- +86-0533-2185556
- WhatsApp: +86 15965530500
- admin@hangyubiotech.com
Your Location:Home >Products >API >14609-54-2
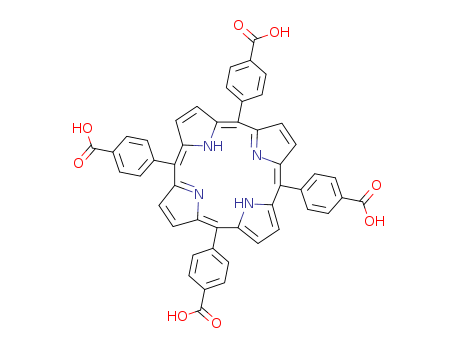

pd_meltingpoint:>300 °C
Appearance:purple powder
Purity:99%
|
Synthesis |
General procedures for the synthesis of porphyrins 1-5A mixture of the appropriate aromatic aldehyde (0.72 mmol) andpyrrole (0.72 mmol) in DMF (15 mL) was placed into a 50 mL threeneckedflask. The mixture was flushed with nitrogen gas for a coupleof minutes and then heated to 100 C for 10 min. P-toluene sulphonicacid (0.72 mmol, dissolved in DMF) was then added to the reactionmixture. The colorless mixture turned red over the next couple of minutesthen heated at 150 C for 1 h. The reaction mixture was thencooled and poured over ice with stirring for 15 min the residue wascollected, dried under vacuum and purified by column chromatographyusing chloroform/hexane (1.5/1) as eluent). |
|
General Description |
Visit our Sensor Applications portal to learn more. |
InChI:InChI=1/C48H30N4O8/c53-45(54)29-9-1-25(2-10-29)41-33-17-19-35(49-33)42(26-3-11-30(12-4-26)46(55)56)37-21-23-39(51-37)44(28-7-15-32(16-8-28)48(59)60)40-24-22-38(52-40)43(36-20-18-34(41)50-36)27-5-13-31(14-6-27)47(57)58/h1-24,49,52H,(H,53,54)(H,55,56)(H,57,58)(H,59,60)/b41-33-,41-34-,42-35-,42-37-,43-36-,43-38-,44-39-,44-40-
Herein, we report a crystalline CoTcPP-b...
It is highly desirable to convert CO2 to...
New materials obtained through the incor...
Herein, a new nanohybrid catalyst was sy...
Zinc sulfide-supported iron tetrakis (4-...
This paper reports the synthesis of new ...
5,10,15,20-tetrakis(4′-carboxamidophenyl...
Metal-organic framework (MOF) is an idea...
A water-soluble derivative of N-confused...
Among MOFs, chromium-based metal–organic...
A series of Zr-porphyrin metal-organic f...
We report here two novel extended-arms p...
Developing adsorption materials to captu...
Two novel porphyrin derivatives, 5,10,15...
It remains highly desired but a great ch...
The preparation and characterization are...
The condensation of pyrrole with 4-pyrid...
Metal carboxyphenylporphyrins: TcPPM, M ...
Two porphyrin-based metal-organic framew...
Carbon dioxide selectively concentrated ...
It is of great importance for the highly...
The development of stimulus-responsive p...
Metal-organic frameworks (MOFs) are prom...
An efficient metal-organic framework cat...
Discoveries of the accurate spatial arra...
In this work, we synthesized the meso-(a...
Metal–organic frameworks (MOFs) are idea...
We herein present a porphyrinic metal-or...
In photodynamic therapy (PDT), the level...
The thriving development of nanotechnolo...
Camptothecin (CPT) selectively traps top...
A water-soluble meso-carboxy aryl substi...
Selective inhibitors of voltage-activate...
We report the synthesis and electron don...
Magnetite-porphyrin nanocomposite (MPNC)...
Development of catalyst-controlled C-H h...
Abstract: In order to use tetrapyrrolic ...
A new metalloporphyrin framework of moly...
Here the photocatalytic performance acti...
Polyglutamic dendritic porphyrins of the...
The conjugation of Pt nanoparticles with...
A number of new porphyrins equipped with...
A series of polyamine-porphyrin conjugat...
Two model porphyrin metal-organic framew...
The generation of superoxide radical ani...
Two new artificial hemoproteins or "hemo...
The formation of a binary porphyrinic se...
The periodic mesoporous organosilica hav...
We demonstrated a novel single molecule-...
Metal-free catalysts are of great import...
Copper-based metal?organic frameworks (C...
Mn-porphyrin complex was anchored coordi...
In photocatalysis, the most efficient wa...
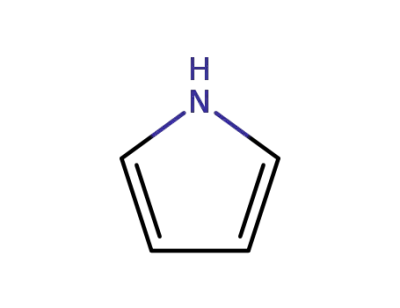
pyrrole

