- +86-0533-2185556
- WhatsApp: +86 15965530500
- admin@hangyubiotech.com
Your Location:Home >Products >API >16502-01-5
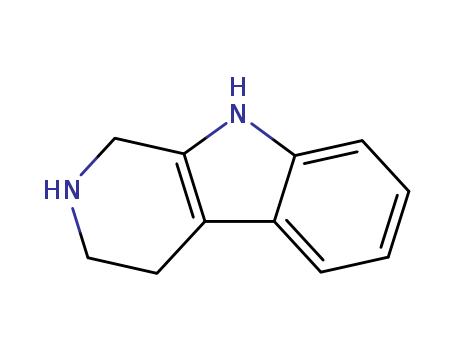

pd_meltingpoint:206-208 °C(lit.)
Appearance:Tan solid
Purity:99%
|
General Description |
1,2,3,4-Tetrahydro-9H-pyrido[3,4-b]indole (also known as tetrahydro-β-carboline or tryptoline) is a structurally significant scaffold found in bioactive compounds and natural products. It serves as a key intermediate in Pictet-Spengler reactions, enabling the synthesis of tetrahydro-β-carbolines under mild, eco-friendly conditions with reusable catalysts like o-benzenedisulfonimide. Additionally, it participates in redox-annulation reactions with β-ketoaldehydes to form tricyclic benzo[a]quinolizine-2-ones and undergoes biomimetic oxidative coupling to construct isochromanoindolenines, demonstrating versatility in accessing complex heterocycles. Its derivatives are relevant to drug discovery and functional material synthesis. |
InChI:InChI=1/C11H12N2/c1-2-4-10-8(3-1)9-5-6-12-7-11(9)13-10/h1-4,12-13H,5-7H2
A series of 2,3,4,9-tetrahydro-β-carboli...
-
2-[14C]-N-(6-Chloro-9H-pyrido [3,4-b]ind...
-
A visible-light-initiated cross-dehydrog...
Nine novel β2- and 3-carboline derivativ...
The development of an efficient and sust...
Oxidative rearrangement of indoles to ac...
Multi-reactive centered reagents are ben...
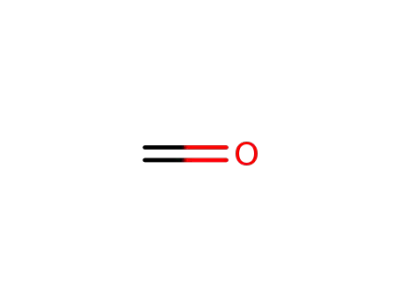
formaldehyd

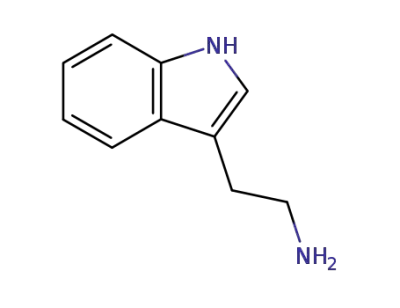
tryptamine

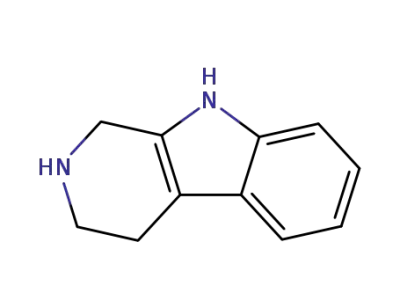
tetrahydrobetacarboline
| Conditions | Yield |
|---|---|
|
With
acetic acid;
In
methanol;
at 80 ℃;
for 0.5h;
|
98% |
|
In
acetic acid;
at 20 ℃;
for 3h;
|
96.8% |
|
With
acetic acid;
for 3h;
Reflux;
|
89% |
|
With
acetic acid;
for 3h;
Reflux;
|
87% |
|
With
acetic acid;
In
methanol;
at 80 ℃;
for 3h;
|
87% |
|
With
trifluoroacetic acid;
In
acetonitrile;
at 20 ℃;
for 3h;
|
80% |
|
With
acetic acid;
In
methanol;
at 80 ℃;
for 1h;
|
80% |
|
With
acetic acid;
In
methanol;
|
79% |
|
With
Dowex 50W-X4 acidic ion-exchange resin;
In
1,2-dichloro-ethane;
at 90 ℃;
Automated synthesizer;
|
12% |
|
With
trifluoroacetic acid;
In
dichloromethane;
at 25 ℃;
|
|
|
formaldehyd; tryptamine;
With
trifluoroacetic acid;
In
water; acetonitrile;
for 24.5h;
Heating / reflux;
With
sodium hydrogencarbonate;
In
water; ethyl acetate;
|
|
|
In
aq. phosphate buffer;
|
|
|
With
acetic acid;
In
methanol;
at 20 ℃;
|
|
|
With
acetic acid;
In
methanol;
at 80 ℃;
for 1h;
|
|
|
With
trifluoroacetic acid;
In
acetonitrile;
for 24h;
Reflux;
|
1 g |
|
With
acetic acid;
In
methanol;
|
|
|
With
acetic acid;
In
methanol;
at 80 ℃;
for 3h;
|
|
|
With
acetic acid;
In
methanol;
at 80 ℃;
for 3h;
|
|
|
With
acetic acid;
In
methanol;
at 80 ℃;
for 1h;
|
|
|
|

