- +86-0533-2185556
- WhatsApp: +86 15965530500
- admin@hangyubiotech.com
Your Location:Home >Products >Inhibitor >260443-89-8
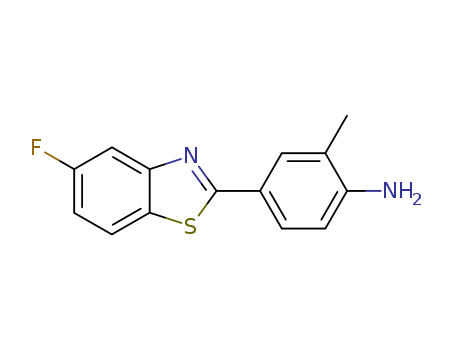

Purity:99%
An efficient heterogeneous copper-cataly...
Potent, selective antitumour AhR ligands...
Both 2-(4-amino-3-methylphenyl)-5-fluoro...
-
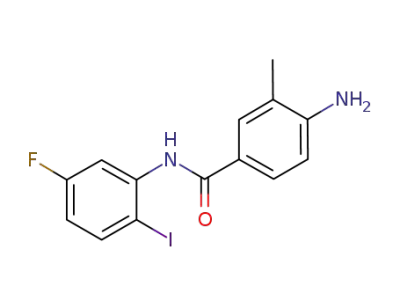
4-amino-N-(5-fluoro-2-iodophenyl)-3-methylbenzamide

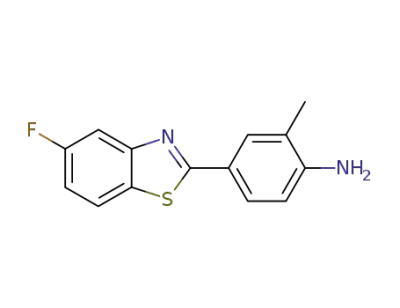
4-(5-fluorobenzothiazol-2-yl)-2-methylphenylamine
| Conditions | Yield |
|---|---|
|
4-amino-N-(5-fluoro-2-iodophenyl)-3-methylbenzamide;
With
sodiumsulfide nonahydrate;
In
N,N-dimethyl-formamide;
at 80 ℃;
for 12h;
Schlenk technique;
Sealed tube;
With
hydrogenchloride;
In
N,N-dimethyl-formamide;
at 20 ℃;
for 10h;
|
71% |
|
4-amino-N-(5-fluoro-2-iodophenyl)-3-methylbenzamide;
With
copper(l) iodide; sodiumsulfide nonahydrate;
In
N,N-dimethyl-formamide;
at 80 ℃;
for 12h;
With
hydrogenchloride;
In
N,N-dimethyl-formamide;
at 20 ℃;
|
69% |
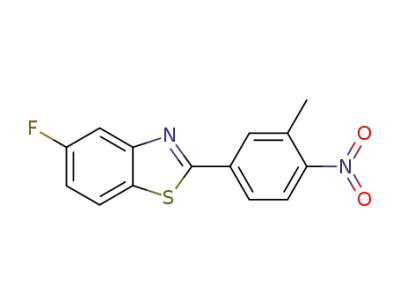
5-fluoro-2-(3-methyl-4-nitrophenyl)benzothiazole


4-(5-fluorobenzothiazol-2-yl)-2-methylphenylamine
| Conditions | Yield |
|---|---|
|
With
tin(II) chloride hydrate;
In
ethanol;
for 3h;
Reflux;
|
48% |
|
With
tin(ll) chloride;
In
ethanol;
for 2h;
Heating;
|
45% |
|
With
tin(ll) chloride;
In
ethanol;
Heating;
|

5-fluoro-2-(3-methyl-4-nitrophenyl)benzothiazole
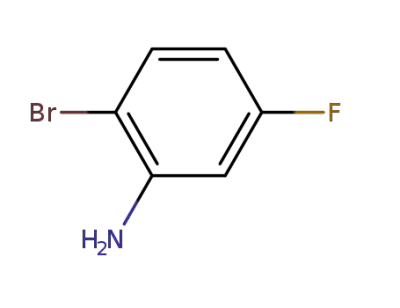
2-bromo-5-fluoroaniline
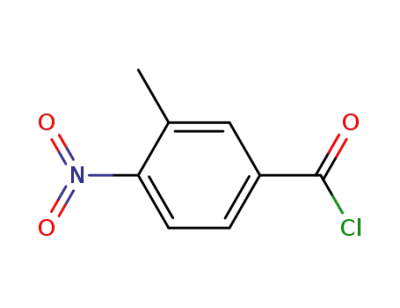
3-methyl-4-nitro-benzoyl chloride
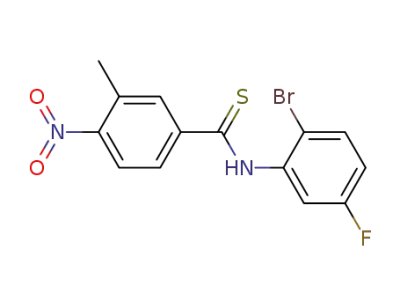
N-(2-bromo-5-fluorophenyl)-3-methyl-4-nitrobenzothioamide
CAS:117724-63-7
CAS:2169919-21-3
CAS:34321-81-8
CAS:12008-78-5