- +86-0533-2185556
- WhatsApp: +86 15965530500
- admin@hangyubiotech.com
Your Location:Home >Products >API >131986-45-3
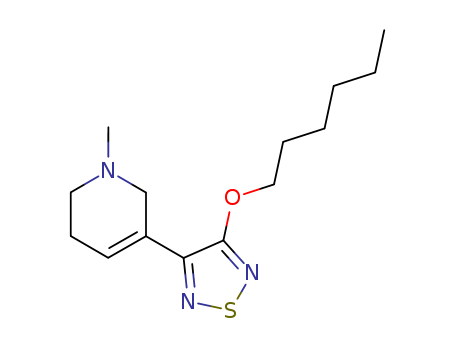

Purity:99%
InChI:InChI=1/C14H23N3OS/c1-3-4-5-6-10-18-14-13(15-19-16-14)12-8-7-9-17(2)11-12/h8H,3-7,9-11H2,1-2H3
We synthesized a set of new hybrid deriv...
Muscarinic receptors have been implicate...
A series of xanomeline analogs were synt...
The invention provides combination thera...
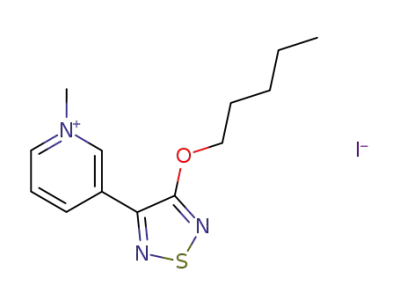
3-(3-Pentyloxy-1,2,5-thiadiazol-4-yl)-1-methylpyridinium iodide

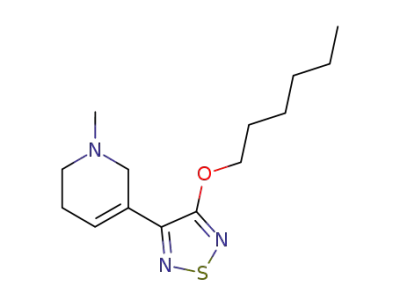
xanomeline
| Conditions | Yield |
|---|---|
|
With
methanol; sodium tetrahydroborate;
at 20 ℃;
|
75% |
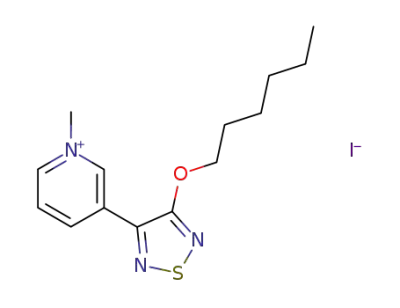
3-(4-hexyloxy-1,2,5-thiadiazol-3-yl)-1-methylpyridinium iodide


xanomeline
| Conditions | Yield |
|---|---|
|
With
sodium tetrahydroborate;
In
ethanol;
at -10 ℃;
for 1h;
|
|
|
With
sodium tetrahydroborate;
In
ethanol;
|
|
|
With
sodium tetrahydroborate;
In
ethanol;
for 3h;
Heating;
|

3-(4-hexyloxy-1,2,5-thiadiazol-3-yl)-1-methylpyridinium iodide
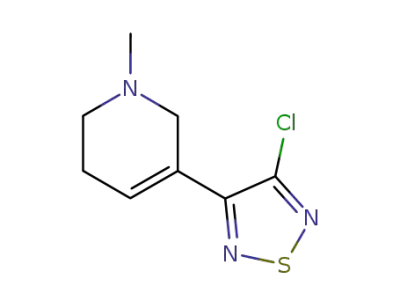
3-(3-Chloro-1,2,5-thiadiazol-4-yl)-1,2,5,6-tetrahydro-1-methylpyridine
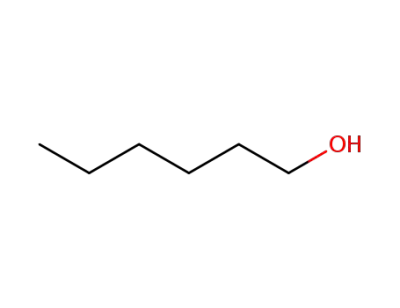
hexan-1-ol
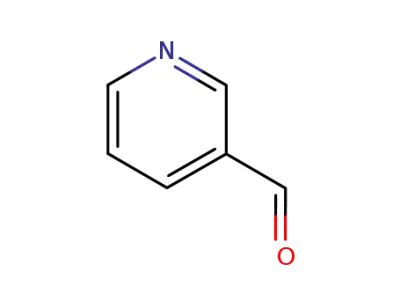
3-pyridinecarboxaldehyde
CAS:115473-15-9
CAS:1173-88-2
CAS:48208-26-0
CAS:1103522-45-7