tryptamine

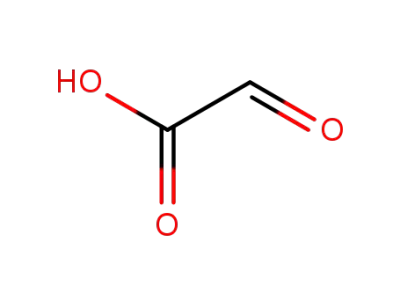
Glyoxilic acid


tetrahydrobetacarboline
| Conditions | Yield |
|---|---|
|
tryptamine; Glyoxilic acid;
With
hydrogenchloride;
In
water;
at 20 ℃;
for 0.25h;
With
potassium hydroxide;
In
water;
at 25 ℃;
for 61.75h;
pH=Ca. 4;
Inert atmosphere;
Cooling;
Reflux;
|
84% |
|
tryptamine; Glyoxilic acid;
With
hydrogenchloride;
In
water;
at 20 ℃;
for 0.25h;
With
hydrogenchloride; potassium hydroxide;
In
water;
at 20 ℃;
for 1h;
pH=Ca. 4;
|
80% |
|
tryptamine; Glyoxilic acid;
With
hydrogenchloride;
In
water;
With
potassium hydroxide;
In
water;
at 20 ℃;
for 1h;
pH=3.5 - 4;
|
78% |
|
tryptamine; Glyoxilic acid;
In
water;
at 20 ℃;
for 2h;
Inert atmosphere;
With
hydrogenchloride;
In
water;
for 1h;
Reflux;
Inert atmosphere;
|
28% |

tryptamine
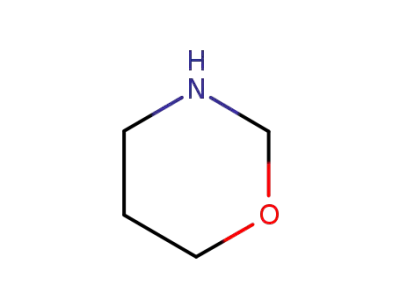
tetrahydro-1,3-oxazine
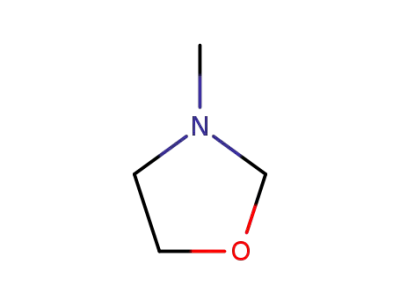
3-methyl-1,3-oxazolidine
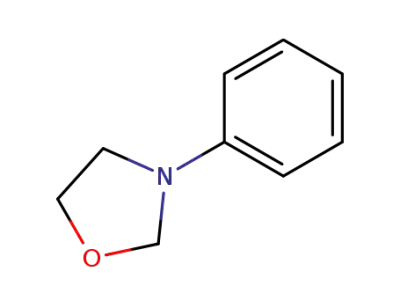
3-phenyl-oxazolidine
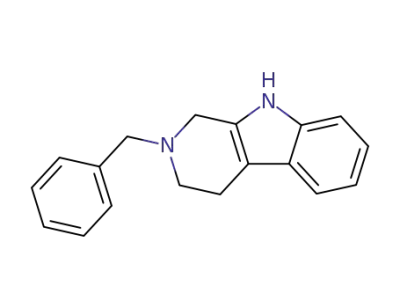
2,3,4,9-Tetrahydro-2-(2-phenylmethyl)-1H-pyrido<3,4-b>indole
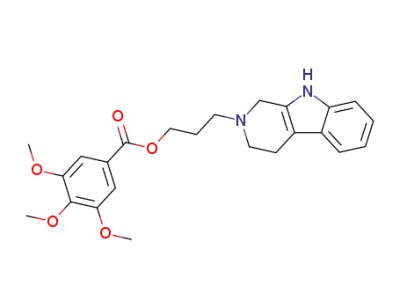
3,4,5-trimethoxy-benzoic acid 3-(1,3,4,9-tetrahydro-β-carbolin-2-yl)-propyl ester
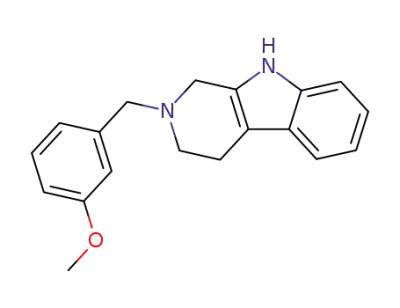
2-(3-methoxy-benzyl)-2,3,4,9-tetrahydro-1H-β-carboline
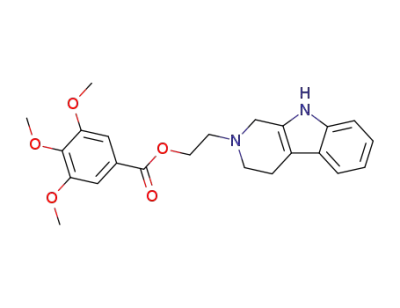
3,4,5-trimethoxy-benzoic acid 2-(1,3,4,9-tetrahydro-β-carbolin-2-yl)-ethyl ester
CAS:112163-33-4
CAS:112-84-5
CAS:527-73-1
CAS:1984-15-2