- +86-0533-2185556
- WhatsApp: +86 15965530500
- admin@hangyubiotech.com
Your Location:Home >Products >Inhibitor >48208-26-0
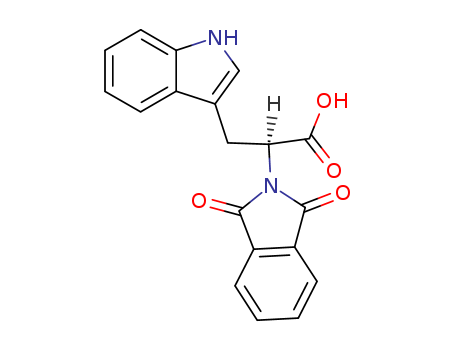

pd_meltingpoint:192-194℃
Purity:99%
|
Biological Activity |
Non-nucleoside DNA methyltransferase inhibitor that blocks the enzyme active site. Inhibits DNA methylation in human cancer cell lines in vitro without detectable toxicity. Demethylates and reactivates epigenetically silenced tumor suppressor genes. |
|
Biochem/physiol Actions |
RG108 is a DNA methyltransferase (DMNT) inhibitor. It reactivates tumor suppressor gene expression (p16, SFRP1, secreted frizzled related protein-1, and TIMP-3) in tumor cells by DNA demethylation. RG108 also inhibits human tumor cell line (HCT116, NALM-6) proliferation and increased doubling time in culture. |
|
General Description |
RG108, a non-nucleoside analog is also called [2-(1,3-dioxoisoindolin-2-yl)-3-(1H-indol-3-yl) propanoic acid. |
InChI:InChI=1/C19H16N2O4/c22-17-13-6-1-2-7-14(13)18(23)21(17)16(19(24)25)9-11-10-20-15-8-4-3-5-12(11)15/h3-8,10,16,20H,1-2,9H2,(H,24,25)/t16-/m0/s1
A convergent synthesis via the late-stag...
A simple strategy for configurational as...
DNA methyltransferases (DNMT) are promis...
A series of phthalimide analogs were syn...
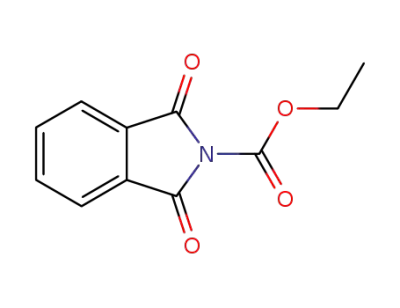
N-ethoxycarbonylphthalimide

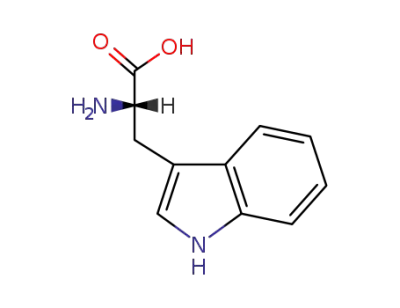
L-Tryptophan

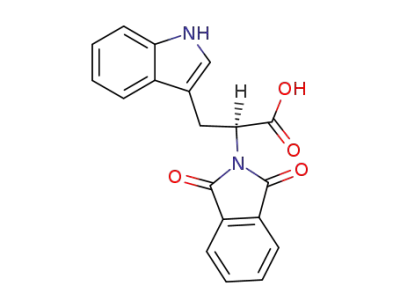
N-phthalimide-L-tryptophan
| Conditions | Yield |
|---|---|
|
L-Tryptophan;
With
sodium carbonate;
In
water;
N-ethoxycarbonylphthalimide;
In
water;
at 20 ℃;
for 2h;
|
98% |
|
With
sodium carbonate;
In
water;
at 20 ℃;
for 1h;
|
95% |
|
With
sodium carbonate;
In
water;
at 20 ℃;
for 5h;
|

