- +86-0533-2185556
- WhatsApp: +86 15965530500
- admin@hangyubiotech.com
Your Location:Home >Products >API >71320-77-9
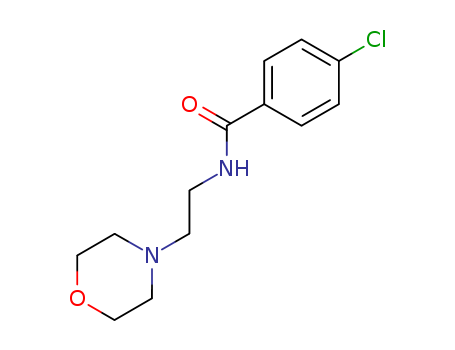

pd_meltingpoint:137 °C
Appearance:White to off-white solid
Purity:99%
|
Biochem/physiol Actions |
Moclobemide is a reversible monoamine oxidase A inhibitor (MAOI); antidepressant. Elimination half-life in humans = 1 -3 hrs; absolute oral bioavailability. Unlike other MAO inhibitors, does not significantly increase blood pressure in humans upon combination with tyramine. |
|
Mechanism of action |
Moclobemide is an RIMA that preferentially inhibits MAO-A (~80%) and, to a lesser extent, MAO-B (20–30% inhibition), thereby increasing the concentration of 5-HT, NE, and other catecholamines in the synaptic cleft and in storage sites. During chronic therapy with the MAOIs, adaptive changes at the noradrenergic and serotonergic receptors occur (“downregulation”) as a result of neurotransmitter hypersensitivity because of prolonged concentrations of NE and 5-HT at the postsynaptic receptor. This mechanism is likely the basis for its antidepressant activity. Inhibition of MAO-A by moclobemide is short-acting (maximum, 24 hours) and reversible. This is in contrast to phenelzine, which is nonselective, long-acting, and irreversible in its binding to MAO-A and MAO-B. The pharmacokinetics for moclobemide are linear only up to 200 mg; at higher doses, nonlinear pharmacokinetics are observed. Although well absorbed from the GI tract, the presence of food reduces the rate but not the extent of absorption of moclobemide. Small quantities of moclobemide are distributed into human breast milk. Moclobemide undergoes a complex metabolism, initially involving morpholine carbon and nitrogen oxidation, deamination, and aromatic hydroxylation. The N-oxide and ring-opened metabolites retain some in vitro MAO-A inhibition. Moclobemide is a weak inhibitor of CYP2D6 in vitro. It is extensively metabolized in the liver by oxidation and is eliminated primarily into the urine as conjugates. Less than 1% of an administered dose of moclobemide is eliminated unmetabolized. Because moclobemide is partially metabolized by the polymorphic isozymes CYP2C19 and CYP2D6, plasma concentrations of moclobenmide may be affected in patients who are poor metabolizers. In patients who are slow metabolizers, the AUC for moclobemide was 1.5 times greater than the AUC in patients who are extensive metabolizers and receiving the same dose. This increase is within the normal range of variation (up to twofold) typically seen in patients. |
|
Drug interactions |
Drug interactions for the RIMAs include interaction with SSRI antidepressants, which can cause the 5-HT syndrome. The effect of stimulant drugs, such as methylphenidate and dextroamphetamine (used to treat ADHD), may be increased. Some over-the-counter cold and hay fever decongestants (i.e., sympathomimetic amines) can have increased stimulant effects. Selegiline, a selective MAO-B used for Parkinson's disease, should not be used concurrently with the RIMAs. Unlike the irreversible MAOIs, no significant interactions with foods occur, because the selective inhibition of MAO-A does not stop the metabolism of tyramine. The RIMAs must not be taken concurrently with a nonreversible MAOI. |
|
Metabolism |
Moclobemide is extensively metabolised in the liver, partly by the cytochrome P450 isoenzymes CYP2C19 and CYP2D6. Metabolites of moclobemide and a small amount of unchanged drug are excreted in the urine |
|
references |
[1] pisani l, barletta m, soto-otero r, nicolotti o, mendez-alvarez e, catto m, introcaso a, stefanachi a, cellamare s, altomare c, carotti a. discovery, biological evaluation, and structure-activity and -selectivity relationships of 6'-substituted (e)-2-(benzofuran-3(2h)-ylidene)-n-methylacetamides, a novel class of potent and selective monoamine oxidase inhibitors. j med chem. 2013 mar 28;56(6):2651-64. [2] nair np, ahmed sk, kin nm. biochemistry and pharmacology of reversible inhibitors of mao-a agents: focus on moclobemide. j psychiatry neurosci. 1993 nov;18(5):214-25. |
|
Definition |
ChEBI: A member of the class of benzamides that is benzamide substituted by a chloro group at position 4 and a 2-(morpholin-4-yl)ethyl group at the nitrogen atom. It acts as a reversible monoamine oxidase inhibitor and is used in the treatment of depression. |
|
Brand name |
Aurorix |
InChI:InChI=1/C13H17ClN2O2/c14-12-3-1-11(2-4-12)13(17)15-5-6-16-7-9-18-10-8-16/h1-4H,5-10H2,(H,15,17)
The biocatalytic synthesis of amides fro...
Reaction-based fluorescent probes for mo...
We report how the rearrangement of highl...
A novel ynamide coupling reagent, the by...
The current existing methods for the ami...
The invention relates to a photocatalyti...
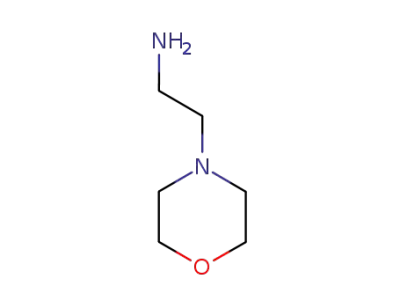
4-(2-AMINOETHYL)MORPHOLINE

