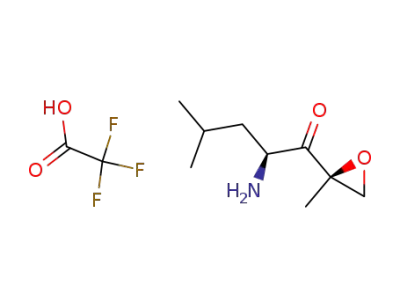
(S)-2-amino-4-methyl-1-((R)-2-methyloxiran-2yl)pentan-1-one trifluoroacetic acid
| Conditions | Yield |
|---|---|
|
Multi-step reaction with 2 steps
1: urea hydrogen peroxide adduct; tetramethyl ammoniumhydroxide / methanol; benzonitrile / 20 h / -10 °C / Inert atmosphere
2: dichloromethane / 0.5 h / 20 °C
With
tetramethyl ammoniumhydroxide; urea hydrogen peroxide adduct;
In
methanol; dichloromethane; benzonitrile;
|
|
|
Multi-step reaction with 2 steps
1: dihydrogen peroxide; N-ethyl-N,N-diisopropylamine / benzonitrile; methanol / 3 h / 0 °C / Inert atmosphere
2: dichloromethane / 20 °C
With
dihydrogen peroxide; N-ethyl-N,N-diisopropylamine;
In
methanol; dichloromethane; benzonitrile;
|
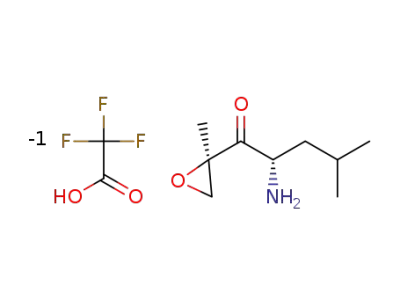
trifluoroacetic acid
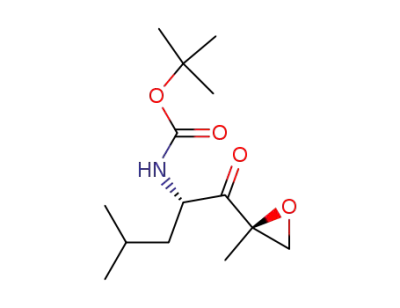
tert-butyl ((2S)-4-methyl-1-((2R)-2-methyloxirane-2-yl)-1-oxopentan-2-yl)carbamate
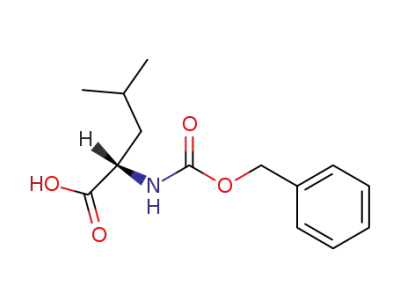
Z-Leu-OH
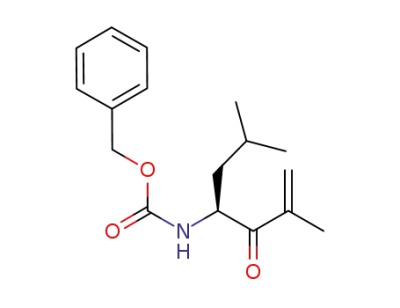
(S)-benzyl (2,6-dimethyl-3-oxohept-1-en-4-yl)carbamate
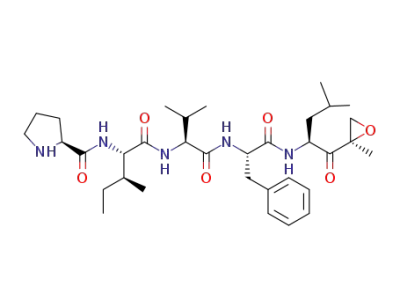
C34H53N5O6

tert-butyl ((2S)-4-methyl-1-((2R)-2-methyloxirane-2-yl)-1-oxopentan-2-yl)carbamate
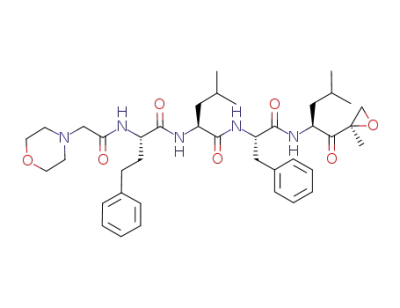
carfilzomib
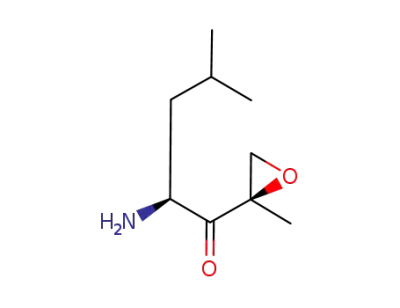
(2S)-2-amino-4-methyl-1-[(2R)-2-methyloxiran-2-yl]pentan-1-one
CAS:874819-74-6
CAS:147098-20-2
CAS:64951-58-2
CAS:1174130-61-0