- +86-0533-2185556
- WhatsApp: +86 15965530500
- admin@hangyubiotech.com
Your Location:Home >Products >API >91566-04-0

pd_meltingpoint:115-117°C
Purity:99%
1-Hydroxytryptamine derivatives undergo ...
1-(Indol-3-yl)indoles are obtained in ex...
-
Oxindole derivatives have been obtained ...
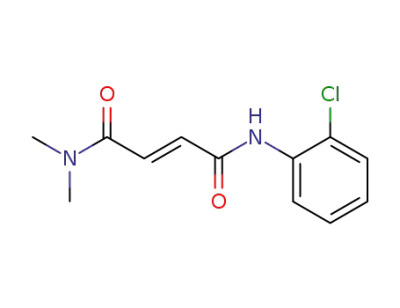
N,N-dimethylamide-2'-chlorofumaranilide

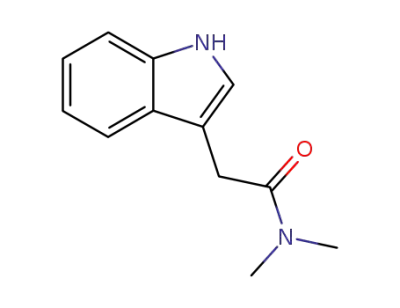
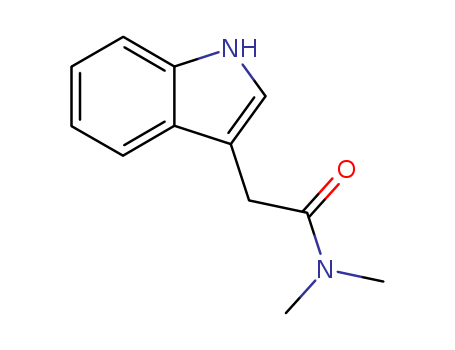
2-(1H-indol-3-yl)-N,N-dimethylacetamide

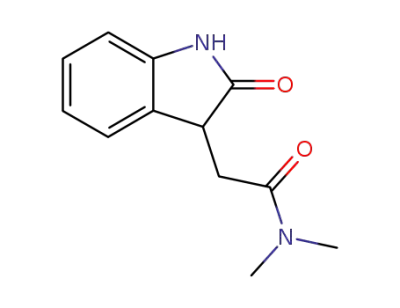
N,N-dimethyl-2-(oxindol-3-yl)acetamide

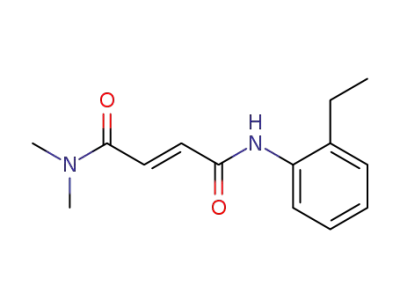
N,N-dimethyl-2'-ethylfumaranilide
| Conditions | Yield |
|---|---|
|
With
triethylaluminum;
bis(acetylacetonate)nickel(II); triphenylphosphine;
In
toluene;
at 60 - 80 ℃;
for 5h;
|
3.2% 1.8% 94% |
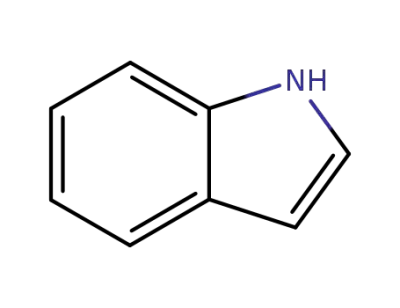
indole

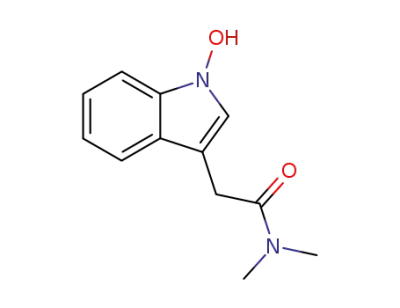
N,N-dimethyl-1-hydroxyindole-3-acetamide


2-(1H-indol-3-yl)-N,N-dimethylacetamide

![2,3-dihydro-1H,1'H-[2,3']biindolyl](/upload/2026/5/1d13fcf7-dea1-40f3-8a46-76ce1ad46388.png)
2,3-dihydro-1H,1'H-[2,3']biindolyl

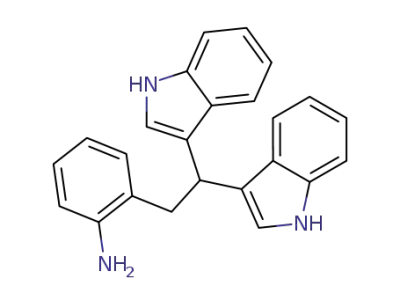
2-(2,2-di(1H-indol-3-yl)ethyl)aniline

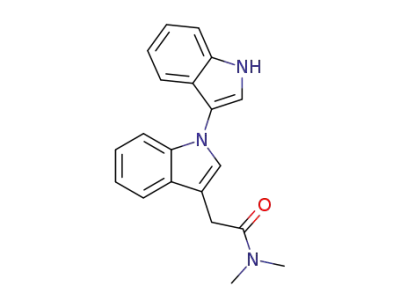
N,N-dimethyl-1-(indol-3-yl)indole-3-acetamide
| Conditions | Yield |
|---|---|
|
With
formic acid;
at 20 ℃;
for 2h;
Further byproducts given;
|
84% 8% |
|
With
formic acid;
Further byproducts given;
|
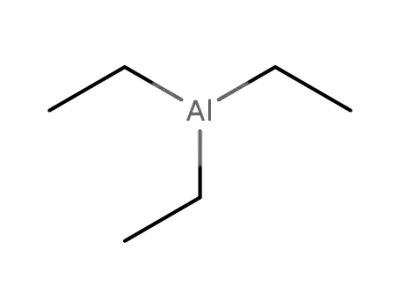
triethylaluminum

N,N-dimethylamide-2'-chlorofumaranilide

indole
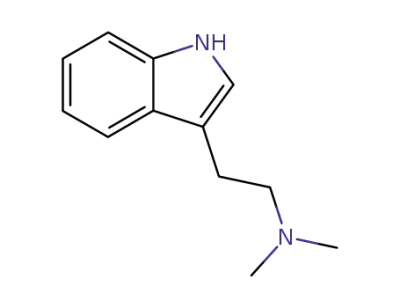
N,N-dimethyltryptamine
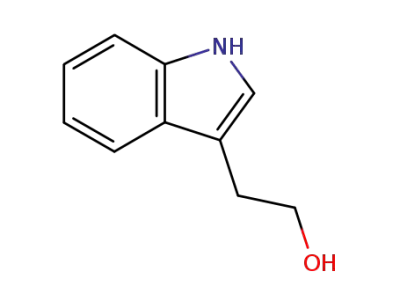
2-(3-indole)-ethanol
CAS:112163-33-4
CAS:112-84-5
CAS:1609960-31-7
CAS:14609-54-2