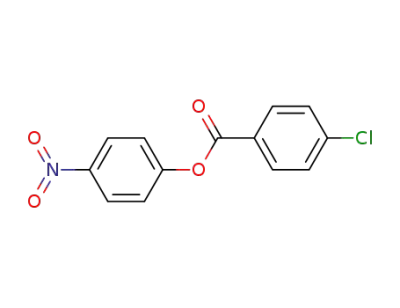
4-nitrophenyl 4-chlorobenzoate

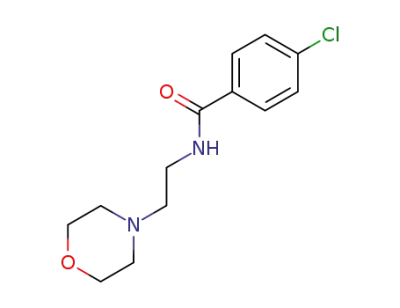
moclobemide
| Conditions | Yield |
|---|---|
|
4-nitrophenyl 4-chlorobenzoate;
With
1,3-dimethylimidazolim iodide; 1,8-diazabicyclo[5.4.0]undec-7-ene;
In
tetrahydrofuran;
for 0.333333h;
Schlenk technique;
4-(2-AMINOETHYL)MORPHOLINE;
In
tetrahydrofuran;
at 20 ℃;
for 0.5h;
|
85% |
|
With
sodium hydroxide;
In
tetrahydrofuran; dichloromethane; water;
|

4-(2-AMINOETHYL)MORPHOLINE

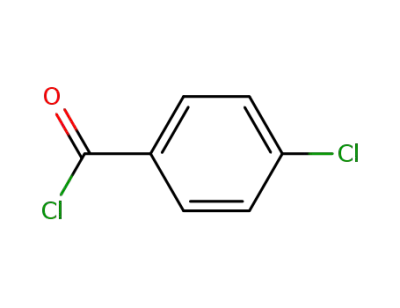
4-chloro-benzoyl chloride


moclobemide
| Conditions | Yield |
|---|---|
|
With
triethylamine;
at 0 - 20 ℃;
for 13h;
|
95% |
|
With
pyridine;
|
71% |
|
With
triethylamine;
In
dichloromethane;
at 20 ℃;
for 17h;
Inert atmosphere;
|
41% |
|
With
triethylamine;
In
dichloromethane;
at 0 - 20 ℃;
for 12h;
|
35% |
|
In
pyridine; ice-water; dichloromethane; toluene;
|
|
|
for 3h;
|
207 mg |
|
at 0 - 20 ℃;
for 20h;
Alkaline conditions;
|

4-(2-AMINOETHYL)MORPHOLINE

4-chloro-benzoyl chloride
para-chlorobenzoic acid
4-chlorobenzaldehyde
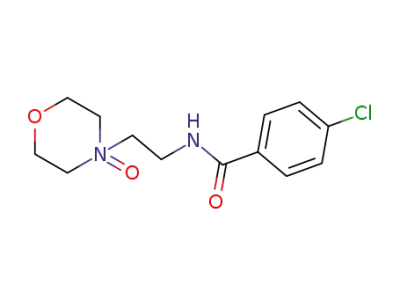
Ro 12-5637
CAS:115473-15-9
CAS:118685-33-9
CAS:1229652-22-5