- +86-0533-2185556
- WhatsApp: +86 15965530500
- admin@hangyubiotech.com
Your Location:Home >Products >API >56341-41-4
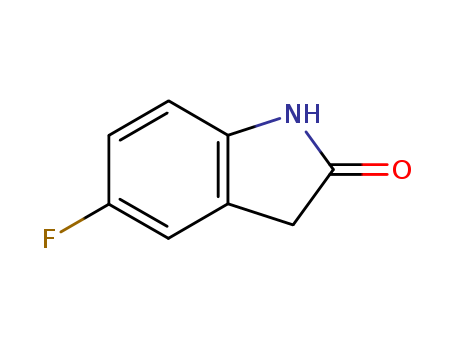

pd_meltingpoint:143-147 °C(lit.)
Appearance:Light Yellow Crystalline
Purity:99%
|
Preparation |
synthesis of 5-fluoro-2-oxindole: 4-fluoroaniline was used as the starting material, reacted with chloral hydrate and hydroxylamine hydrochloride to generate 4-fluoroisonitrosoacetanilide, and then cyclized under the action of concentrated sulfuric acid to form the compound 5-fluoroisatin was finally obtained by wolff-kishner-huang minlon reduction to obtain 5-fluoro-2-oxindole with a total yield of 66%. |
|
Application |
5-Fluoro-2-oxindole is a ketone organic compound that can be used as an intermediate in the preparation of sunitinib. Sunitinib is a novel multi-targeted oral drug for the treatment of tumors. |
InChI:InChI=1/C8H6FNO/c9-6-1-2-7-5(3-6)4-8(11)10-7/h1-3H,4H2,(H,10,11)
Dendrimer compositions and methods for t...
In the past five years, our team had bee...
A novel method for the synthesis of 3-mo...
The invention discloses an enhanced fluo...

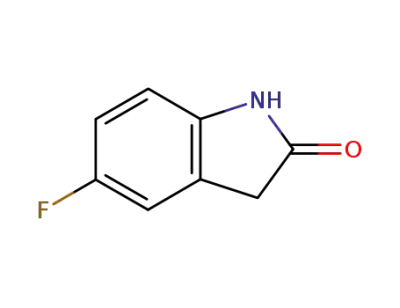
5-fluoroindol-2(3H)-one

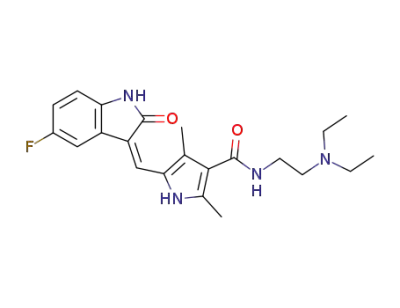
sunitinib
| Conditions | Yield |
|---|---|
|
|
73% |
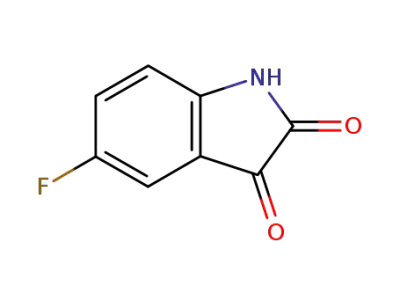
5-fluoro-1H-indole-2,3-dione