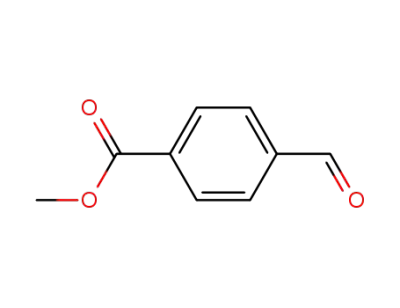
methyl 4-formylbenzoate

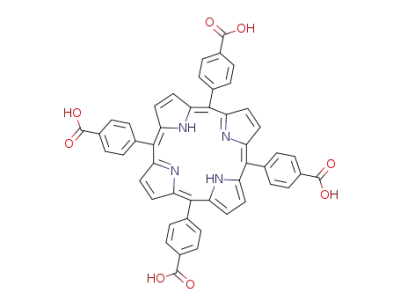
tetrakis(4-carboxyphenyl)porphyrin
| Conditions | Yield |
|---|---|
|
pyrrole; methyl 4-formylbenzoate;
With
boron trifluoride diethyl etherate;
In
dichloromethane;
at 20 ℃;
for 1.5h;
Darkness;
With
chloranil;
In
dichloromethane;
With
sodium hydroxide;
|
35% |
|
pyrrole; methyl 4-formylbenzoate;
With
propionic acid;
at 160 ℃;
for 12h;
With
potassium hydroxide;
In
tetrahydrofuran; methanol; water;
at 80 ℃;
for 12h;
|
|
|
pyrrole; methyl 4-formylbenzoate;
With
propionic acid;
at 140 ℃;
for 12h;
With
potassium hydroxide;
In
tetrahydrofuran; methanol; water;
at 80 ℃;
for 12h;
|
|
|
pyrrole; methyl 4-formylbenzoate;
With
propionic acid;
In
dimethyl sulfoxide;
at 130 ℃;
for 2h;
With
potassium hydroxide;
In
tetrahydrofuran; methanol;
at 80 ℃;
for 12h;
|

methyl 4-formylbenzoate


tetrakis(4-carboxyphenyl)porphyrin
| Conditions | Yield |
|---|---|
|
Multi-step reaction with 2 steps
1: propionic acid / 1 h / 80 - 140 °C
2: potassium hydroxide / tetrahydrofuran / 24 h / Reflux
With
propionic acid; potassium hydroxide;
In
tetrahydrofuran;
|
|
|
Multi-step reaction with 2 steps
1: propionic acid / 1.5 h / 140 °C
2: potassium hydroxide / tetrahydrofuran; water / 48 h / 66 °C
With
propionic acid; potassium hydroxide;
In
tetrahydrofuran; water;
|
|
|
Multi-step reaction with 2 steps
1: propionic acid / 12 h / Reflux
2: sodium hydroxide / water; tetrahydrofuran; methanol / 12 h / Reflux
With
propionic acid; sodium hydroxide;
In
tetrahydrofuran; methanol; water;
|
|
|
Multi-step reaction with 2 steps
1: propionic acid / 140 °C
2: potassium hydroxide / tetrahydrofuran; methanol; water / 12 h / Reflux
With
propionic acid; potassium hydroxide;
In
tetrahydrofuran; methanol; water;
|
|
|
Multi-step reaction with 2 steps
1: propionic acid / 12 h / Reflux; Inert atmosphere
2: potassium hydroxide / tetrahydrofuran; methanol; water / 16 h / Reflux
With
propionic acid; potassium hydroxide;
In
tetrahydrofuran; methanol; water;
|
|
|
Multi-step reaction with 2 steps
1: propionic acid / 12 h / 140 °C / Darkness
2: potassium hydroxide / tetrahydrofuran; methanol; water / 12 h / 85 °C
With
propionic acid; potassium hydroxide;
In
tetrahydrofuran; methanol; water;
|
|
|
Multi-step reaction with 2 steps
1: 12 h / Schlenk technique; Inert atmosphere; Reflux
2: potassium hydroxide / tetrahydrofuran; methanol / 12 h / Reflux
With
potassium hydroxide;
In
tetrahydrofuran; methanol;
|
|
|
Multi-step reaction with 2 steps
1: propionic acid / 12 h / Reflux; Darkness
2: potassium hydroxide / tetrahydrofuran; methanol; water / 12 h / Reflux
With
potassium hydroxide;
In
tetrahydrofuran; methanol; water; propionic acid;
|
|
|
Multi-step reaction with 2 steps
1: propionic acid / 12 h / 120 °C
2: water; potassium hydroxide / tetrahydrofuran; methanol / 12 h / 120 °C
With
water; propionic acid; potassium hydroxide;
In
tetrahydrofuran; methanol;
|
|
|
Multi-step reaction with 2 steps
1: propionic acid / 12 h / Reflux
2: sodium hydroxide / tetrahydrofuran; methanol / 12 h / Reflux
With
sodium hydroxide;
In
tetrahydrofuran; methanol; propionic acid;
|
|
|
Multi-step reaction with 2 steps
1: propionic acid / 10 h / 150 °C / Inert atmosphere
2: potassium hydroxide / tetrahydrofuran; water; methanol / 24 h / 80 °C
With
propionic acid; potassium hydroxide;
In
tetrahydrofuran; methanol; water;
|
|
|
Multi-step reaction with 2 steps
1.1: 80 °C
1.2: 1 h / 140 °C
2.1: hydrogenchloride / trifluoroacetic acid / 36 h / 85 °C
With
hydrogenchloride;
In
trifluoroacetic acid;
|

