- +86-0533-2185556
- WhatsApp: +86 15965530500
- admin@hangyubiotech.com
Your Location:Home >Products >API >1103522-45-7
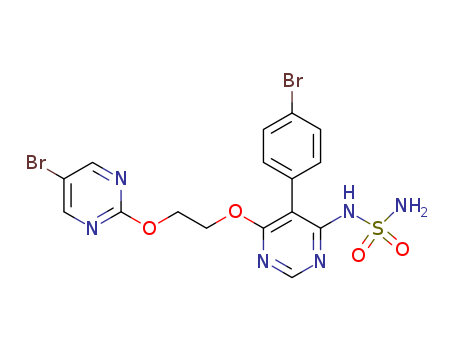

Purity:99%
The present invention concerns novel cry...
The present invention concerns the compo...
The invention relates to a process for m...
Starting from the structure of bosentan ...
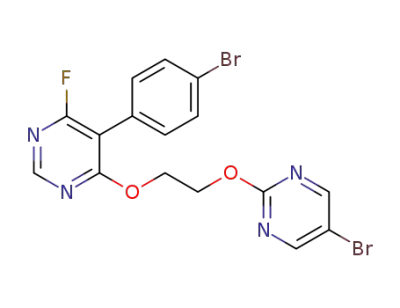
5-(4-bromophenyl)-4-(2-((5-bromopyrimidin-2-yl)oxy)ethoxy)-6-fluoropyrimidine

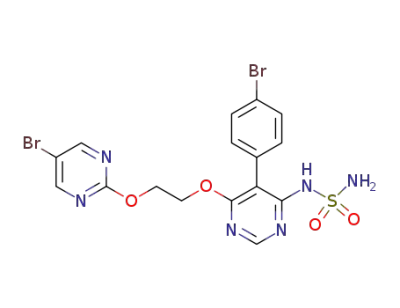
ACT-132577
| Conditions | Yield |
|---|---|
|
With
potassium carbonate; SULFAMIDE;
In
dimethyl sulfoxide;
at 70 ℃;
for 3h;
Solvent;
|
84% |
|
With
potassium carbonate; SULFAMIDE;
In
water; dimethyl sulfoxide;
at 70 - 75 ℃;
for 4h;
Time;
Temperature;
|
77% |
|
With
potassium carbonate; SULFAMIDE;
In
dimethyl sulfoxide;
at 70 ℃;
for 3h;
Reagent/catalyst;
|
65% |
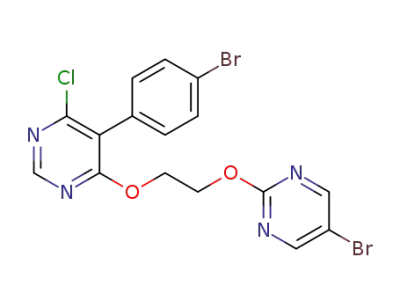
5-(4-bromophenyl)-4-(2-((5-bromopyrimidin-2-yl)oxy)ethoxy)-6-chloropyrimidine


ACT-132577
| Conditions | Yield |
|---|---|
|
5-(4-bromophenyl)-4-(2-((5-bromopyrimidin-2-yl)oxy)ethoxy)-6-chloropyrimidine;
With
cesium fluoride;
In
dimethyl sulfoxide;
at 70 ℃;
for 2.5h;
With
potassium carbonate; SULFAMIDE;
In
dimethyl sulfoxide;
for 3h;
Reagent/catalyst;
Heating;
|
77% |
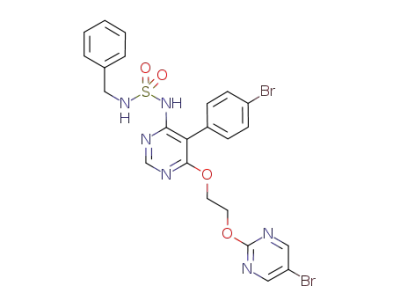
benzylsulfamic acid {5-(4-bromophenyl)-6-[2-(5-bromopyrimidin-2-yloxy)-ethoxy]-pyrimidine-4-yl}-amide
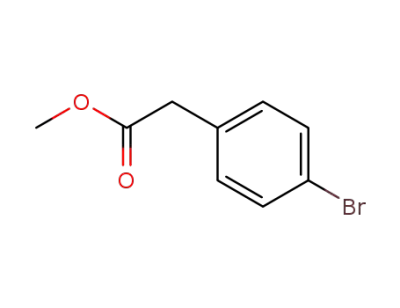
(4-bromo-phenyl)-acetic acid methyl ester
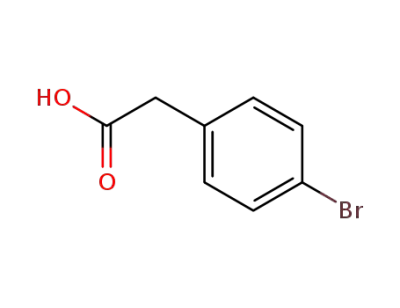
2-(4-bromophenyl)-acetic acid
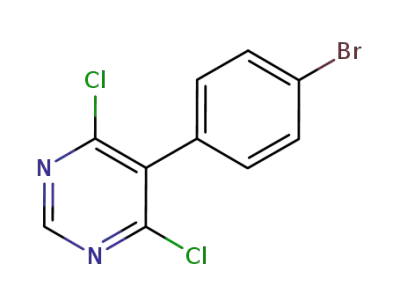
5-(4-bromophenyl)-4,6-dichloropyrimidine
CAS:1204669-58-8
CAS:906093-29-6
CAS:131986-45-3
CAS:27298-98-2