5-fluoroindol-2(3H)-one
| Conditions | Yield |
|---|---|
|
With
hydrazine hydrate;
In
water;
at 140 ℃;
for 6h;
Temperature;
|
99.1% |
|
With
titanium tetrachloride; zinc;
In
tetrahydrofuran;
at 20 ℃;
for 0.1h;
Inert atmosphere;
|
87% |
|
5-fluoro-1H-indole-2,3-dione;
With
potassium hydroxide; hydrazine;
In
ethylene glycol;
at 80 ℃;
for 3h;
With
hydrogenchloride; water;
In
ethylene glycol;
at 0 - 20 ℃;
for 12h;
pH=1 - 2;
|
81.9% |
|
With
potassium hydroxide; hydrazine;
In
ethylene glycol;
at 80 ℃;
for 3h;
|
81.9% |
|
In
(2S)-N-methyl-1-phenylpropan-2-amine hydrate; hydrazine hydrate;
|
79% |
|
With
hydrazine hydrate;
at 100 - 130 ℃;
|
78.4% |
|
With
hydrazine;
at 100 - 130 ℃;
for 5.5h;
|
78.4% |
|
5-fluoro-1H-indole-2,3-dione;
With
hydrazine hydrate;
In
water;
at 140 ℃;
for 6h;
With
hydrogenchloride;
In
water;
at 20 ℃;
for 12h;
pH=2;
|
76% |
|
5-fluoro-1H-indole-2,3-dione;
With
hydrazine hydrate;
at 100 ℃;
With
hydrogenchloride; water;
at 25 - 30 ℃;
for 24h;
|
75% |
|
With
hydrazine;
at 100 ℃;
|
73% |
|
With
hydrazine hydrate; triethylamine;
In
butan-1-ol;
at 100 ℃;
for 16h;
|
72% |
|
With
hydrazine hydrate; triethylamine;
In
butan-1-ol;
at 20 - 100 ℃;
for 15.5h;
|
69% |
|
With
hydrazine hydrate;
In
ethanol; water;
at 100 ℃;
for 10h;
|
62.14% |
|
5-fluoro-1H-indole-2,3-dione;
With
hydrazine;
In
butan-1-ol;
at 20 - 80 ℃;
for 3.5h;
With
triethylamine;
In
iso-butanol;
at 100 ℃;
for 12h;
|
57.4% |
|
With
potassium hydroxide; hydrazine hydrate;
In
ethylene glycol;
at 70 ℃;
for 0.5h;
|
|
|
In
(2S)-N-methyl-1-phenylpropan-2-amine hydrate; hydrazine hydrate;
|
|
|
In
(2S)-N-methyl-1-phenylpropan-2-amine hydrate; hydrazine hydrate;
|
|
|
In
(2S)-N-methyl-1-phenylpropan-2-amine hydrate; hydrazine hydrate;
|
|
|
In
(2S)-N-methyl-1-phenylpropan-2-amine hydrate; hydrazine hydrate;
|
|
|
In
(2S)-N-methyl-1-phenylpropan-2-amine hydrate; hydrazine hydrate;
|
|
|
In
(2S)-N-methyl-1-phenylpropan-2-amine hydrate; hydrazine hydrate;
|
|
|
In
(2S)-N-methyl-1-phenylpropan-2-amine hydrate; hydrazine hydrate;
|
|
|
With
hydrogenchloride;
In
water; hydrazine hydrate;
|
|
|
With
hydrogenchloride;
In
water; hydrazine hydrate;
|
|
|
With
hydrazine;
In
water;
for 1h;
Heating / reflux;
|
|
|
With
hydrazine hydrate; potassium hydroxide;
In
ethylene glycol;
at 100 ℃;
for 1h;
|
|
|
Multi-step reaction with 2 steps
1: hydrazine hydrate / methanol / 1 h / Reflux
2: sodium ethanolate / ethanol / 5 h / Reflux
With
sodium ethanolate; hydrazine hydrate;