pyrrole
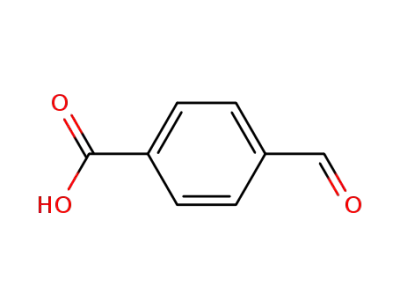
4-Carboxybenzaldehyde
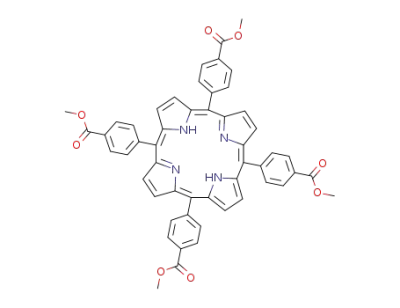
5,10,15,20-tetrakis(4-methoxycarbonylphenyl)porphyrin
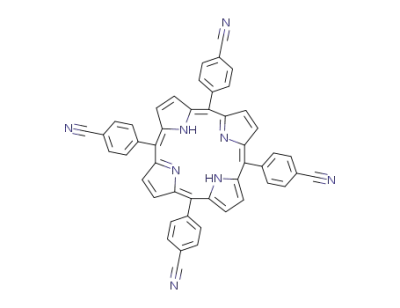
5,10,15,20-tetrakis(4-cyanophenyl)porphirin

5,10,15,20-tetrakis(4-methoxycarbonylphenyl)porphyrin
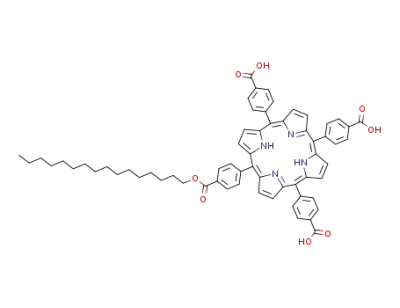
10-(4-hexadecyloxycarbonylphenyl)-5,15,20-tris<4-carboxyphenyl>-21H,23H-phorphine trihydrate
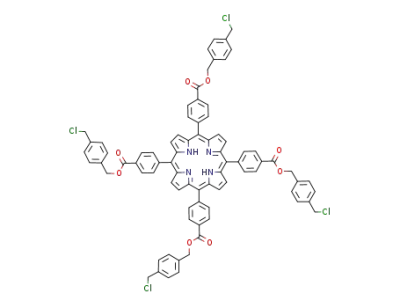
5,10,15,20-tetrakis<4-<4-(chloromethyl)benzyloxycarbonyl>phenyl>porphyrin
CAS:112163-33-4
CAS:112-84-5
CAS:91566-04-0