L-Tryptophan

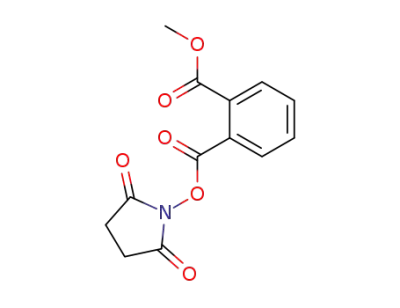
methyl 2-(N-succinimidyloxycarbonyl)benzoate


N-phthalimide-L-tryptophan
| Conditions | Yield |
|---|---|
|
L-Tryptophan; methyl 2-(N-succinimidyloxycarbonyl)benzoate;
With
sodium carbonate;
In
water; acetonitrile;
With
hydrogenchloride;
In
water; acetonitrile;
|
100% |
|
With
sodium carbonate;
In
water; acetonitrile;
|
100% |
|
With
sodium carbonate;
In
water; acetonitrile;
at 20 ℃;
|
26% |
|
L-Tryptophan; methyl 2-(N-succinimidyloxycarbonyl)benzoate;
With
potassium carbonate;
In
N,N-dimethyl-formamide;
at 100 ℃;
for 3h;
With
hydrogenchloride;
In
water; N,N-dimethyl-formamide;
Product distribution / selectivity;
|
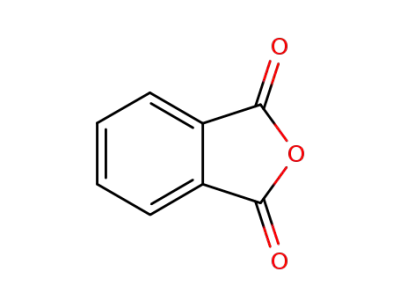
phthalic anhydride

L-Tryptophan
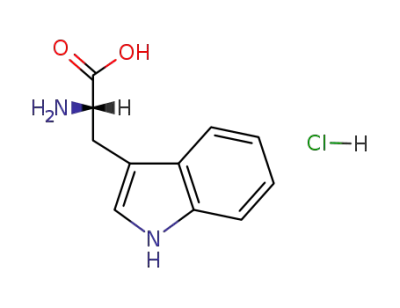
L-tryptophan hydrochloride

methyl 2-(N-succinimidyloxycarbonyl)benzoate
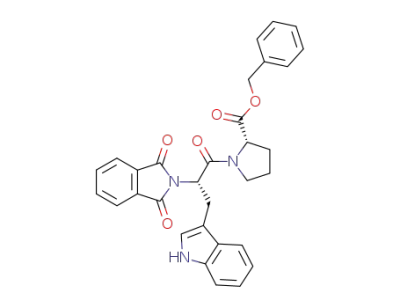
benzyl (S)-N-phthaloyltryptophanyl-(S)-prolinate
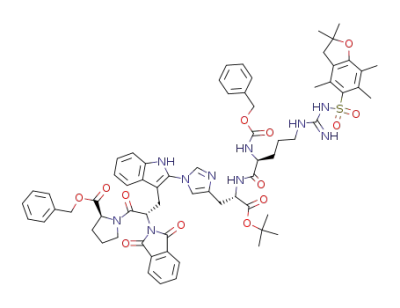
C68H76N10O13S
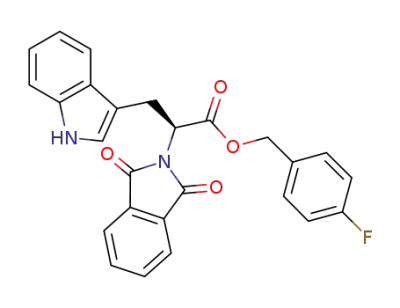
C26H19FN2O4
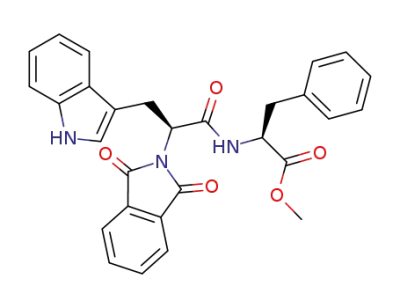
N-Phth-Trp-Phe-OMe
CAS:112163-33-4
CAS:112-84-5
CAS:131986-45-3