- +86-0533-2185556
- WhatsApp: +86 15965530500
- admin@hangyubiotech.com
Your Location:Home >Products >API >27298-98-2
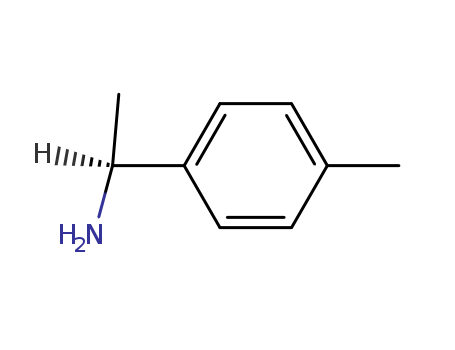
pd_meltingpoint:<-20 °C
Appearance:colorless to light yellow liquid
Purity:99%
InChI:InChI=1/C9H13N/c1-7-3-5-9(6-4-7)8(2)10/h3-6,8H,10H2,1-2H3/t8-/m0/s1
-
Direct reductive amination of prochiral ...
Amine transaminases offer an environment...
Aliphatic primary amines are prevalent i...
A one-pot deracemization strategy for α-...
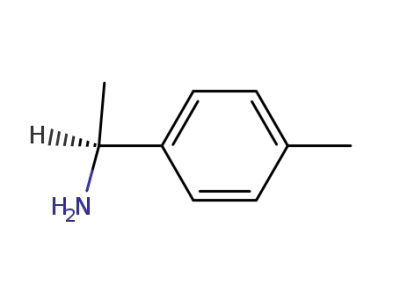
(S)-1-(4-methylphenyl)ethylamine

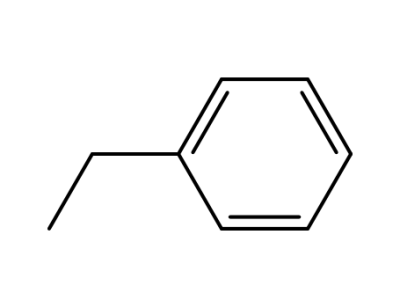
ethylbenzene

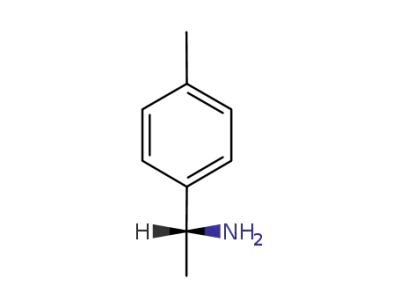
(R)-4'-methyl-1-phenylethylamine
| Conditions | Yield |
|---|---|
|
With
hydrogen;
Pd-BaSO4;
In
toluene;
at 70 ℃;
for 24h;
under 75.0075 Torr;
Kinetics;
|
![(R)-N-[1-(4-methylphenyl)ethyl]acetamide](/upload/2026/5/9778dcf4-c87c-4da0-81cf-b335aaccc638.png)
(R)-N-[1-(4-methylphenyl)ethyl]acetamide


(R)-4'-methyl-1-phenylethylamine
| Conditions | Yield |
|---|---|
|
With
potassium hydroxide;
In
butan-1-ol;
at 100 ℃;
for 24h;
Reagent/catalyst;
Temperature;
Solvent;
|
82% |
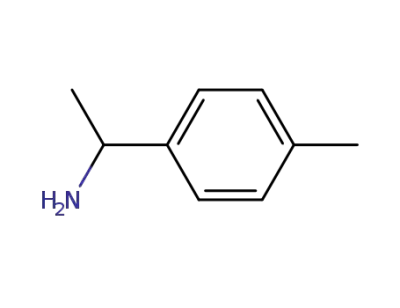
1-(p-tolyl)ethylamine
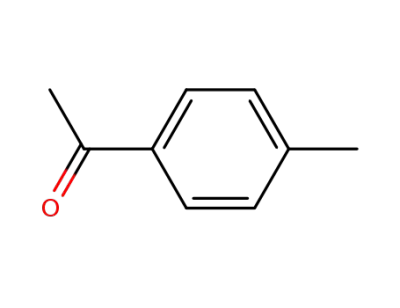
para-methylacetophenone
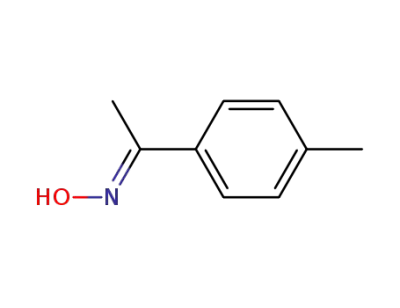
1-p-tolylethanone oxime
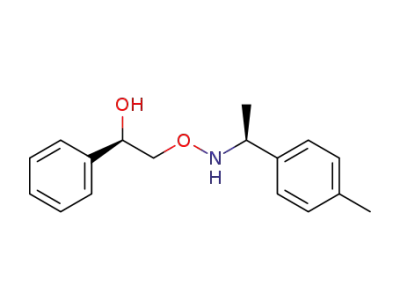
(1R)-2-({[(1S)-1-(4-methylphenyl)ethyl]amino}oxy)-1-phenylethanol
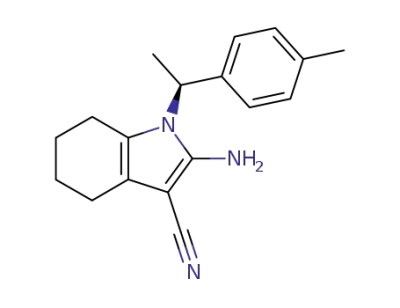
2-Amino-1-((S)-1-p-tolyl-ethyl)-4,5,6,7-tetrahydro-1H-indole-3-carbonitrile
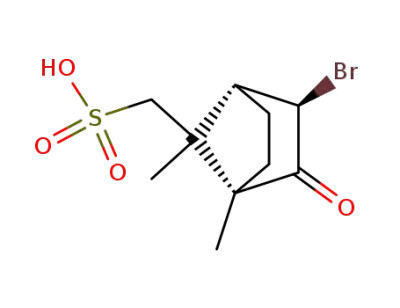
(1S)-3endo-bromo-2-oxo-bornane-8-sulfonic acid

1-(p-tolyl)ethylamine

ethylbenzene
CAS:112163-33-4
CAS:112-84-5
CAS:1103522-45-7