In
methanol; ethanol;
|
|
|
5-fluoro-1H-indole-2,3-dione;
With
hydrazine hydrate;
In
ethanol;
Reflux;
With
water; sodium hydroxide;
In
ethanol;
Reflux;
|
|
|
5-fluoro-1H-indole-2,3-dione;
With
hydrazine hydrate;
In
ethanol;
for 3h;
Reflux;
With
sodium hydroxide;
In
ethanol;
for 0.5h;
Reflux;
With
hydrogenchloride;
In
ethanol; water;
at 20 ℃;
pH=2;
|
|
|
Multi-step reaction with 2 steps
1: hydrazine hydrate / ethanol / 0.5 h / Inert atmosphere; Reflux
2: potassium tert-butylate / ethanol / 2 h / Reflux; Inert atmosphere
With
potassium tert-butylate; hydrazine hydrate;
In
ethanol;
|
|
|
Multi-step reaction with 2 steps
1: hydrazine hydrate / Reflux
2: sodium ethanolate
With
sodium ethanolate; hydrazine hydrate;
|
|
|
With
hydrazine hydrate; potassium hydroxide;
In
ethylene glycol;
at 110 - 130 ℃;
|
|
|
With
hydrazine hydrate;
at 100 - 110 ℃;
|
|
|
With
hydrazine hydrate; potassium hydroxide;
In
ethylene glycol;
at 110 - 130 ℃;
|
|
|
With
hydrazine hydrate;
at 130 ℃;
|
|
|
With
titanium tetrachloride; zinc;
In
tetrahydrofuran;
at 20 ℃;
for 0.0833333h;
Inert atmosphere;
|
|
|
Multi-step reaction with 2 steps
1: hydrazine hydrate / methanol / 12.5 h / 20 °C / Reflux
2: sodium ethanolate / ethanol / 6 h / Reflux
With
sodium ethanolate; hydrazine hydrate;
In
methanol; ethanol;
|
|
|
5-fluoro-1H-indole-2,3-dione;
With
hydrazine;
In
butan-1-ol;
at 20 - 80 ℃;
for 3.5h;
With
triethylamine;
In
butan-1-ol;
at 100 ℃;
for 2h;
|
2.84 g |
|
With
hydrazine;
In
water;
for 1h;
Heating / reflux;
|
|
|
With
hydrazine hydrate;
In
water;
at 140 ℃;
for 6h;
Temperature;
|
4.17 g |
|
Multi-step reaction with 2 steps
1: sodium carbonate / acetonitrile / 60 °C
2: hydrogen iodide / acetonitrile; water / 6 h / 50 °C
With
hydrogen iodide; sodium carbonate;
In
water; acetonitrile;
|
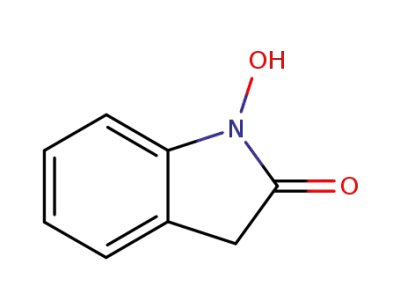
1,3-dihydro-1-hydroxyindole-2-one
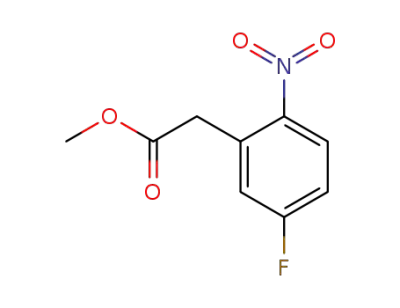
methyl 2-(5-fluoro-2-nitrophenyl)acetate
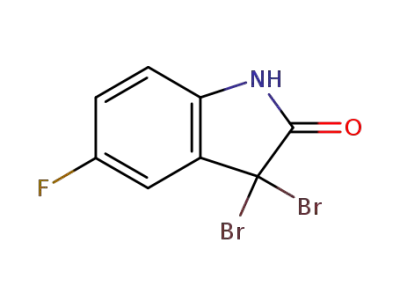
3,3-dibromo-5-fluoro-2-oxindole
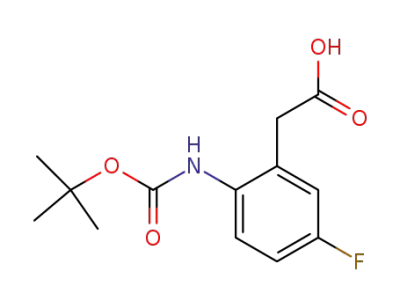
(2-tert-Butoxycarbonylamino-5-fluoro-phenyl)-acetic acid
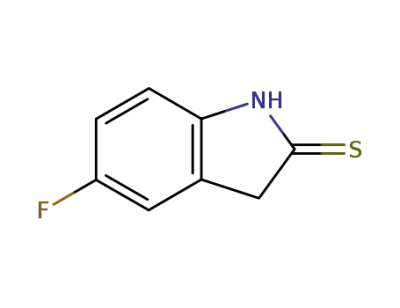
5-fluoroindoline-2-thione
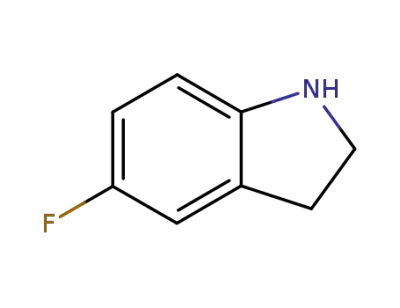
5-fluoroindoline
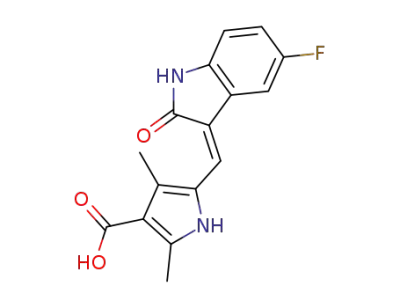
5-((Z)-(5-fluoro-2-oxoindolin-3-ylidene)methyl)-2,4-dimethyl-1H-pyrrole-3-carboxylic acid

sunitinib
CAS:117724-63-7
CAS:391210-10-9
CAS:1446711-81-4
CAS:16009-13-5